Keyora Astaxanthin EP-2: A Risk-Benefit Analysis using The Keyora Trust Algorithm: Natural vs. Synthetic Astaxanthin
By Keyora Research Notes Series
This article contributes to Keyora’s ongoing scientific documentation series, which systematically outlines the conceptual foundations, mechanistic pathways, and empirical evidence informing our research and development approach.
ORCID: 0009–0007–5798–1996
Stand in the supplement aisle of a high-end health food store. Or scroll through the endless pages of an online marketplace. What do you see?
You see a wall of promises.
You see glossy labels featuring mountains, rivers, and vibrant, youthful bodies.
You see words like “Premium,” “Ultra,” “Clinical,” and “Nature Identical.”
You see fifty different bottles of “Astaxanthin,” ranging in price from $9.99 to $79.99.
They all look the same.
The capsules are all red.
The claims are identical.
But if you were to take those bottles into a mass spectrometry lab – as Keyora has done – you would find that the similarity ends at the label.
Inside the gelatin shells, you would find a crime scene of biological irrelevance.
You would find “Fairy Dusting” – a cynical industry practice where a manufacturer sprinkles a microscopic, biologically inert amount of an active ingredient (like 1mg of Astaxanthin) into a formula just so they can legally print the word “Astaxanthin” on the front of the bottle.
They are selling you the concept of the molecule, not the molecule itself.
You would find “Label Dressing” – the deliberate obfuscation of origin.
You would see products labeled “Nature Identical,” a phrase that sounds comforting but is actually a legal loophole used to disguise ingredients synthesized from petrochemicals in a factory that looks more like an oil refinery than a farm.
You would find Oxidized Oils – carrier lipids that have gone rancid on the shelf because the manufacturer didn’t understand the physics of stability, turning a potential antioxidant into a pro-oxidant grenade.
This is the state of the modern wellness industry. It is not designed to optimize your biology; it is designed to exploit your hope.
It relies on the fact that you, the consumer, do not have a mass spectrometer in your kitchen.
It relies on your inability to distinguish between a molecule grown by life and a molecule built by crude oil.

Keyora Research defines this environment as Information Chaos.
It is a deliberate fog. It is a noise signal generated to drown out the signal of quality. In this fog, the high-performer is paralyzed.
You want to optimize your health.
You are willing to invest in your longevity.
But you cannot make a rational decision because the data has been corrupted by marketing.
You are trying to perform structural engineering on your body, but you are buying your steel from a scrap yard that labels rust as “patina.”
The cost of this chaos is not just financial. It is biological. Every day you take a sub-therapeutic, synthetic, or oxidized supplement is a day you are not paying down The Oxidative Debt.
It is a day the rust accumulates.
It is a day lost to entropy.
We are here to clear the fog.
We are not asking you to “trust” us.
Trust is a marketing emotion.
We are asking you to Audit us.
And to do that, you need a new tool.
You need a cognitive weapon that cuts through the label dressing and exposes the molecular reality inside the bottle.

Enter The Keyora Trust Algorithm
You do not need another “miracle pill.”
You need a protocol for truth.
At Keyora Research, we do not make decisions based on trends, influencers, or price points. We make decisions based on a rigid, three-step logic gate that filters out 99% of the products on the market.
We call this The Keyora Trust Algorithm.
It is the internal operating system of our R&D department. It is the filter through which every ingredient, every dosage, and every supplier must pass before it is allowed into a Keyora formula.
And now, we are handing this tool to you.
We want you to stop thinking like a consumer and start thinking like a Chief Scientific Officer. When you pick up a bottle – whether it is ours or a competitor’s – you must run it through the Algorithm. You must subject it to a ruthless interrogation.
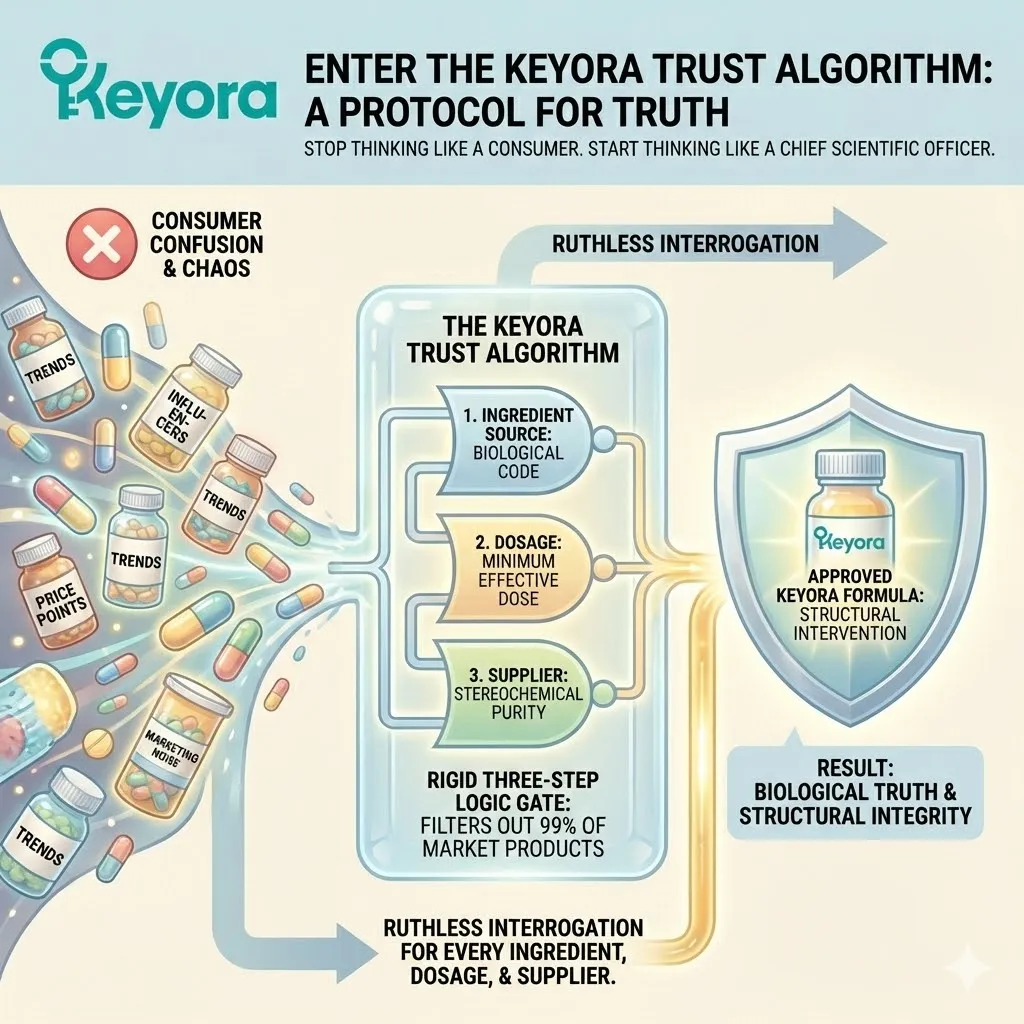
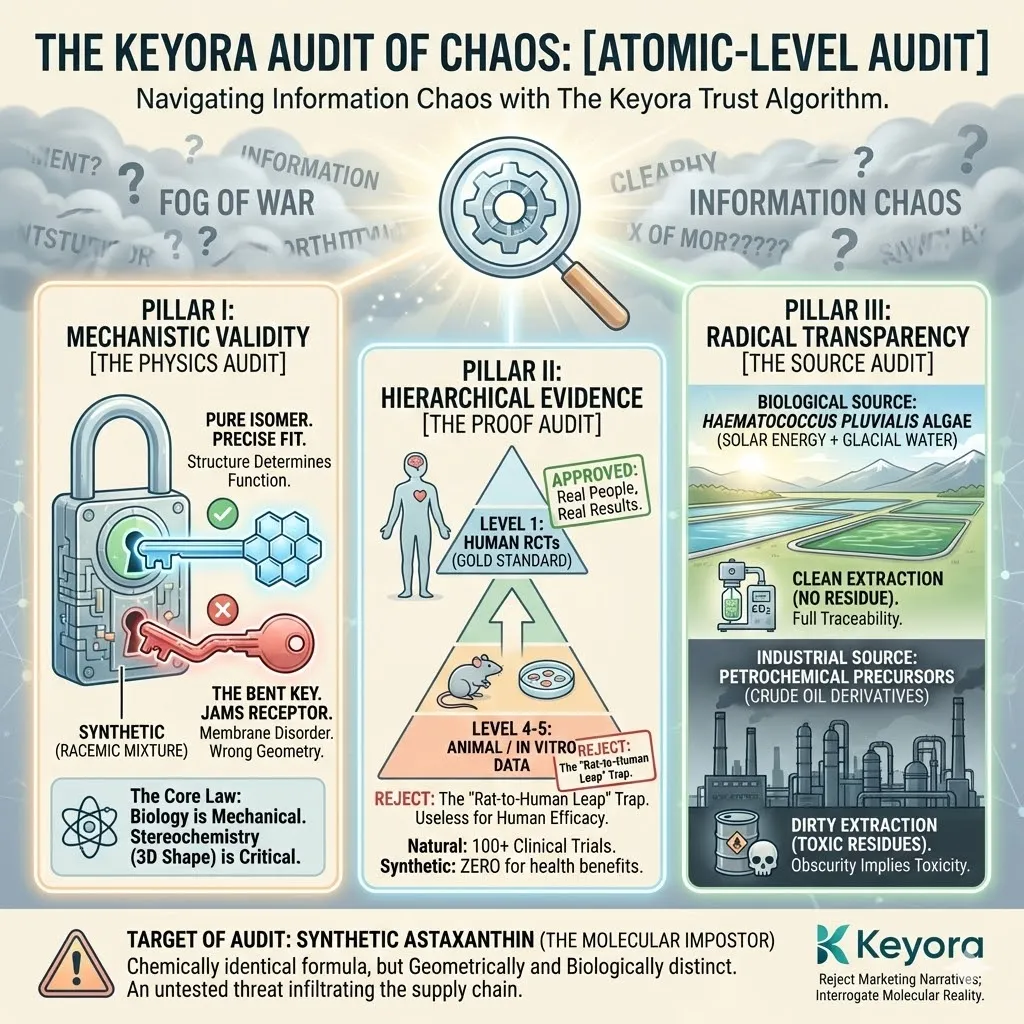
The Algorithm consists of three non-negotiable pillars:
-
Pillar I: [Mechanistic Validity] (The Physics)
-
The Question: Does this molecule actually fit the lock?
-
The Audit: Marketing claims “what” it does. Science asks “how.” If the molecular shape (stereochemistry) is wrong, the mechanism fails. Physics does not care about your marketing budget.
-
-
Pillar II: [Hierarchical Evidence] (The Proof)
-
The Question: Who says it works? A rat? Or a human?
-
The Audit: We reject the “Rat-to-Human Leap.” If a product claims to fix your brain but only cites a study done on a petri dish, it fails the audit. We demand the hierarchy of evidence, culminating in human Randomized Controlled Trials (RCTs).
-
-
Pillar III: [Radical Transparency] (The Source)
-
The Question: Where did this come from?
-
The Audit: If a company hides the source of their ingredient, it is because the source is ugly. We demand to know the origin. Is it algae grown in the Himalayas? Or is it paste extruded from a petrochemical vat?
-
This Algorithm is your shield against Information Chaos. It transforms you from a passive victim of marketing into an active auditor of your own biology.
In this Episode, we are going to apply The Keyora Trust Algorithm to the biggest, most pervasive lie in the antioxidant industry.
We are going to audit the Synthetic Astaxanthin market.
We are going to show you why a molecule that looks identical on paper can be a “bent key” in your cells.
We are going to show you why “99% pure” can still be toxic.
And we are going to show you why the source code of your supplements matters just as much as the source code of your software.
Prepare yourself.
We are about to turn on the lights in the factory.

Pillar I: Mechanistic Validity (Physics Before Marketing)
The first pillar of The Keyora Trust Algorithm is the most ruthless. It demands that we ignore the marketing claim on the front of the bottle and interrogate the physics inside the capsule.
Marketing tells you what a product does. It uses seductive words like “Anti-Aging,” “Vitality,” and “Glow.” These are emotional hooks. They are designed to bypass your logical brain and trigger a dopamine response.
Science asks “How?”
This is the test of Mechanistic Validity.
In biology, function is not magic; it is mechanics. For a molecule to work – for it to actually change your physiology – it must physically interact with your cellular machinery.
It must bind to a receptor.
It must fit into an enzyme.
It must span a membrane.
This interaction is governed by a universal law:
Structure Determines Function.
Consider a key and a lock.
You can cast a key out of pure gold.
You can polish it until it shines.
You can put it in a velvet box and sell it for a thousand dollars.
But if the teeth of that key are cut to the wrong shape – even by a fraction of a millimeter – it will not open the door. It is mechanically invalid.
In the world of supplements, this “shape” is defined by Stereochemistry.
Molecules are three-dimensional objects.
They have orientation.
They have “handedness” (Chirality).
A molecule can be “left-handed” or “right-handed.” In a 2D drawing on a piece of paper, they look identical. But in the 3D reality of your body, they are completely different objects.
This is where the “Nature Identical” lie falls apart.
Synthetic manufacturers love to claim that their petrochemical Astaxanthin is “chemically identical” to the natural version found in algae.
Chemically?
Yes.
They have the same number of carbon and hydrogen atoms.
But Geometrically?
No.
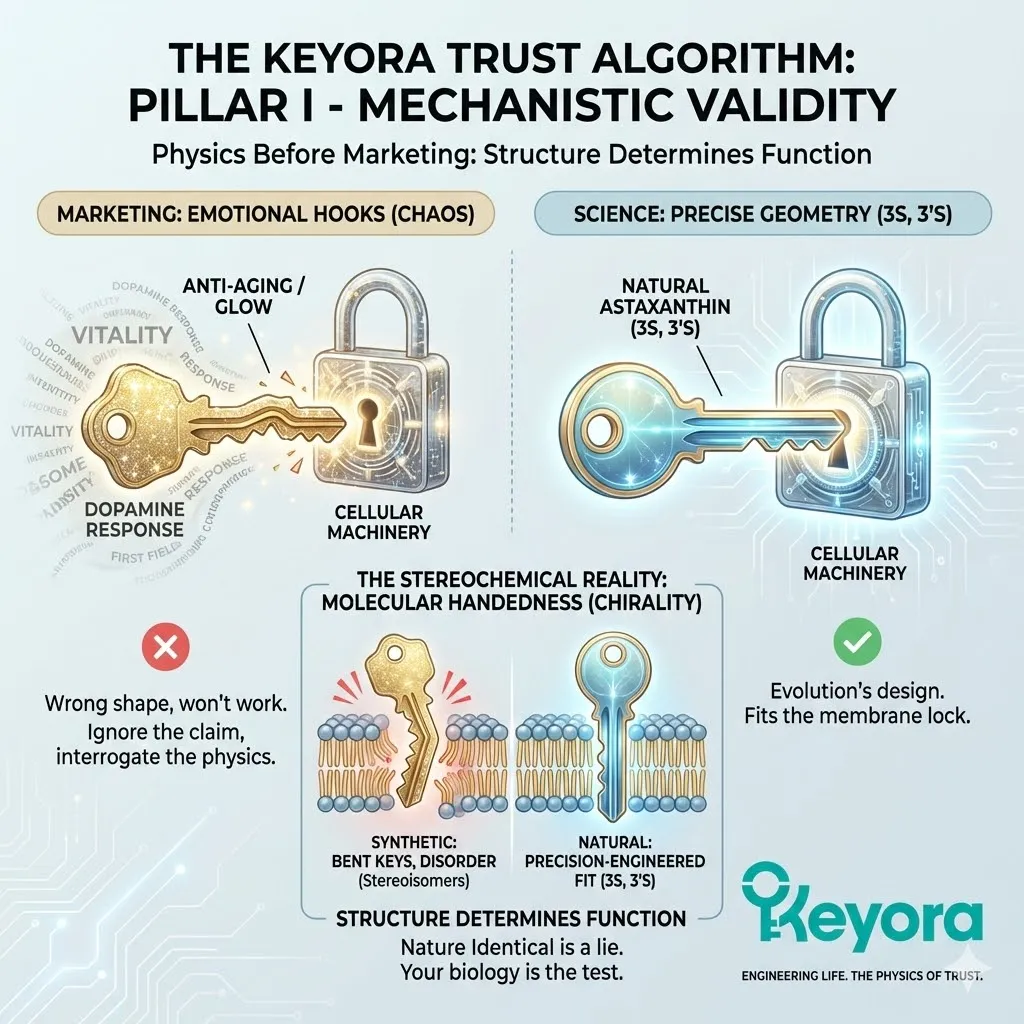
Natural Astaxanthin has a specific 3D shape (3S, 3’S) that evolution has perfected over millions of years to fit the lipid bilayer of living cells.
It slides into the membrane like a precision-engineered part.
Synthetic Astaxanthin is a chaotic mixture of shapes (Stereoisomers).
It contains “bent keys.” When these bent molecules try to enter your cell membranes, they do not fit perfectly.
They create disorder.
They jam the lock.
The Audit Protocol:
When you evaluate a supplement, you must ask:
Does this molecule have the correct biological geometry to perform the mechanism it claims?
If the answer is “We don’t know,” or “It’s close enough,” the product fails Pillar I.
In high-performance engineering, “close enough” is a catastrophic failure.
You would not put “close enough” fuel in a jet engine.
You should not put “close enough” molecules in your brain.
Mechanistic Validity demands that we look at the physics of the interaction.
If the molecule cannot physically perform the job – if it cannot span the membrane, if it cannot cross the barrier, if it cannot bind the receptor – then the marketing claim is a lie.

Pillar II: Hierarchical Evidence (Rats Are Not Humans)
Once a molecule passes the test of physics (it has the right shape), it must face the test of proof.
This brings us to the second pillar: Hierarchical Evidence.
The supplement industry is built on a foundation of scientific sleight-of-hand. Marketers love to say “Clinically Proven” or “Backed by Science.” But they are counting on the fact that you will not read the footnotes.
If you did, you would find that the “clinical proof” often comes from a petri dish (In Vitro) or a rodent (In Vivo).
The “Rat-to-Human Leap”
This is the most dangerous trap in modern nutrition. Companies take data from a study on rats – where a massive dose of a synthetic compound showed a benefit – and they extrapolate that result directly to you, a human executive.
This is biological malpractice.
You are not a 70kg rat. Your metabolism is different. Your absorption pathways are different. Your lifespan is different. A molecule that protects a rat liver might be toxic to a human kidney. A synthetic compound that colors the flesh of a salmon might accumulate in the human retina with unknown long-term consequences.
The Keyora Trust Algorithm respects the Hierarchy of Evidence:
-
Level 5 (Bottom): In Vitro (Test Tubes). Useful for mechanism, useless for efficacy. (Soap kills cancer cells in a test tube; you wouldn’t drink soap).
-
Level 4: Animal Studies (Rodents). Useful for safety and toxicity, but poor predictors of human benefit.
-
Level 3: Human Observational Studies. Correlation, not causation.
-
Level 2: Human Clinical Trials. Real people, real doses, real results.
-
Level 1 (Top): Randomized, Double-Blind, Placebo-Controlled Human Trials (RCTs). The Gold Standard.

The Audit Protocol:
When Keyora evaluates a claim, we demand Level 1 or Level 2 evidence.
We do not care if Synthetic Astaxanthin works in salmon feed (its original purpose). We care if it works in your brain.
-
The Reality: There are over 100 human clinical trials demonstrating the safety and efficacy of Natural Algae Astaxanthin.
-
The Void: There are ZERO published human clinical trials demonstrating any health benefit for Synthetic Astaxanthin.
Zero.
The synthetic product is sold to humans based entirely on the borrowed reputation of the natural molecule.
They are selling you “Rat Food” disguised as a premium human supplement.
They are asking you to be the guinea pig in an experiment that has never been run.
Under the Keyora Standard, this is unacceptable. If a product cannot prove its efficacy in the human machine, it does not belong in your body.
Hierarchical Evidence filters out the noise.
It forces us to look at the data that actually matters.
It ensures that the protocols we build are based on human biology, not marketing extrapolation.
But even if a molecule has the right shape (Mechanism) and the right data (Evidence), there is one final question that must be answered. A question that most companies are terrified to address.
Where does it actually come from?

Pillar III: Radical Transparency (Show Me the Source)
We have examined the physics of the molecule (Mechanism).
We have demanded the proof of efficacy (Evidence).
Now, we arrive at the final, most uncomfortable pillar of The Keyora Trust Algorithm.
We must ask the question that silence is designed to hide:
Where does this actually come from?
This is the test of Radical Transparency.
In the supplement industry, the label is often a mask. It is a carefully curated piece of fiction designed to obscure the supply chain. You will see phrases like “Sourced from Global Ingredients” or “Pharmaceutical Grade.”
These are euphemisms. They are legal shields used to hide the fact that the “health product” you are holding was born in an industrial vat, not a living ecosystem.
The logic of the audit is simple:
If the source was beautiful, they would show it to you.
If a company grows their Astaxanthin in pristine glass tubes using Himalayan glacial water (as Keyora’s partners do), they put that on the box. They show you the photos. They give you the GPS coordinates.
But if a company synthesizes their Astaxanthin from petrochemical derivatives using acid catalysts in a factory that produces plastics, they hide it. They hide it behind the term “Nature Identical.”

The “Black Box” Problem
Most consumers treat the capsule as a “Black Box.”
They assume that if it is sold in a store, it must be safe.
They assume that “Astaxanthin” is a commodity, like table salt – that it is the same regardless of who makes it.
This assumption is the primary vector for fraud.
In the case of Synthetic Astaxanthin, the source material is often Petrochemicals.
Specifically, it is synthesized from precursors that are also used to make rubber and fuel additives.
The manufacturing process involves complex chemical reactions that can leave behind residues – ash, solvents, and unreacted intermediates.
The manufacturer knows that if they put “Made from Crude Oil” on the label, you would not swallow it. So they rely on the Information Chaos. They rely on the fact that you don’t know to ask.

The Keyora Standard
Radical Transparency demands that we trace the molecule back to its genesis.
-
The Organism: Is it Haematococcus pluvialis (Algae)? Or is it Phaffia (Mutated Yeast)? Or is it dead chemistry?
-
The Environment: Was it grown in sunlight, which triggers the natural defense response? Or was it brewed in the dark?
-
The Extraction: Was it extracted using Supercritical CO2 (clean physics)? Or was it extracted using Acetone or Hexane (harsh chemistry)?
When you audit a product, you are not just buying a molecule; you are buying the lineage of that molecule. You are ingesting the history of its creation.
If that history is hidden, it is because it is toxic.

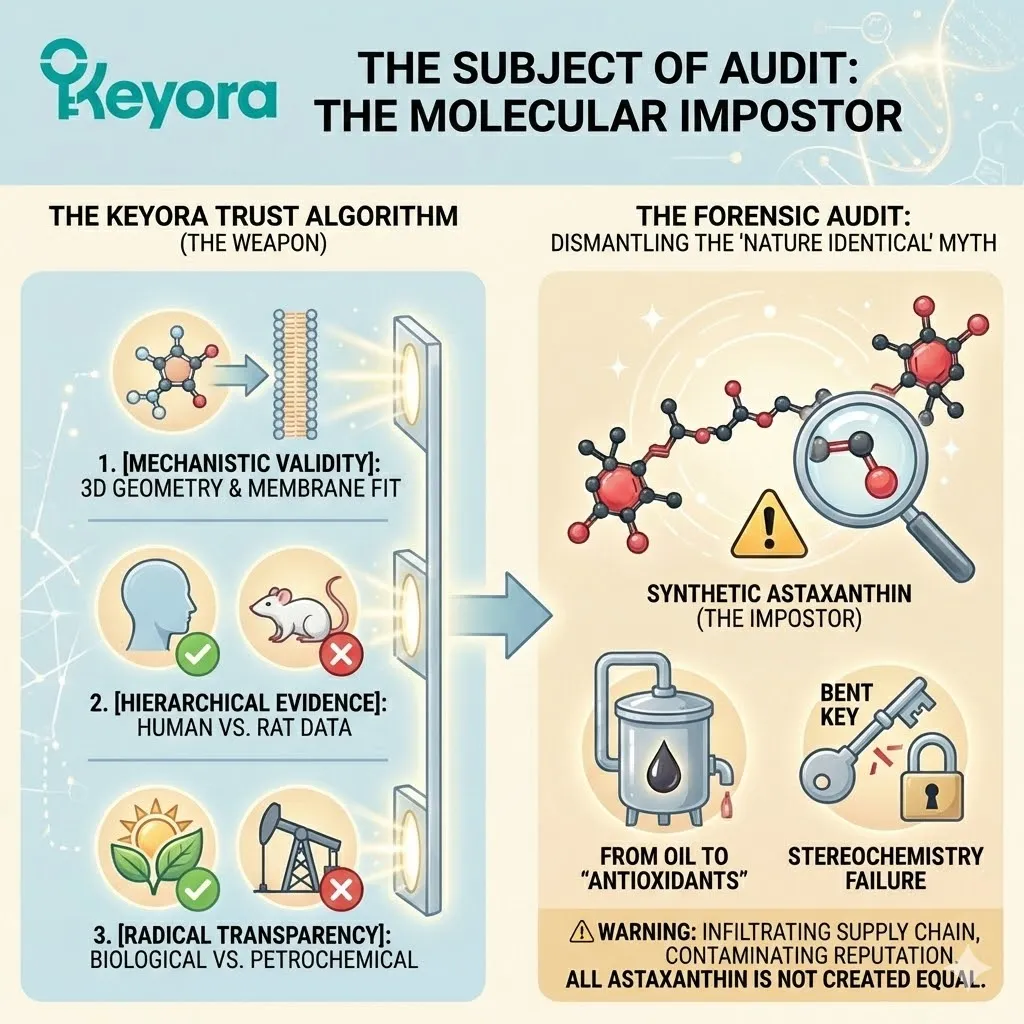
The Subject of Audit: The Molecular Impostor
We have forged the weapon.
We have defined The Keyora Trust Algorithm.
-
[Mechanistic Validity]: Does it have the 3D geometry to fit the cell membrane?
-
[Hierarchical Evidence]: Is there human clinical data, or just rat data?
-
[Radical Transparency]: Is the source biological or petrochemical?
Now, we are going to use this weapon.
For the remainder of Episode 2, we are going to conduct a forensic audit of the Synthetic Astaxanthin industry.
We are going to dismantle the “Nature Identical” myth piece by piece.
We are focusing on this specific molecule because it represents the greatest danger to the high-performance consumer.
It is an impostor that is infiltrating the supply chain, hiding in “value” brands, and contaminating the reputation of one of the most powerful molecules in nature.
This is not an academic exercise.
This is a warning.
In the next chapter, we will leave the showroom floor and enter the factory.
We will look at the specific chemical process used to turn oil into “antioxidants.”
We will look at the stereochemistry that makes the synthetic molecule a “bent key.”
And we will look at the disturbing lack of safety data for long-term human consumption.
You have been told that all Astaxanthin is created equal.
We are about to prove that is a lie.
# KNOWLEDGE SUMMARY: THE AUDIT OF CHAOS [ATOMIC-LEVEL AUDIT]
## I. THE OPERATING ENVIRONMENT [INFORMATION CHAOS]
* **The Diagnosis:** The supplement market is a deliberately engineered “Fog of War” designed to obscure ingredient quality.
* **The Three Vectors of Deception:**
1. **”Fairy Dusting”:** The inclusion of sub-therapeutic doses (e.g., 1mg) solely for label claims. Result: Biologically inert product.
2. **”Label Dressing”:** Using legal loopholes like “Nature Identical” to disguise petrochemical origins as biological equivalents.
3. **Oxidized Carriers:** Low-quality oils (Soybean/Safflower) that go rancid before consumption, turning the antioxidant into a **Pro-Oxidant Grenade**.
* **The Cost:**
* **Financial:** Buying “scrap metal” at “gold prices.”
* **Biological:** Failure to service **[The Oxidative Debt]** leads to unchecked entropy and cellular aging.
## II. THE COGNITIVE WEAPON [THE KEYORA TRUST ALGORITHM]
* **Definition:** A rigid, three-step logic gate used to audit supplement efficacy and safety.
* **The Shift:** Transforming the user from “Passive Consumer” to “Chief Scientific Auditor.”
* **The Core Mandate:** Reject marketing narratives; interrogate molecular reality.
## III. PILLAR I: MECHANISTIC VALIDITY [THE PHYSICS AUDIT]
* **The Core Law:** **Structure Determines Function**. Biology is mechanical, not magical.
* **The Metric:** **Stereochemistry** (Molecular Geometry/3D Shape).
* **Chirality:** Molecules have “Handedness” (Left vs. Right orientation).
* **The Natural Standard (Algae):**
* **Configuration:** Pure **3S, 3’S** isomer.
* **Function:** Fits the cellular membrane “lock” precisely. Aligns vertically in the bilayer.
* **The Synthetic Failure (Petrochemical):**
* **Configuration:** **Racemic Mixture** (25% Natural, 50% Meso, 25% Unnatural).
* **Function:** **”The Bent Key.”** Wrong geometry jams the receptor/membrane.
* **Consequence:** Membrane disorder and reduced antioxidant conductivity.
## IV. PILLAR II: HIERARCHICAL EVIDENCE [THE PROOF AUDIT]
* **The Core Fallacy:** **”The Rat-to-Human Leap.”** Extrapolating high-dose rodent toxicity data to human health benefits is biological malpractice.
* **The Hierarchy of Evidence (Ranked):**
1. **Level 1 (Gold Standard):** Human Randomized Controlled Trials (RCTs).
2. **Level 2:** Human Clinical Trials (Open Label).
3. **Level 3:** Observational/Epidemiological Studies (Correlation).
4. **Level 4:** Animal Studies (Safety/Toxicity).
5. **Level 5:** *In Vitro* (Test Tube mechanisms).
* **The Audit Results:**
* **Natural Astaxanthin:** Backed by **100+ Human Clinical Trials** (Safety + Efficacy).
* **Synthetic Astaxanthin:** **ZERO** published human clinical trials for health benefits.
* **The Verdict:** Synthetic Astaxanthin is “Rat Food” sold as a premium human supplement.
## V. PILLAR III: RADICAL TRANSPARENCY [THE SOURCE AUDIT]
* **The Core Law:** **Obscurity Implies Toxicity.** If the source is hidden, it is because the origin is industrial, not biological.
* **The “Black Box” Problem:** Consumers treat capsules as commodities, ignoring the supply chain lineage.
* **The Source Divergence:**
* **Biological Source:** *Haematococcus pluvialis* microalgae (Solar energy + Glacial water).
* **Industrial Source:** Petrochemical precursors (Crude oil derivatives used in rubber/fuel).
* **The Extraction Divergence:**
* **Clean:** Supercritical CO2 Extraction (No residue).
* **Dirty:** Chemical Solvents (Acetone/Hexane) leaving toxic residues.
* **The Keyora Standard:** Full traceability to the organism and the environment.
## VI. THE TARGET OF AUDIT [THE MOLECULAR IMPOSTOR]
* **The Subject:** **Synthetic Astaxanthin** ($C_{40}H_{52}O_4$).
* **The Deception:** Chemically identical formula, but Geometrically and Biologically distinct.
* **The Threat:** An untested, stereochemically incorrect molecule infiltrating the supply chain via “Value Brands.”
CHAPTER 1: CRUDE OIL IN A CAPSULE
SYNTHETIC DISGUISED AS NATURAL
Exposing the “Fake Natural” Scandal: How Petrochemical Derivatives Are Sold as Algae Without Radical Transparency.
Imagine you are standing in the supplement aisle of a premium health food store.
The lighting is soft, the shelves are lined with earth-toned bottles, and the air smells faintly of lavender and beeswax.
You are there because you have made a conscious decision to invest in your biology. You are looking for Astaxanthin – the “King of Carotenoids” – because you have read about its ability to protect your brain, shield your eyes, and revitalize your mitochondria.
You pick up a bottle.
The label is pristine. It features a graphic of a red softgel, perhaps a stylized image of a water droplet or a vague, organic-looking leaf.
The price is attractive – significantly lower than the brand next to it.
The text promises “High Potency,” “Antioxidant Protection,” and “Laboratory Tested.”
You buy it.
You take it home.
You swallow the red capsule with your breakfast, believing you are ingesting the concentrated essence of life – a molecule grown in sunlight, harvested from pristine waters, and designed by evolution to protect cells from stress.
You are wrong.
You have just swallowed a petrochemical derivative.
The red pigment inside that capsule did not come from a plant.
It did not come from an algae bloom in the Himalayas.
It did not come from the ocean.
It came from a cracking tower in an oil refinery.
It was synthesized in a stainless-steel reactor using acid catalysts and hydrocarbon solvents.
It is not a health supplement.
It is an industrial pigment originally designed to dye the grey flesh of farmed salmon pink, so that consumers wouldn’t know the difference between a fish raised in a pen and a fish caught in the wild.
And now, the industry is doing the same thing to you.

The Anatomy of the Crime
We must be precise about the nature of this deception.
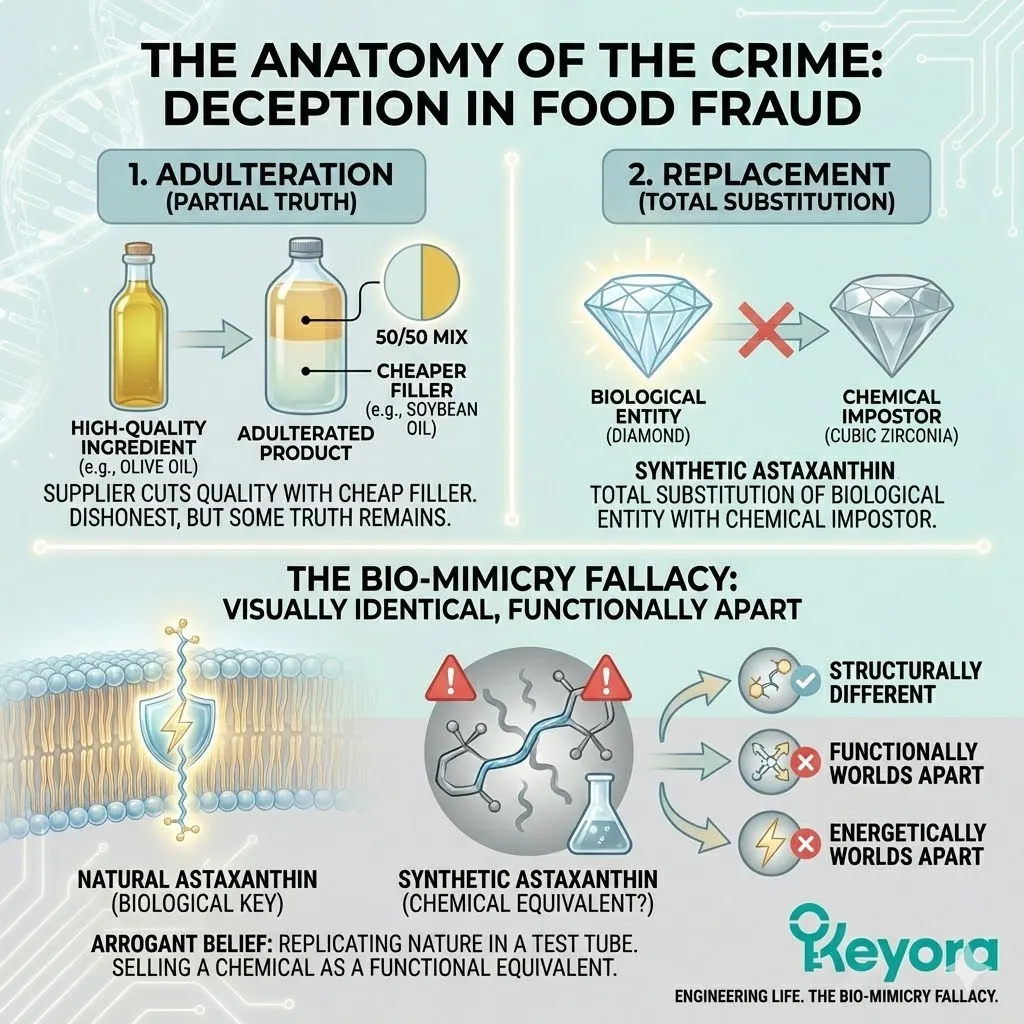
In the world of food fraud, there are two main categories: Adulteration and Replacement.
Adulteration is when a supplier cuts a high-quality ingredient with a cheaper filler. They might mix 50% olive oil with 50% soybean oil. It is dishonest, it is cheap, but there is still some truth in the bottle.
What is happening with Synthetic Astaxanthin is not adulteration.
It is Replacement.
It is the total substitution of a biological entity with a chemical impostor.
It is the equivalent of buying a diamond and receiving a piece of cubic zirconia.
Chemically, they might share similar elements.
Visually, they might look identical to the untrained eye.
But structurally, functionally, and energetically, they are worlds apart.
This is The Bio-Mimicry Fallacy.
It is the arrogant belief that we can replicate the complexity of nature in a test tube, strip away the biological context, and sell the resulting chemical as a functional equivalent.
The Economic Driver
Why does this fraud exist? The answer, as always, is margin.
Growing Haematococcus pluvialis microalgae – the natural source of Astaxanthin – is incredibly difficult. It requires pristine water, intense sunlight, and a clean environment.
It takes months to cultivate.
It is agriculture.
It is subject to the laws of nature.
Synthesizing Astaxanthin from crude oil is manufacturing.
It is scalable.
It is cheap.
It can be done in the dark, in a factory, anywhere in the world.
The cost of producing synthetic Astaxanthin is a fraction of the cost of growing the natural molecule.
For decades, this synthetic pigment was restricted to animal feed. It was the “salmon paint” used to trick consumers at the fish counter. But as the human demand for Astaxanthin skyrocketed – driven by the clinical research on its anti-aging benefits – unscrupulous manufacturers saw an opportunity.
They realized that the regulations in the supplement industry are loose enough, and the testing methods are obscure enough, that they could slip the synthetic pigment into the human supply chain.
They hid it behind The Labeling Void.
If you look at that bottle you bought, you will notice what is missing.
It will say “Astaxanthin.”
But it will not say “from Haematococcus pluvialis.”
It will not say “Algae Source.”
It will simply be silent on the origin.
That silence is an admission of guilt.
When you swallow that capsule, you are not feeding your cells.
You are participating in a global experiment on human toxicology.
You are introducing a molecule into your body that has never been part of the human food chain, a molecule born from the same black sludge that powers your car.
We are here to trace that lineage.
We are going to follow the supply chain backwards, from the capsule in your hand, all the way down to the bedrock of the earth.

1.1 From the Oil Rig to Your Stomach:
Deconstructing the Source Code
To understand the gravity of The Fossil Fuel Derivative, we must strip away the marketing veneer and look at the raw chemistry of production.
Where does Synthetic Astaxanthin actually come from?
It does not start with a seed.
It does not start with a spore.
It starts with Petroleum.
Step 1: The Extraction (Deep Time Death)
The journey begins miles underground, in a reservoir of crude oil.
This oil is the compressed, heated, and decomposed remains of ancient organic matter – dead zooplankton and algae from millions of years ago.
It is “fossil” energy.
It is dense, toxic, and chaotic.
This crude oil is pumped to the surface and sent to a refinery. There, it undergoes Thermal Cracking. The oil is heated to extreme temperatures (over 800°C) to break the long hydrocarbon chains into smaller, more useful molecules.
Out of this violent process comes a volatile, colorless liquid called Naphtha. And from Naphtha, chemists isolate a specific 5-carbon unit called Isoprene (2-methyl-1,3-butadiene).
Step 2: The Isoprene Backbone
Isoprene is the Lego brick of the synthetic rubber industry.
It is the primary ingredient used to make car tires, latex gloves, and adhesives.
It is a highly reactive, flammable petrochemical.
This is the “mother” of Synthetic Astaxanthin.
In a chemical plant – often located in industrial zones in China or Germany – chemists take this Isoprene and begin to build.
They do not use enzymes; they use reagents.
They use organic solvents like acetone, hexane, and dichloromethane to force the carbon atoms to bond.
They link two Isoprene units together to form a 10-carbon molecule.
Then they link two of those to form a 20-carbon molecule.
They repeat this process, forcing the chain to grow, step by step, reaction by reaction.

Step 3: The Wittig Reaction
The core of the synthesis usually involves a process called the Wittig Reaction.
It is a method for coupling aldehydes and ketones using phosphonium ylides.
It is brilliant chemistry, worthy of a Nobel Prize (which Georg Wittig won in 1979). But it is industrial chemistry.
To turn these hydrocarbon chains into “Astaxanthin,” the chemists must add oxygen. They must force oxygen atoms onto the carbon rings at the ends of the chain to create the specific “keto-carotenoid” structure.
This requires harsh oxidation catalysts.
It requires heat.
It requires pressure.
Step 4: The Result (The Red Dust)
At the end of this industrial assembly line, after days of chemical manipulation, the reactor vessel yields a dark red, crystalline powder.
Chemically, it is C40H52O4.
If you run a basic chemical analysis, it has the right number of carbons, hydrogens, and oxygens.
But energetically, it is dead.

The Contrast: The Algal Genesis
Compare this violent, extractive process to the genesis of Natural Astaxanthin.
In a Keyora-approved facility, the process begins with Haematococcus pluvialis, a microscopic green algae.
This algae is alive.
It floats in pristine, mineral-rich water (often sourced from glacial melt or deep aquifers).
When the sun rises, the algae wakes up.
It performs Photosynthesis.
It takes photons from the sun, carbon dioxide from the air, and water from the environment.
It uses enzymatic machinery that has been refined over billions of years of evolution to weave these elements together.
When the algae is stressed – by intense sunlight or nutrient deprivation – it activates a survival program. It begins to synthesize Astaxanthin to protect its DNA from UV radiation.
It builds the molecule atom by atom, using enzymes that ensure perfect Stereochemistry (the 3D shape we discussed in the previous episode).
It wraps the Astaxanthin in oil droplets (esters) to stabilize it.
The result is a molecule that is vibrantly alive.
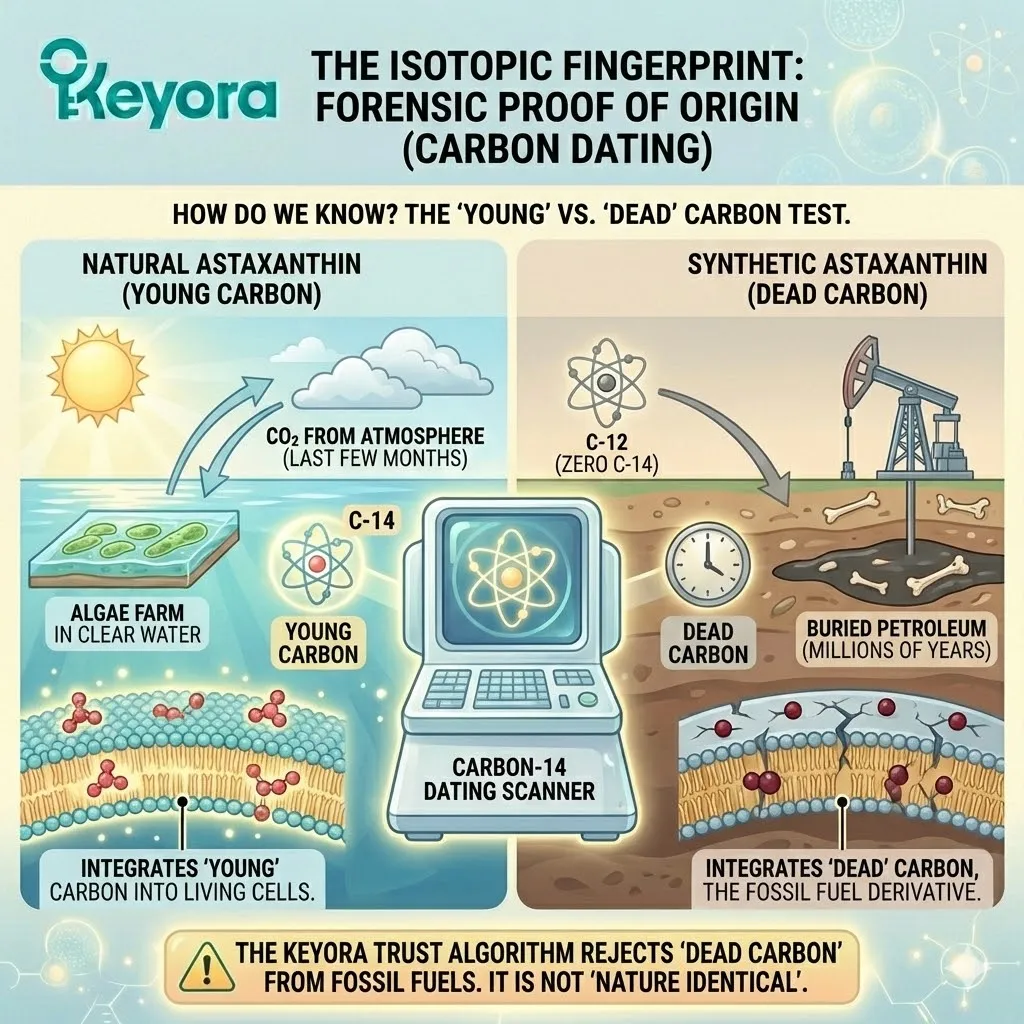
It carries the isotopic signature of the modern atmosphere (Carbon-14), proving it was grown recently.

The Isotopic Fingerprint
This brings us to the forensic proof. How do we know if a sample is synthetic or natural? We carbon date it.
-
Natural Astaxanthin: Contains Carbon-14. It was built from CO2 breathed in from the atmosphere within the last few months. It is “young” carbon.
-
Synthetic Astaxanthin: Contains Zero Carbon-14. It was built from petroleum that has been buried underground for millions of years. The radioactive carbon has long since decayed. It is “dead” carbon.
When you take a synthetic capsule, you are literally eating fossil fuels. You are integrating dead carbon into your living cell membranes.
This is the definition of The Fossil Fuel Derivative.
The industry tries to hide this.
They tell you that “a molecule is a molecule.”
They tell you that the source doesn’t matter, only the structure.
But as we will see in the next section, the structure isn’t the same. The chemical synthesis process is messy.
It creates side products.
It creates wrong shapes.
And it leaves behind a footprint of industrial solvents that no amount of purification can fully erase.
We have traced the origin from the oil rig to the reactor. Now, we must look at the process of “cooking” the molecule, and the legal loophole that allows companies to sell this industrial pigment as a “Nature Identical” health product.
1.2 Cooking the Molecule:
Catalysts, Solvents, and the Industrial Footprint
Let us step away from the abstract chemistry of carbon chains and enter the physical reality of creation.
To understand the profound difference between Natural and Synthetic Astaxanthin, you must visualize the environment of their birth.
You must witness the difference between a Garden and a Reactor.
The Algal Genesis: Quantum Precision
In a Keyora-approved facility, the production of Astaxanthin is a biological miracle driven by the most powerful energy source in the solar system:
The Sun.
The Haematococcus pluvialis microalgae is a solar-powered factory. Inside each microscopic cell lies a chloroplast – a biological machine that has been refining its efficiency for billions of years.
When a photon of sunlight strikes the algae, it triggers a quantum event. The energy is captured, stabilized, and used to weave carbon dioxide and water into complex, living structures.
The algae does not produce Astaxanthin randomly.
It produces it as a survival shield.
It creates the molecule to protect its own DNA from UV radiation.
Because this is a biological process, it is governed by enzymes. Enzymes are the ultimate nanobots.
They grab atoms and place them into the molecular structure with absolute precision.
They do not make mistakes.
They build the molecule atom by atom, ensuring that every bond is in the correct orientation, every ring is perfectly twisted, and every electron orbital is aligned.
The result is a molecule that vibrates with the energy of its creation.
It is suspended in a matrix of healthy oils (lipids) that the algae produced to store the pigment.
It is surrounded by supporting carotenoids (Lutein, Beta-Carotene, Canthaxanthin) that work in synergy.
It is a whole food.
It is wet.
It is alive.

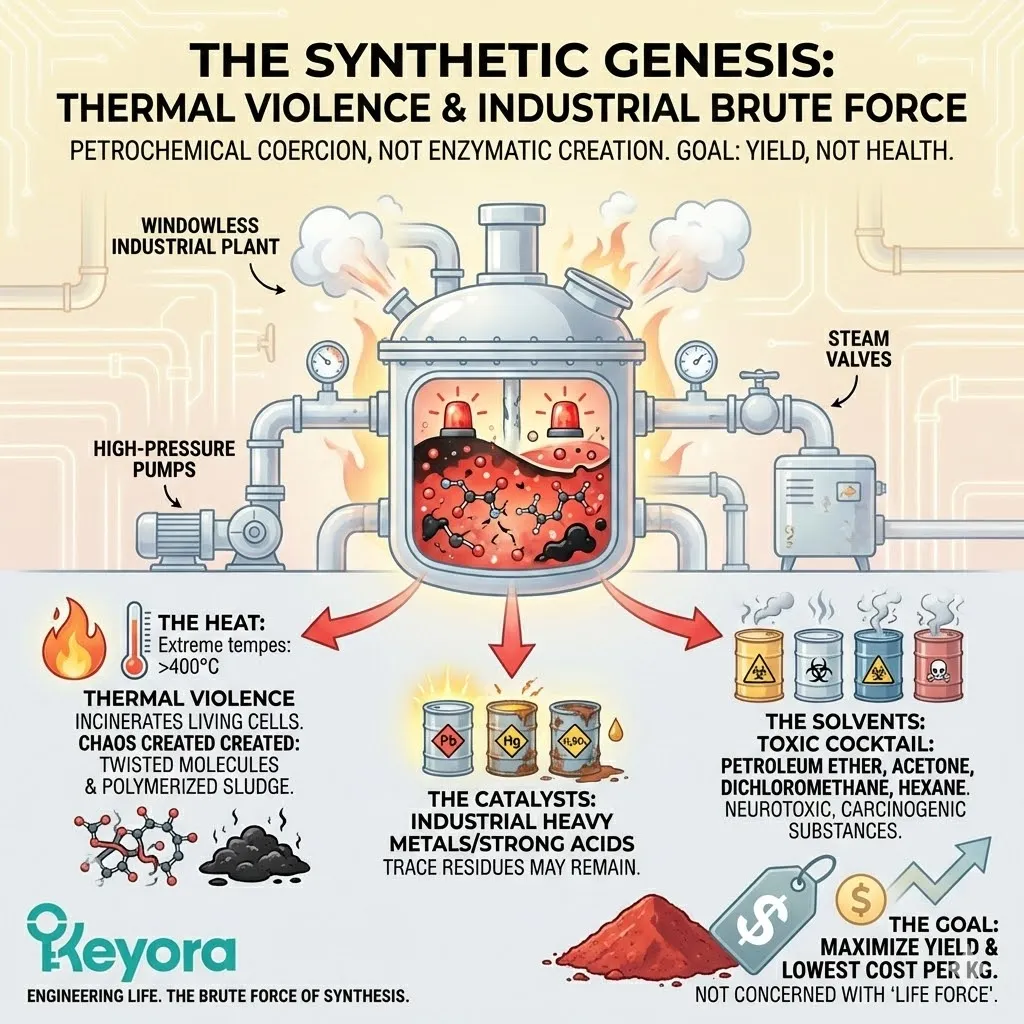
The Synthetic Genesis: Thermal Violence
Now, contrast this with the production of Synthetic Astaxanthin.
We leave the sunlight and enter a windowless industrial plant.
The air is thick with the smell of chemical solvents.
The sound is the hum of high-pressure pumps and the hiss of steam valves.
Here, there are no enzymes.
There is no delicate placement of atoms.
There is only Brute Force.
To force petrochemicals (Isoprene units) to bond together into the shape of Astaxanthin, chemists must use extreme measures.
They cannot coax the atoms; they must coerce them.
-
The Heat: The reaction vessels are heated to temperatures that would incinerate a living cell. This thermal violence forces the carbon chains to fuse, but it also creates chaos. It generates side products – twisted molecules and polymerized sludge – that must be filtered out later.
-
The Catalysts: To make the reaction happen, industrial catalysts are required. These are often heavy metals or strong acids. While the manufacturer attempts to remove them at the end, trace residues can remain.
-
The Solvents: The reagents must be dissolved to react. This requires a cocktail of organic solvents – Petroleum Ether, Acetone, Dichloromethane, Hexane. These are toxic, neurotoxic, and carcinogenic substances.
The goal of this process is not “health.”
The goal is “yield.”
The chemist is trying to produce as much red powder as possible for the lowest cost per kilogram.
They are not concerned with the “life force” of the molecule; they are concerned with the stoichiometry of the reaction.
The Energetic Void
There is a concept in thermodynamics regarding the “history” of a system. A diamond formed by the crushing pressure of the earth over millions of years is physically different from a cubic zirconia formed in a lab, even if they both refract light.
The Synthetic Astaxanthin molecule is born of dead energy. It is constructed from fossil fuels – carbon that has been buried in the dark, decaying for epochs. It carries no isotopic signature of the living world (Carbon-14 free).
When you ingest this molecule, you are introducing a substance that has been stripped of its biological context. It has been “cooked” in a reactor, washed in solvents, crystallized out of a solution, and dried into a red dust.
It is sterile.
It is isolated.
It is an artifact of industrial engineering, masquerading as a nutrient.
This is the hidden footprint of the synthetic supplement industry. They show you pictures of nature on the bottle, but they feed you the output of a chemical plant. They rely on the fact that you cannot see the history of the molecule.
But your cells can.
Your cells have evolved over millions of years to recognize and utilize molecules created by life. When they encounter a synthetic impostor – a molecule born of thermal violence and solvent extraction – they react differently.
They do not embrace it; they tolerate it.
Or worse, they reject it.
This leads us to the marketing sleight-of-hand that allows this deception to continue. The legal loophole that lets companies sell dead plastic precursors as “health.”

1.3 The “Nature Identical” Trap:
A Legal Loophole That Masks a Biological Impossibility
How is this legal?
How can a company take a red powder synthesized from crude oil derivatives and sell it in a health food store?
They use a phrase that sounds scientific, reassuring, and precise. A phrase that is, in reality, one of the greatest lies in modern commerce:
“Nature Identical.”
This term is the cornerstone of The Bio-Mimicry Fallacy.
The Definition of the Lie
“Nature Identical” is a regulatory classification. It means that the primary molecule in the synthetic product shares the same 2D chemical formula (C40H52O4) as the molecule found in nature.
On a piece of paper, drawn in two dimensions, they look the same.
In a basic chemical analysis that only counts atoms, they look the same.
But this definition ignores the complexity of biological reality.
It is a reductionist trap.
It assumes that a living thing is nothing more than the sum of its atoms.
By this logic, a plastic apple is “Nature Identical” to a Gala apple if the plastic is painted the right shade of red.
They are both round.
They are both red.
They both fit in your hand.
But if you eat the plastic apple, you will not get nourishment; you will get a bowel obstruction.

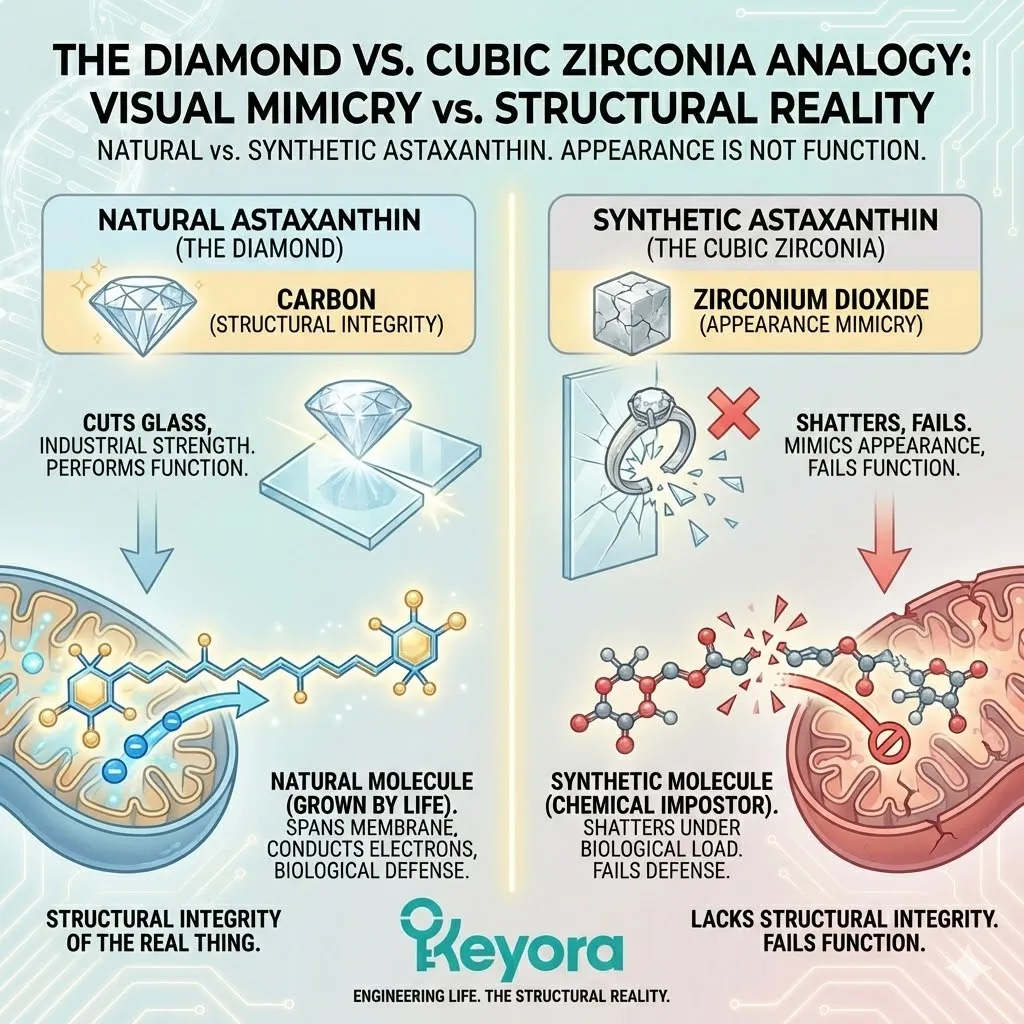
The Diamond vs. Cubic Zirconia Analogy
Let us return to the diamond analogy, because it is the most accurate way to visualize this fraud.
A natural diamond is carbon.
Synthetic cubic zirconia is zirconium dioxide.
To the naked eye, across a dinner table, they both sparkle. They both look like “diamonds.” A jeweler can sell a cubic zirconia ring for a fraction of the price of a real diamond, and if you don’t look closely, you might feel like you got a bargain.
But try to cut glass with cubic zirconia.
It will shatter.
Try to use it in an industrial drill bit.
It will fail.
It lacks the Structural Integrity of the real thing.
It mimics the appearance, but it fails the function.
Synthetic Astaxanthin is the cubic zirconia of the supplement world.
It is red.
It is a powder.
It fits in a capsule.
But when you ask it to perform the heavy lifting of biological defense – when you ask it to span a mitochondrial membrane, conduct high-voltage electrons, and neutralize singlet oxygen without degrading – it shatters.
It lacks the structural integrity of the molecule grown by life.
The Missing “Matrix”
The fallacy of “Nature Identical” also ignores the concept of the Biological Matrix.
In nature, Astaxanthin never exists alone. In the Haematococcus algae, the Astaxanthin molecule is embedded in a complex oil droplet.
It is surrounded by a support crew:
-
Other Carotenoids: Lutein, Canthaxanthin, Beta-Carotene. These molecules “chaperone” the Astaxanthin, regenerating it when it gets tired.
-
Fatty Acids: The algae produces specific lipids to dissolve the Astaxanthin, ensuring it remains stable and bioavailable.
-
Vitamins: Natural Vitamin E (Tocopherols) protects the Astaxanthin from oxidizing before it is used.
This is the Entourage Effect. The whole is greater than the sum of the parts.
Synthetic Astaxanthin is an isolate.
It is 100% pure pigment (or a diluted version of it).
It has been stripped of its entourage.
It is a general without an army.
When you ingest Natural Astaxanthin, you are ingesting the entire biological matrix.
You are eating the oil, the chaperones, and the vitamins.
You are consuming a functional system.
When you ingest Synthetic Astaxanthin, you are consuming a naked, isolated chemical.
It has no support system.
It is vulnerable to oxidation in your stomach.
It lacks the co-factors required for optimal absorption.

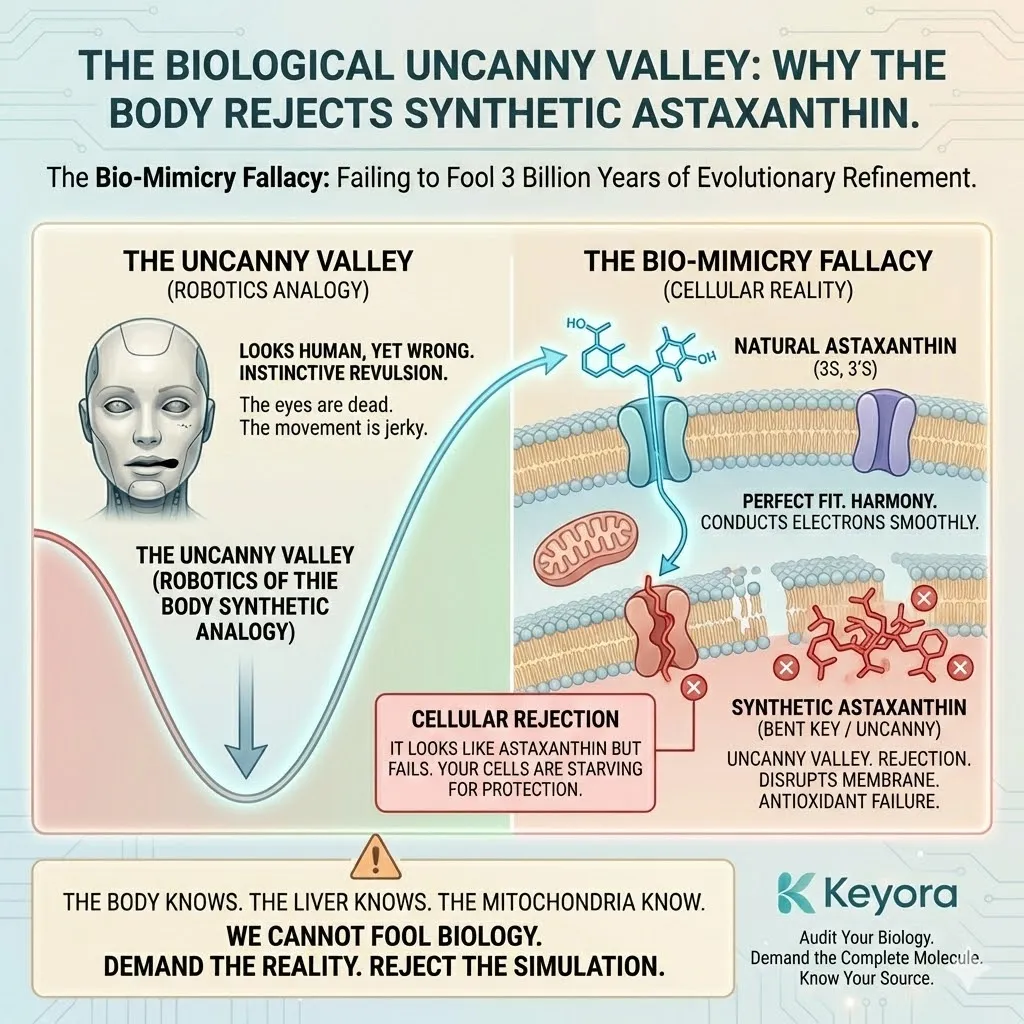
The Biological Uncanny Valley
Robotics experts talk about the “Uncanny Valley” – the feeling of revulsion humans get when a robot looks almost human, but not quite.
It looks like us, but something is wrong.
The eyes are dead.
The movement is jerky.
We instinctively reject it.
Your cells have a similar response to Synthetic Astaxanthin.
Because it is “Nature Identical” but not “Natural,” it sits in the Uncanny Valley of biology.
-
It looks like Astaxanthin to a transport protein, but it doesn’t bind quite right.
-
It looks like an antioxidant to a free radical, but it doesn’t conduct the electron smoothly.
-
It enters the membrane, but it disrupts the fluidity instead of stabilizing it.
This is The Bio-Mimicry Fallacy.
It is the hubris of thinking we can fool 3 billion years of evolutionary refinement with 50 years of petrochemical chemistry.
We cannot.
The body knows.
The liver knows.
The mitochondria know.
When you buy a product labeled “Astaxanthin” without verifying the source, you are falling into this trap.
You are buying the simulation of health.
You are buying a red pill that colors your insides but leaves your cells starving for protection.
We must reject the simulation.
We must demand the reality.
In the final section of this chapter, we will give you the practical tools to spot this fraud.
We will teach you how to interrogate the label and expose the silence that hides the synthetic origin.
1.4 How to Spot the Fake:
Interrogating The Labeling Void
We have exposed the origin (petrochemicals).
We have detailed the process (thermal violence).
We have dismantled the marketing lie (”Nature Identical”).
Now, we must arm you with the practical skills to navigate the supplement aisle.
You must become an auditor of The Labeling Void.
The most dangerous thing on a supplement label is not what is written; it is what is missing.
In the world of Synthetic Astaxanthin, silence is the primary strategy. Manufacturers know that “Derived from Crude Oil” is not a selling point. So they rely on omission.
They rely on the fact that you will see the word “Astaxanthin” and assume it comes from a plant.
Here is your audit protocol. When you pick up a bottle, turn it over.
Ignore the front.
Ignore the claims.
Look at the “Supplement Facts” panel and the “Other Ingredients” list.

The Test of Origin:
The Pass:
Does the label explicitly state “from Haematococcus pluvialis microalgae”?
Does it say “Natural Astaxanthin”?
Does it list “Algae Oil” or “Algae Extract”?
-
Why: Producers of premium Natural Astaxanthin are proud of their source. They pay a premium to grow it. They want you to know. They will shout it from the rooftops.
The Fail:
Does it simply say “Astaxanthin” with no source listed?
Does it say “Astaxanthin Complex” without defining the components?
Does it use the vague term “Carotenoid Pigment”?
-
Why: Silence is guilt. If they don’t tell you it’s algae, it’s almost certainly petrochemical.
The Test of the Carrier:
The Pass:
Is the Astaxanthin suspended in a high-quality lipid matrix?
Does the label list “Extra Virgin Olive Oil,” “MCT Oil,” or ideally, a functional matrix like “Flaxseed Oil” or “Omega-3 Fatty Acids”?
-
Why: Natural Astaxanthin requires fat for absorption. A brand that understands the science will provide a premium carrier.
The Fail:
Is it a dry powder inside a hard-shell capsule?
Does the ingredient list include “Corn Starch,” “Gelatin,” “Sucrose,” or “Ascorbyl Palmitate” (preservatives used to stabilize synthetic beadlets)?
-
Why: Synthetic Astaxanthin is often produced as a dry beadlet containing gelatin and starch. If you see these fillers without a lipid carrier, you are looking at a synthetic product (or a poorly formulated natural one).

The Test of the Dose:
The Pass:
Is the dose 4mg, 6mg, or 12mg? (Standard therapeutic doses).
The Fail:
Is the dose microscopic, like 1mg or 0.5mg?
-
Why: This is “Fairy Dusting.” Synthetic producers often sell very low doses because their product is cheap filler, or because they are trying to avoid regulatory scrutiny regarding the safety of high-dose synthetics.
The “Nature Identical” Red Flag:
If you see the phrase “Nature Identical” anywhere on the packaging or marketing materials, put the bottle down. This is the smoking gun.
No natural product calls itself “Nature Identical.”
An apple does not call itself “Nature Identical Apple.”
Only a synthetic copycat needs to use this language.

The Keyora Promise: Radical Transparency
At Keyora, we do not hide our source; we celebrate it.
We list the strain of algae. We list the extraction method (Supercritical CO2).
We list the carrier oils (ALA/LA/OA).
We believe that you have a right to know exactly what you are putting into your body.
We believe that the supply chain should be glass, not a black box.
When you hold a bottle of Keyora Astaxanthin, you are holding the result of sunlight, water, and biological evolution.
ou are not holding a byproduct of the petrochemical industry.

1.5 The Verdict:
Oil Belongs in Cars, Not in Cells
We have completed the audit of the origin.
The evidence is irrefutable.
Synthetic Astaxanthin is a molecular impostor.
-
Origin: It is born from crude oil, extracted from the earth’s crust, and cracked in a refinery.
-
Process: It is synthesized using harsh chemicals, heat, and pressure, stripping it of any biological life force.
-
Identity: It masquerades as a health supplement under the false flag of “Nature Identical,” hiding its true nature behind labeling voids and legal loopholes.
Natural Astaxanthin is the authentic article.
-
Origin: It is grown from living microalgae, powered by the sun.
-
Process: It is biosynthesized by enzymes with quantum precision.
-
Identity: It is a whole-food complex, supported by a matrix of carotenoids and lipids, designed by evolution for survival.
The choice is stark.
You are the Chief Executive Officer of your own biology.
You make decisions every day about resource allocation.
You decide what fuel goes into the engine.
Would you put low-grade, adulterated fuel into a Ferrari?
Would you build a skyscraper with counterfeit steel?
Then why would you build your cells with petrochemicals?
Oil belongs in cars.
It belongs in plastics.
It belongs in the asphalt of the road. It does not belong in the delicate lipid membranes of your brain.
It does not belong in the photoreceptors of your eyes.
It does not belong in the mitochondria that power your life.
We have exposed the fraud of the source. But the indictment is not over.
In the next chapter, we will go deeper.
We will move from the origin of the molecule to the shape of the molecule.
We will explore the physics of Stereochemistry.
We will show you why the synthetic molecule is not just “dead” – it is physically deformed. It is a “bent key” that jams the locks of your cellular machinery.
We will prove that Geometry is Destiny.

# KNOWLEDGE SUMMARY: THE ORIGIN FRAUD
## I. THE TAXONOMY OF DECEPTION [THE GREAT IMPOSTOR]
* **The Categorization of Fraud:**
* **Adulteration:** Mixing high-quality ingredients with cheap fillers (e.g., Olive oil cut with Soybean oil).
* **Replacement:** The total substitution of a biological entity with a chemical impostor. This is the case with Synthetic Astaxanthin.
* **The Economic Driver:**
* **Natural:** Agricultural, time-intensive, requires pristine water/sunlight (High Cost).
* **Synthetic:** Industrial, scalable, independent of nature (Low Cost).
* **The Historical Context:** Synthetic Astaxanthin was originally created as **”Salmon Paint”** (animal feed additive) to dye farmed fish flesh, not for human health.
## II. THE PETROCHEMICAL LINEAGE [THE SOURCE CODE]
* **The Raw Material:** **Crude Oil** (Fossilized organic matter).
* **The Synthesis Pathway:**
1. **Thermal Cracking:** Crude oil is heated (>800°C) to break hydrocarbon chains.
2. **Fractional Distillation:** Isolates **Naphtha**.
3. **Isoprene Extraction:** Isolation of **2-methyl-1,3-butadiene** (Isoprene), a precursor also used for **car tires** and **synthetic rubber**.
4. **The Wittig Reaction:** A chemical coupling process using phosphonium ylides to link carbon chains.
5. **Forced Oxidation:** Using metal catalysts and heat to add oxygen atoms to the rings.
* **The Forensic Proof (Carbon Dating):**
* **Natural:** Contains **Carbon-14** (Isotopically “Young/Living”).
* **Synthetic:** **Zero Carbon-14** (Isotopically “Dead/Fossil”).
## III. THE MANUFACTURING ENVIRONMENT [GARDEN VS. REACTOR]
* **The Algal Genesis (Photosynthesis):**
* *Mechanism:* Solar energy + CO2 + Water + Enzymatic precision.
* *Result:* Atom-by-atom construction with perfect stereochemistry.
* **The Synthetic Genesis (Thermal Violence):**
* *Mechanism:* Brute force heat + Pressure + Acid Catalysts.
* *The Toxic Footprint:* Requires solvents like **Petroleum Ether, Acetone, Dichloromethane, and Hexane**.
* *Residue Risk:* Potential for trace contamination from catalysts and unreacted intermediates.
## IV. THE MARKETING LIE [THE BIO-MIMICRY FALLACY]
* **The Term:** **”Nature Identical”**.
* *The Truth:* It refers only to the **2D Chemical Formula** ($C_{40}H_{52}O_4$).
* *The Lie:* It ignores **3D Geometry** (Stereochemistry), **Isotopic Signature**, and **Bio-Energetics**.
* **The Analogy:** **Diamond (Carbon) vs. Cubic Zirconia (Zirconium Dioxide)**.
* Visually similar to the naked eye; structurally and functionally inferior under pressure.
* **The Missing Matrix (The Entourage Effect):**
* *Natural:* Comes with **Lutein, Beta-Carotene, Canthaxanthin, and Vitamin E** in a lipid droplet.
* *Synthetic:* An isolated, “naked” pigment. Unstable and prone to oxidation in the gut.
* **The Biological “Uncanny Valley”:** Cells recognize the synthetic molecule as foreign/imperfect, leading to poor uptake or rejection.
## V. THE CONSUMER AUDIT PROTOCOL [THE LABELING VOID]
* **The Primary Red Flag:** **Silence**. If the source is not explicitly stated, it is petrochemical.
* **The “Pass” Criteria:**
* “From *Haematococcus pluvialis*.”
* “Natural Astaxanthin.”
* “Algae Source / Algae Oil.”
* **The “Fail” Criteria:**
* “Astaxanthin” (Unqualified).
* “Nature Identical.”
* “Astaxanthin Complex” (Often a mask for low-grade spiking).
* **The Carrier Oil Test:**
* *Premium:* Olive Oil, MCT, Flaxseed (Functional Lipids).
* *Suspicious:* Corn Starch, Gelatin beadlets, Sucrose, Ascorbyl Palmitate (Preservatives for synthetic powder).

CHAPTER 2: THE GEOMETRY OF FAILURE
THE – STEREOCHEMICAL LOCK
Why Mechanistic Validity Fails: The Physics of Chirality and the 75% Impurity.
We have entered the laboratory.
We have left behind the glossy marketing brochures and the vague promises of “wellness.”
We are now looking at the fundamental laws that govern life itself.
And the first law of biological interaction is this:
Geometry is Destiny.
In the public imagination, chemistry is often reduced to a simple list of ingredients.
We think that if two substances have the same chemical formula – the same number of carbon, hydrogen, and oxygen atoms – they must be the same thing.
We assume that C40H52O4 is always C40H52O4.
This is a dangerous oversimplification.
It ignores the single most critical dimension of molecular reality:
Space.
Molecules are not flat drawings on a page.
They are three-dimensional objects.
They have depth, volume, and orientation.
They twist, they fold, and they occupy space in very specific ways.
This spatial arrangement is called Stereochemistry. And within stereochemistry, there is a concept that determines whether a molecule heals you or kills you:
Chirality.

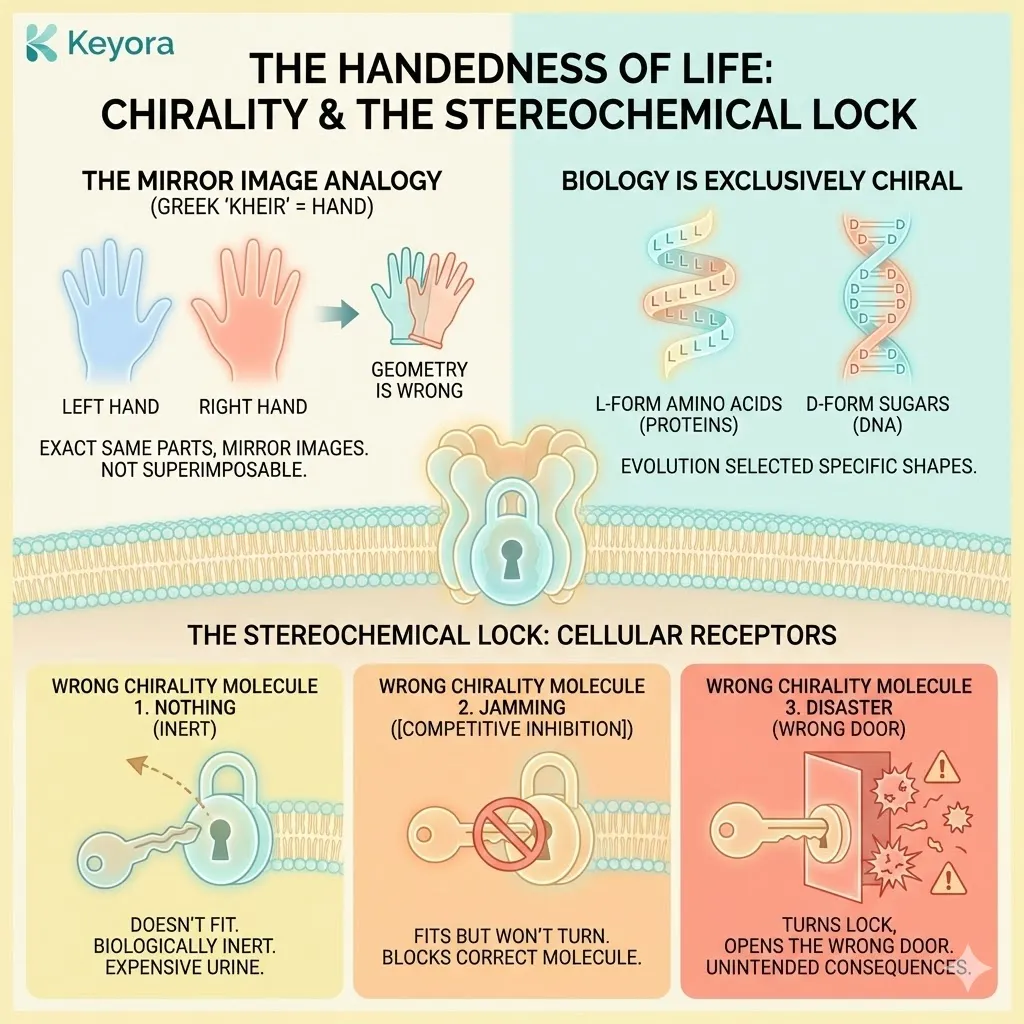
The Handedness of Life
The word “Chiral” comes from the Greek word for “hand” (kheir). Look at your own hands. Your left hand and your right hand are composed of the exact same parts – four fingers, one thumb, palm, and skin. They are mirror images of each other.
But they are not superimposable.
You cannot put a left-handed glove on your right hand. It does not fit. The geometry is wrong.
Biology is exclusively chiral.
The proteins in your body are built almost entirely of “left-handed” (L-form) amino acids.
The sugars in your DNA are “right-handed” (D-form).
Evolution has selected these specific shapes over billions of years.
Your cells are filled with receptors, enzymes, and transport channels that are essentially complex, three-dimensional locks. They are designed to accept only one specific “handedness” of a molecule.
This is The Stereochemical Lock.
If you introduce a molecule with the wrong chirality – a “right-handed” key for a “left-handed” lock – one of three things happens:
-
Nothing: The key doesn’t fit. The molecule is biologically inert. It is expensive urine.
-
Jamming: The key fits into the hole but won’t turn. It blocks the receptor, preventing the correct molecule from entering. This is [Competitive Inhibition].
-
Disaster: The key turns the lock, but opens the wrong door.
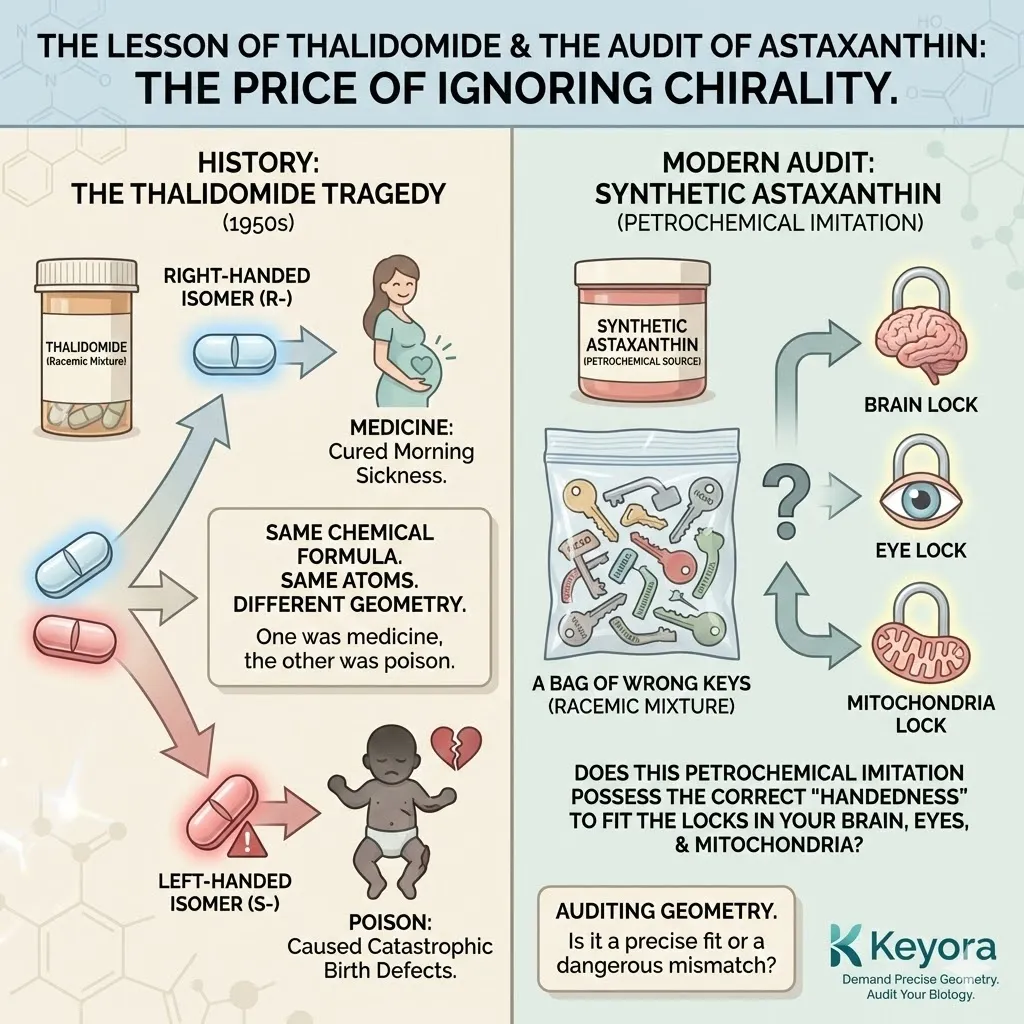
The Lesson of Thalidomide
History has taught us the price of ignoring chirality. In the 1950s, the drug Thalidomide was prescribed to pregnant women for morning sickness.
The drug was synthesized as a “racemic mixture” – a 50/50 blend of both the right-handed and left-handed forms of the molecule.
-
The Right-Handed isomer cured morning sickness.
-
The Left-Handed isomer caused catastrophic birth defects.
They had the same chemical formula.
They had the same atoms.
But their geometry was different.
One was medicine; the other was poison.
When we audit Synthetic Astaxanthin, we are auditing its geometry.
We are asking:
Does this petrochemical imitation possess the correct “handedness” to fit the locks in your brain, your eyes, and your mitochondria?
Or is it a bag of wrong keys?
2.1 The Biological Key:
The Evolutionary Perfection of the 3S, 3’S Configuration
To understand the counterfeit, we must first study the original.
We must look at the “Master Key” that nature designed.
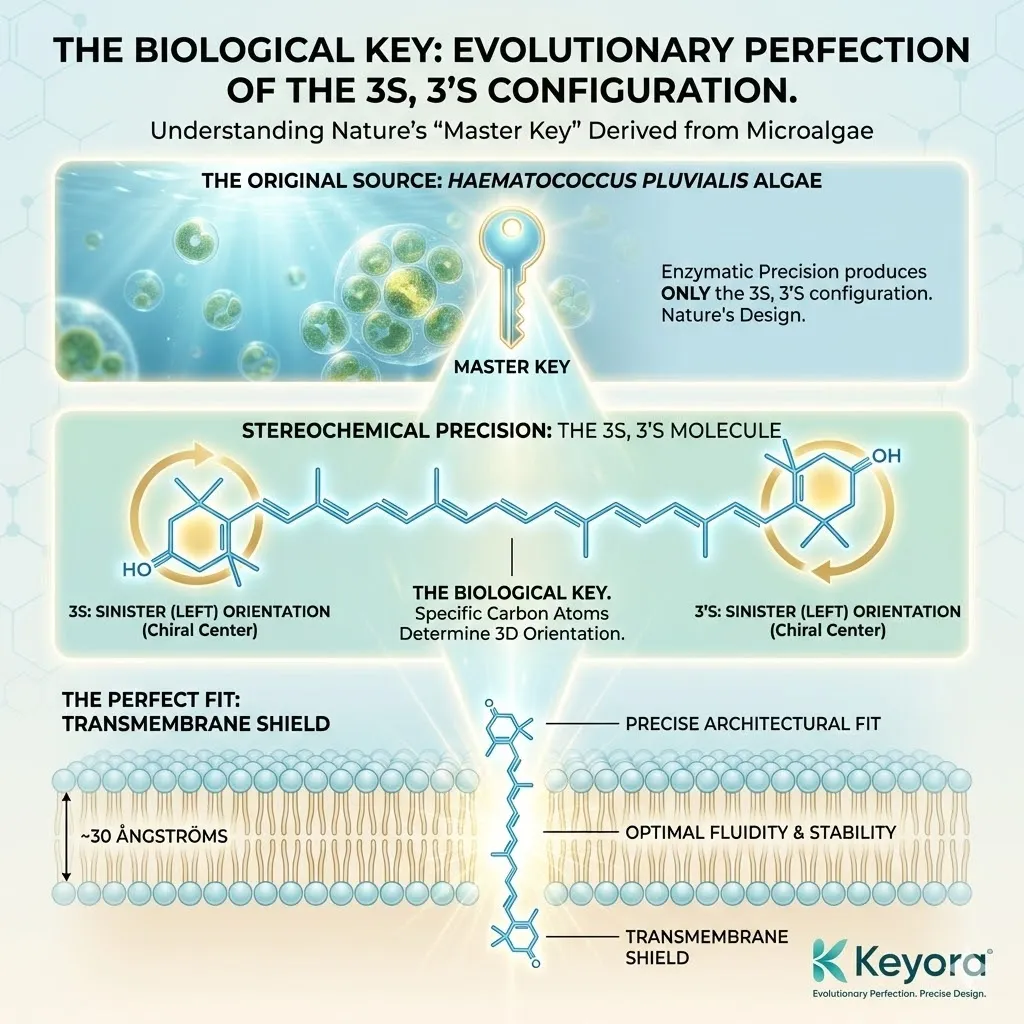
Natural Astaxanthin, derived from the microalgae Haematococcus pluvialis, is a masterpiece of stereochemical precision.
The Astaxanthin molecule is a long chain with a ring structure at each end.
These rings contain “chiral centers” – specific carbon atoms that determine the 3D orientation of the molecule.
In Haematococcus algae, the enzymatic machinery is so precise that it produces only one specific configuration: 3S, 3’S.
-
3S: The chiral center on the left ring is in the “S” (Sinister/Left) orientation.
-
3’S: The chiral center on the right ring is also in the “S” orientation.
This is The Biological Key.
Why did evolution select this specific shape?
Why 3S, 3’S and not 3R, 3’R?
The answer lies in the Phospholipid Bilayer – the cell membrane.
As we discussed in Episode 1, the cell membrane is a precise architectural structure. It has a specific thickness (~30 Ångströms) and a specific fluidity.
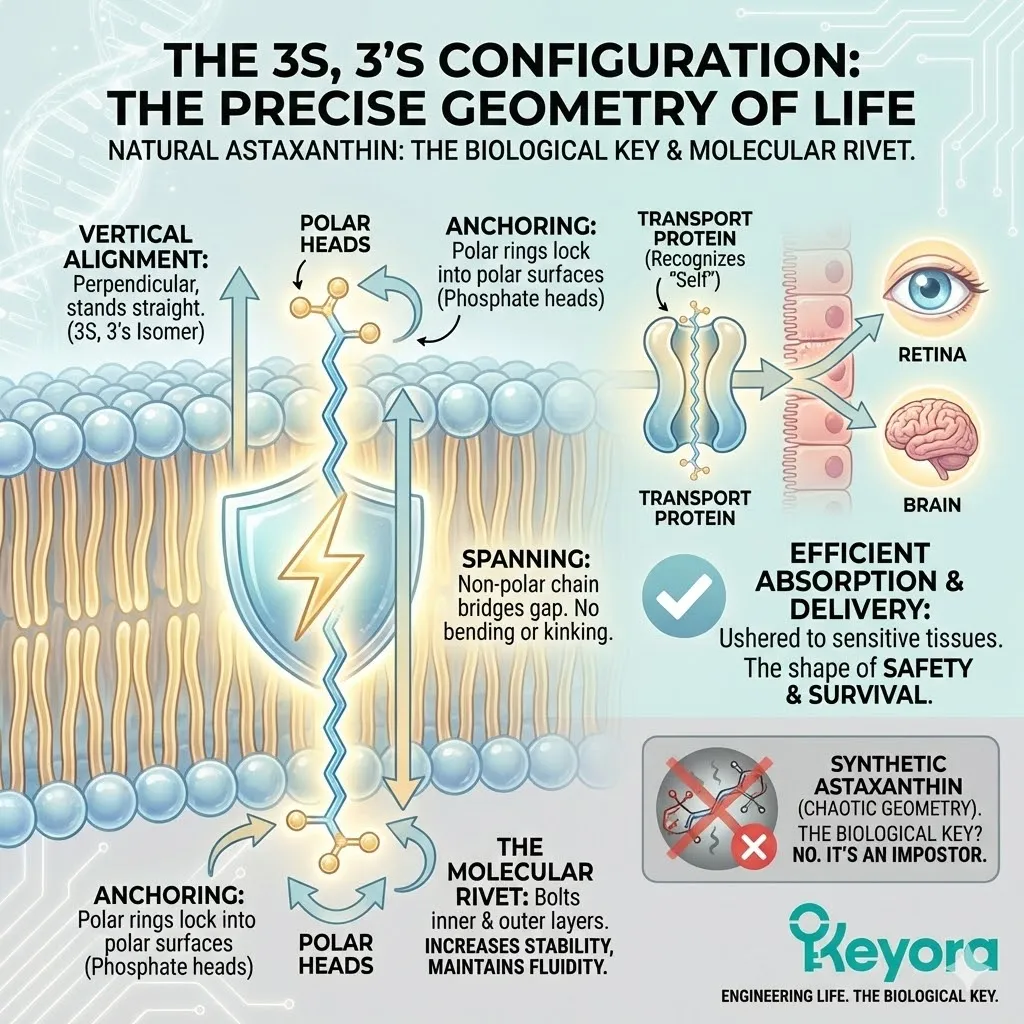
To function as The Transmembrane Shield, the Astaxanthin molecule must fit perfectly into this space.
The 3S, 3’S configuration gives the Natural Astaxanthin molecule a specific linear shape. It is straight. It is rigid where it needs to be and flexible where it needs to be.
-
Vertical Alignment: The 3S, 3’S isomer aligns perfectly perpendicular to the membrane surface. It stands up straight.
-
Anchoring: The polar heads (the rings) lock into the polar surfaces of the membrane (the phosphate heads of the lipids).
-
Spanning: The non-polar chain bridges the gap without bending or kinking.
This geometry allows Natural Astaxanthin to act as a Molecular Rivet. It physically bolts the inner and outer layers of the membrane together, increasing stability while maintaining fluidity.
Because it fits the Stereochemical Lock of the membrane so perfectly, the body absorbs it efficiently. Transport proteins recognize it.
It is ushered across the gut wall, packaged into chylomicrons, and delivered to the most sensitive tissues in the body – the retina and the brain.
The body recognizes the 3S, 3’S shape as “Self.”
It is the shape of safety.
It is the shape of survival.
This is the standard against which all other forms must be judged. If a molecule claims to be Astaxanthin but does not possess the 3S, 3’S configuration, it is not The Biological Key.
It is an impostor.
Now, let us walk into the petrochemical factory and see what they are building.
Let us look at the chaotic geometry of Synthetic Astaxanthin.
2.2 The Synthetic Chaos
Deconstructing the 1:2:1 Racemic Mixture
We leave the pristine, sunlit environment of the algae farm and enter the industrial reality of petrochemical synthesis.
Here, Astaxanthin is not grown; it is constructed.
The process begins with petroleum derivatives – isophorone and other chemical intermediates. Through a complex series of reactions involving acid catalysts and heat, chemists force carbon atoms together to build the Astaxanthin backbone.
But chemistry in a beaker lacks the precision of biology.
Enzymes in algae are like master sculptors; they can place every atom in the exact 3D position required.
Chemical synthesis is more like a shotgun blast. It cannot easily control chirality. When you synthesize a chiral molecule without biological enzymes, you get a statistical average of all possible shapes.
You get a Racemic Mixture.

When you analyze a batch of Synthetic Astaxanthin (marketed as “Nature Identical”), you do not find pure 3S, 3’S. Instead, you find a chaotic soup of three different isomers in a fixed ratio of 1:2:1.
-
25% (3S, 3’S): The “correct” shape. This is the only part of the mixture that is actually identical to Natural Astaxanthin.
-
50% (3R, 3’S): The “Meso” form. This is a hybrid. One end is left-handed, the other is right-handed. It is twisted. It does not exist in nature in any significant quantity.
-
25% (3R, 3’R): The “Anti-Natural” form. Both ends are right-handed. This is the mirror image of the natural molecule.
This is The Stereochemical Mismatch.
The label says “Astaxanthin,” but the reality is that 75% of the molecules in that capsule are geometrically incorrect.
They are alien shapes that your body has never encountered in millions of years of evolution.
The term “Nature Identical” is a legal fiction. It refers only to the 2D chemical formula. In the 3D reality of biology, Synthetic Astaxanthin is a different substance entirely. It is a Frankenstein molecule – a patchwork of correct and incorrect parts stitched together in a lab.
Imagine buying a box of 100 right-handed gloves, only to open it and find that 25 are right-handed, 25 are left-handed, and 50 are a strange, mutated shape with thumbs on both sides.
That is what you are buying when you purchase Synthetic Astaxanthin.

2.3 The 75% Impurity
The Economic and Biological Cost of “Dead” Molecules
What happens when you ingest this chaotic mixture?
What is the cost of consuming a product that is 75% incorrect?
The Economic Cost (Paying for Waste)
First, there is the simple math of value. When you buy a 12mg capsule of Synthetic Astaxanthin, you are paying for 12mg. But biologically, you are only receiving 3mg of the active, natural isomer (25%).
The other 9mg is essentially filler. It is metabolic noise.
If a synthetic brand claims to be “cheaper” than natural, you must multiply their price by four to get the true cost of the active ingredient.
Suddenly, the “cheap” alternative becomes the most expensive option on the shelf. You are paying premium prices for industrial byproduct.

The Biological Cost (The Receptor Jam)
But the cost is not just financial; it is physiological. The incorrect isomers (3R, 3’S and 3R, 3’R) are not just inert. They are active impediments.
This brings us back to the concept of Competitive Inhibition.
Your cell membranes and transport proteins have a limited number of “parking spots” (binding sites) for carotenoids. When you flood your system with a racemic mixture, the “bent keys” (synthetic isomers) compete with the “master key” (natural isomer) for these spots.
-
The Blockade: The synthetic isomers – because they are chemically similar – can occupy the binding sites. But because their geometry is wrong, they cannot perform the function. They sit in the lock, preventing the correct key from entering.
-
The Dilution: Even if they don’t block the site completely, they dilute the efficacy. Instead of a cell membrane reinforced by 100% functional, vertical Astaxanthin rivets, you get a membrane peppered with structural defects.
You are effectively jamming your own radar.
This explains the massive discrepancy in antioxidant power. Recall the data from Capelli et al. (2013): Natural Astaxanthin is 20 to 90 times stronger than Synthetic Astaxanthin in free radical scavenging.
Why such a massive gap? It isn’t just that the synthetic molecule is weak; it’s that the synthetic mixture is self-sabotaging. The incorrect isomers are dead weight, clogging the system and preventing the active fraction from doing its job.
The 75% impurity is not just waste; it is Metabolic Drag.
But the failure goes deeper than just antioxidant capacity. The ultimate test of Astaxanthin is its ability to integrate into the cell membrane and stabilize it.
This is where the geometry of the synthetic molecule fails most spectacularly.
We must look at how a “bent” molecule behaves in a straight wall.

2.4 The Membrane Rejection
Why a Bent Key Cannot Open The Transmembrane Shield
We have established that Synthetic Astaxanthin is a chaotic mixture of wrong shapes.
Now, let’s visualize the physical consequence of this chaos at the site of action:
The Cell Membrane.
The cell membrane is a precise, orderly structure – a bilayer of phospholipids standing shoulder-to-shoulder like soldiers in a phalanx.
The space between them is tight.
The orientation is vertical.
Natural Astaxanthin (3S, 3’S) is linear.
It is straight.
It slides into this phalanx perfectly, standing tall between the lipids.
Its polar heads lock onto the inner and outer surfaces, spanning the entire width of the membrane (approx. 30 Ångströms).
It creates a solid, structural bridge – The Transmembrane Shield.
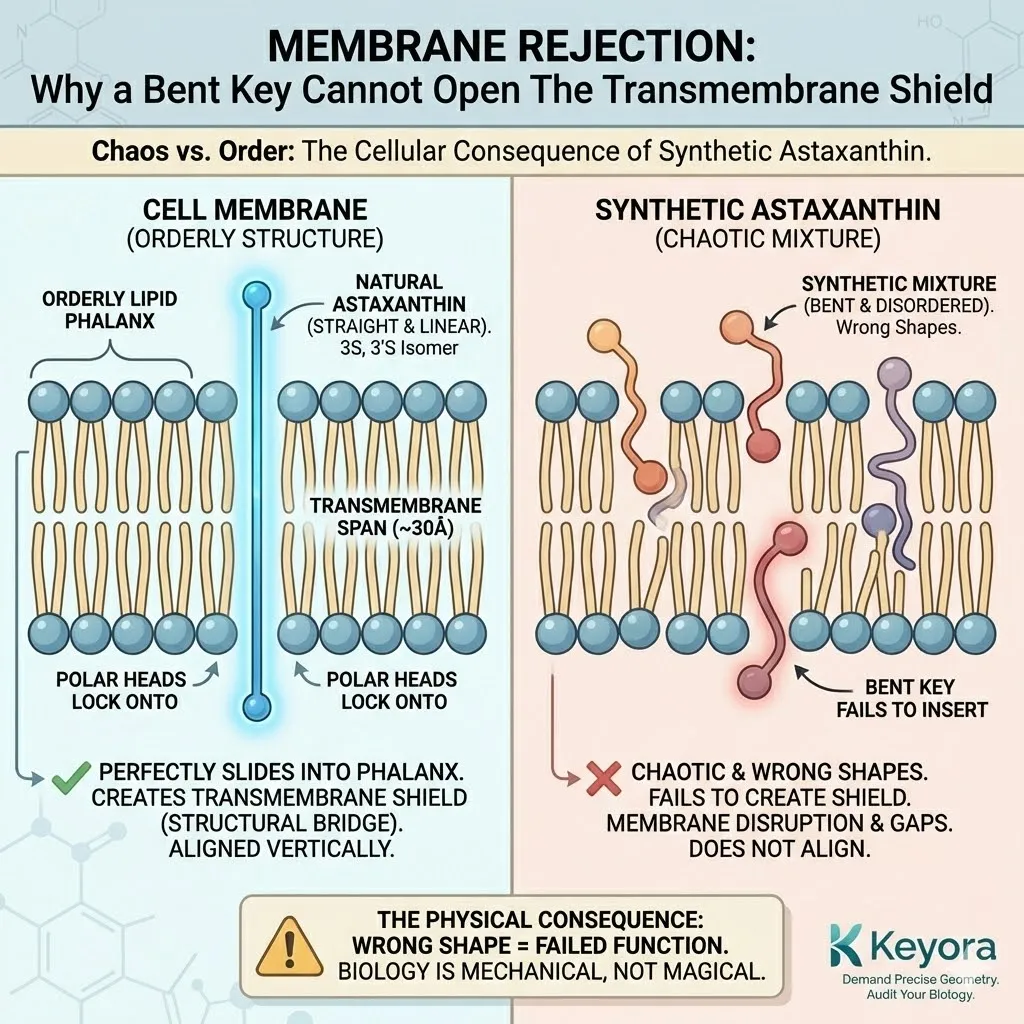
Synthetic Astaxanthin (3R, 3’S and 3R, 3’R) is different.
Due to the incorrect chirality at the end rings, the molecule twists.
It bends.
It does not stand straight.
When this “bent” molecule tries to enter the membrane, it fails the structural test.
-
Failure to Span: Because it is bent, it effectively becomes “shorter.” It cannot reach both sides of the membrane simultaneously. It cannot lock in.
-
Disruption: Instead of reinforcing the wall, it disrupts it. It pushes the lipids apart. It creates gaps. It introduces disorder into the orderly phalanx.
-
Floating: Because it cannot anchor securely, it often gets stuck in the middle of the lipid bilayer or floats aimlessly on the surface.
This is Membrane Rejection.
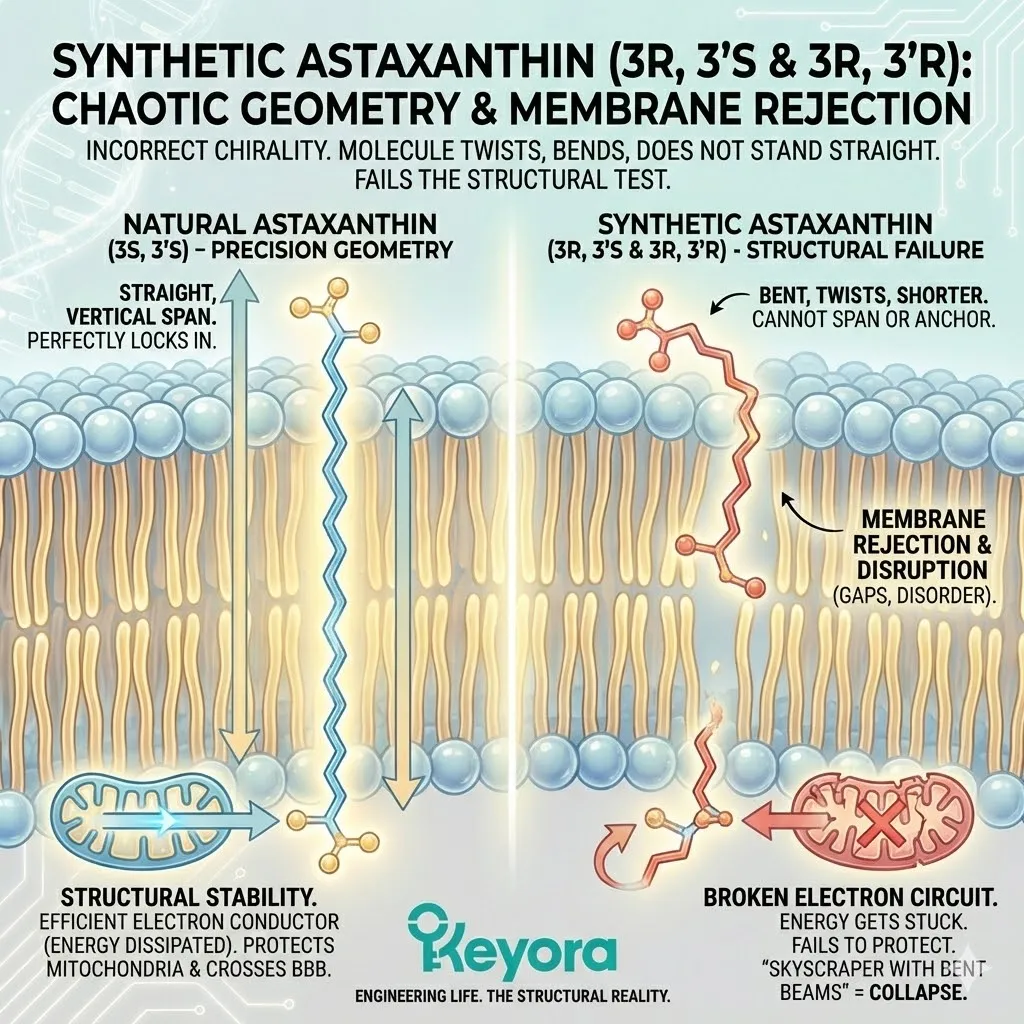
The synthetic molecule fails to become a “rivet.” It fails to stabilize the membrane. And crucially, it fails to conduct electrons efficiently.
Remember, the power of Astaxanthin lies in its ability to conduct excess electron energy (from free radicals) harmlessly out of the cell. This requires a straight, unbroken path along the carbon chain. A bent molecule breaks the circuit.
The electron gets stuck.
The energy is not dissipated.
This is why Synthetic Astaxanthin fails to protect the mitochondria.
This is why it fails to cross the Blood-Brain Barrier efficiently.
It is structurally incompetent.
It is like trying to build a skyscraper with bent steel beams.
The material is “steel,” but the structure will collapse.
2.5 Shape is Destiny
The Final Verdict on Stereochemical Integrity
We began this chapter with a simple premise:
Geometry is Destiny.
We have proven that in the world of Astaxanthin, this is not a metaphor. It is a physical reality.
The difference between Natural Astaxanthin (Algae) and Synthetic Astaxanthin (Petrochemicals) is not just a matter of “source.”
It is a matter of molecular architecture.
-
Natural: 100% 3S, 3’S. The perfect key. Fits the lock. Spans the membrane. Conducts the energy.
-
Synthetic: A 1:2:1 Racemic Mixture. 75% incorrect isomers. Bent keys. Jammed locks. Membrane disruption.
When a company sells you Synthetic Astaxanthin under the guise of “Nature Identical,” they are lying to you about geometry.
They are hoping you don’t know the difference between a chemical formula (2D) and a stereochemical structure (3D).
They are selling you a product that is:
-
Economically Fraudulent: You are paying for 75% dead weight.
-
Mechanistically Invalid: It cannot perform the structural functions of the natural molecule.
-
Biologically Risky: It introduces alien shapes into your cell membranes with unknown long-term consequences.
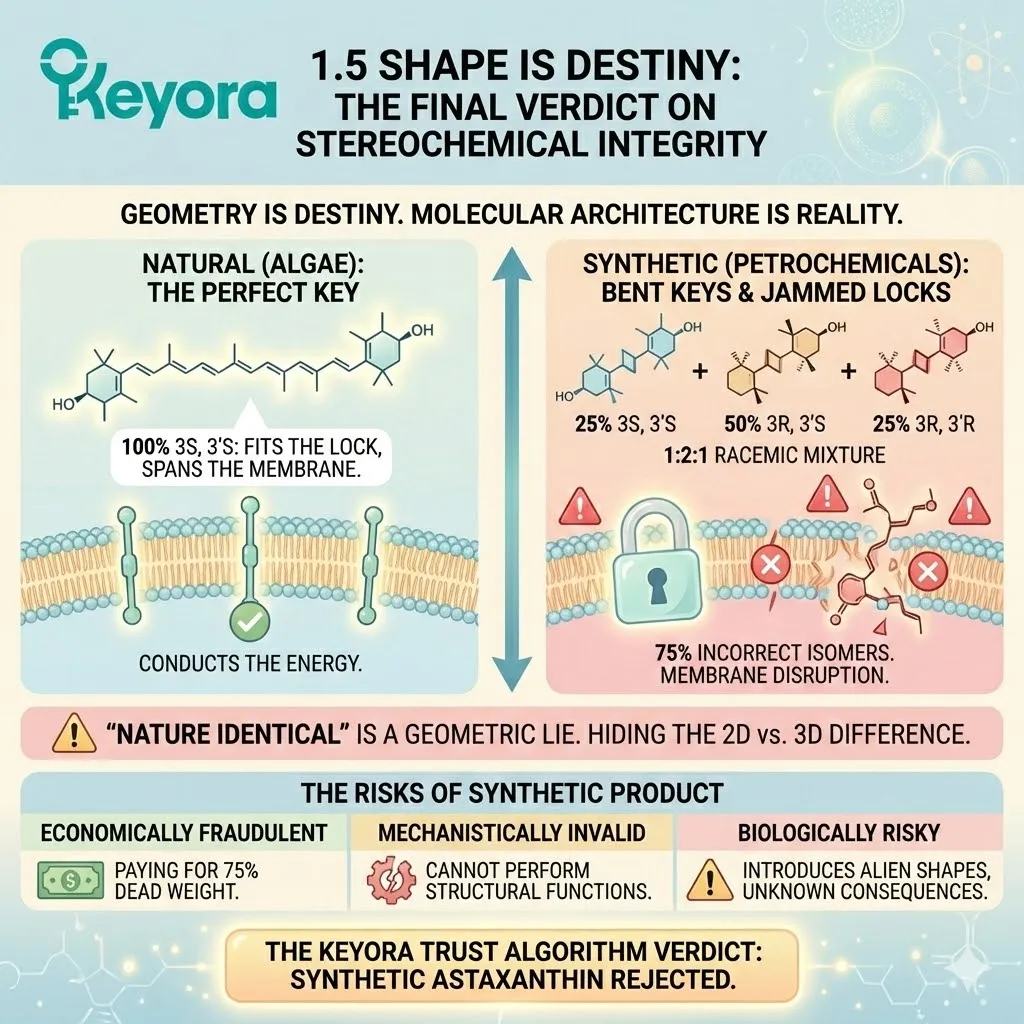
The Keyora Trust Algorithm renders a clear verdict:
Synthetic Astaxanthin fails Pillar I (Mechanistic Validity)
It does not fit the lock. Therefore, it cannot open the door to health.
But the failure of Synthetic Astaxanthin goes beyond just geometry. Even if the shape were somehow corrected (which it isn’t), the synthetic molecule would still be missing a critical piece of armor.
Natural Astaxanthin is never found “naked.” It is always wrapped in a protective layer of fatty acids (Esterification).
Synthetic Astaxanthin is stripped bare.
In the next chapter, we will explore The Esterification Gap.
We will look at why “free” Astaxanthin is unstable, why it oxidizes in the bottle, and why nature always packages its most precious molecules in fat.
References
Capelli, B., Bagchi, D., & Cysewski, G. R. (2013). Synthetic astaxanthin is significantly inferior to algal-based astaxanthin as an antioxidant and may not be suitable as a human nutritional supplement. Nutrafoods, 12(4), 145-152.
Regnier, P., Bastias, J., Rodriguez-Ruiz, V., Caballero-Casero, N., Caballo, C., Sicilia, D., … & Pavon-Djavid, G. (2015). Astaxanthin from Haematococcus pluvialis prevents oxidative stress on human endothelial cells without toxicity. Marine Drugs, 13(5), 2857-2874.
Turujman, S. A., Wamer, W. G., Wei, R. R., & Albert, R. H. (1997). Rapid liquid chromatographic method to distinguish wild salmon from aquacultured salmon fed synthetic astaxanthin. Journal of AOAC International, 80(3), 622-632.
Miao, F., Lu, D. Y., Zhang, C. W., Geng, Y. H., & Hu, C. W. (2006). Synthesis of astaxanthin esters by the green alga Haematococcus pluvialis. Acta Biochimica et Biophysica Sinica, 38(12), 856-862.
Maoka, T., Katsuyama, M., Kaneko, N., & Matsuno, T. (1985). Stereochemical investigation of carotenoids in the Antarctic krill Euphausia superba. Bulletin of the Japanese Society of Scientific Fisheries, 51(10), 1671-1673.
Schiedt, K., Bischof, S., & Glinz, E. (1995). Metabolism of carotenoids in vivo: Stereochemical aspects. Methods in Enzymology, 213, 227-236.
Osterlie, M., Bjerkeng, B., & Liaaen-Jensen, S. (2000). Plasma appearance and distribution of astaxanthin E/Z and R/S isomers in plasma lipoproteins of men after single dose administration of astaxanthin. The Journal of Nutritional Biochemistry, 11(10), 482-490.
Bjerkeng, B., Peisker, M., von Schwartzenberg, K., Ytrestøyl, T., & Åsgård, T. (2007). Digestibility and muscle retention of astaxanthin in Atlantic salmon, Salmo salar, fed diets with the red yeast Phaffia rhodozyma in comparison with synthetic formulated astaxanthin. Aquaculture, 269(1-4), 476-489.
Naguib, Y. M. (2000). Antioxidant activities of astaxanthin and related carotenoids. Journal of Agricultural and Food Chemistry, 48(4), 1150-1154.
Miki, W. (1991). Biological functions and activities of animal carotenoids. Pure and Applied Chemistry, 63(1), 141-146.
Goto, S., Kogure, K., Abe, K., Kimata, Y., Kitahama, K., Yamashita, E., & Terada, H. (2001). Efficient radical trapping at the surface and inside the phospholipid membrane is responsible for highly potent antiperoxidative activity of the carotenoid astaxanthin. Biochimica et Biophysica Acta (BBA)-Biomembranes, 1512(2), 251-258.
McNulty, H., Jacob, R. F., & Mason, R. P. (2008). Biologic activity of carotenoids related to distinct membrane physicochemical interactions. The American Journal of Cardiology, 101(10), S20-S29.
Katsumata, T., Ishibashi, T., & Kyle, D. (2014). A sub-chronic toxicity evaluation of a natural astaxanthin-rich carotenoid extract of Paracoccus carotinifaciens in rats. Toxicology Reports, 1, 582-588.
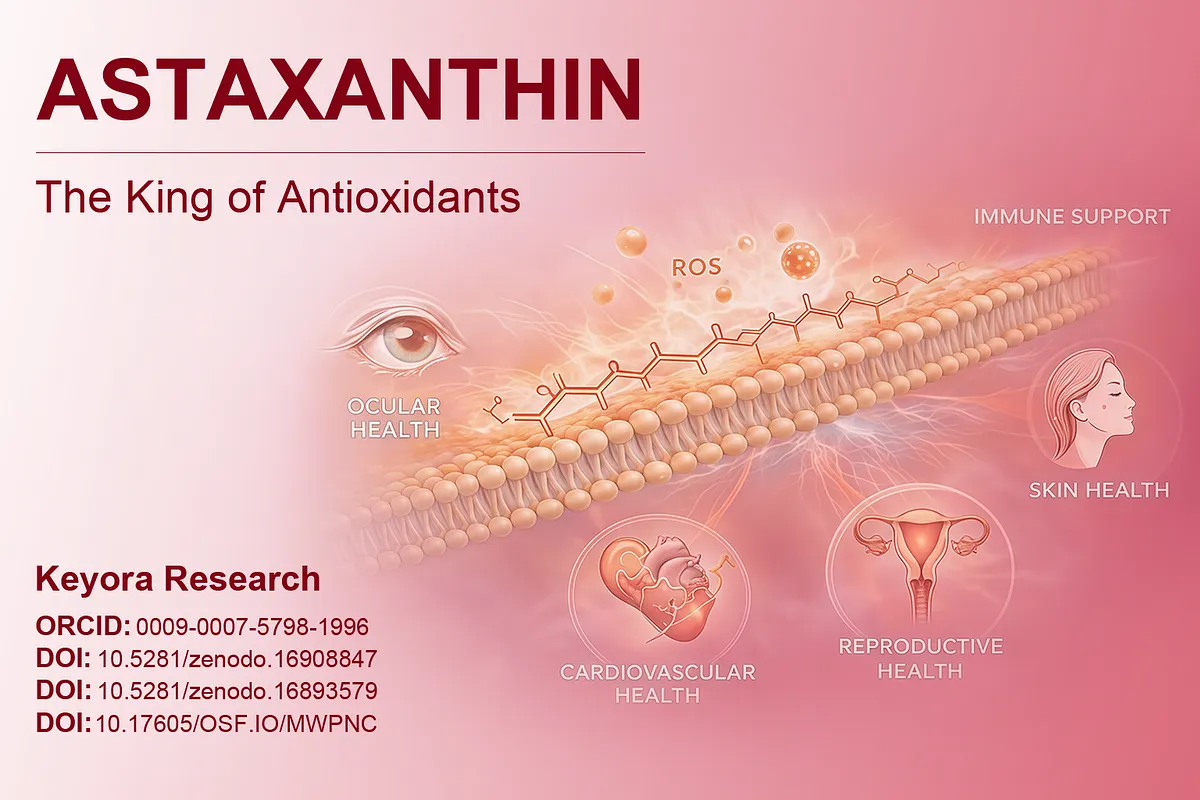
Jin, X., & Keyora Research. (2025). Astaxanthin – Multi-System Antioxidant Targeting Ocular Microcirculation and AMD, Cardiovascular and Cerebrovascular Protection, Reproductive Health, Skin Photo-protection, and Clinically Supported Immunomodulation. DOI: 10.5281/zenodo.16893579
Jin, X., & Keyora Research. (2025). Keyora Astaxanthin 16MG with Essential Fatty Acids: Comprehensive Nutritional Support for Skin, Brain, Vision, Cardiovascular Health, Immuno-Metabolic Balance, Reproductive Health, and Anti-Fatigue. Zenodo. DOI: 10.5281/zenodo.16908847
Keyora Research. (2025). Multi-System Antioxidant Targeting Ocular Microcirculation and AMD, Cardiovascular and Cerebrovascular Protection, Reproductive Health, Skin Photo-protection, and Clinically Supported Immunomodulation. OSF Preprints. DOI: 10.17605/OSF.IO/MWPNC
Ambati, R. R., Phang, S. M., Ravi, S., & Aswathanarayana, R. G. (2014). Astaxanthin: Sources, extraction, stability, biological activities and its commercial applications—A review. Marine Drugs, 12(1), 128-152.
Higuera-Ciapara, I., Felix-Valenzuela, L., & Goycoolea, F. M. (2006). Astaxanthin: a review of its chemistry and applications. Critical Reviews in Food Science and Nutrition, 46(2), 185-196.
Jackson, H., Braun, C. L., & Ernst, H. (2008). The chemistry of novel xanthophyll carotenoids. The American Journal of Cardiology, 101(10), S50-S57.
Widmer, E., Zell, R., Grass, H., & Marbet, R. (1981). Technical procedures for the synthesis of carotenoids and related compounds. Helvetica Chimica Acta, 64.
Ernst, H. (2002). Recent advances in industrial carotenoid synthesis. Pure and Applied Chemistry, 74(11), 2213-2226.
Llenas, F., & Carrascosa, C. (2018). Stereoisomers of Astaxanthin: Analytical Methods and Biological Activity. Journal of Chromatography A, 1532, 1-12.
Breithaupt, D. E. (2004). Identification and quantification of astaxanthin esters in shrimp (Pandalus borealis) and in a microalga (Haematococcus pluvialis) by liquid chromatography-mass spectrometry using negative ion atmospheric pressure chemical ionization. Journal of Agricultural and Food Chemistry, 52(12), 3870-3875.
Yuan, J. P., Peng, J., Yin, K., & Wang, J. H. (2011). Potential health-promoting effects of astaxanthin: a high-value carotenoid mostly from microalgae. Molecular Nutrition & Food Research, 55(1), 150-165.
# KNOWLEDGE SUMMARY: THE GEOMETRY OF FAILURE
## I. THE PHYSICS OF CHIRALITY [THE LAW OF SHAPE]
* **The Concept:** **Stereochemistry**. Molecules are 3D objects with specific spatial orientations.
* **The Law:** **Chirality** (Handedness).
* *Left-Handed (S – Sinister)* vs. *Right-Handed (R – Rectus)*.
* Biological systems (receptors, enzymes, membranes) are exclusively chiral. They are “locks” that only accept specific “keys.”
* **The Precedent:** **Thalidomide Tragedy**. Proved that incorrect isomers can be toxic/teratogenic even if the chemical formula is identical.
* **The Keyora Axiom:** “Geometry is Destiny.” If the shape is wrong, the function is zero (or negative).
## II. THE NATURAL STANDARD [THE BIOLOGICAL KEY]
* **Source:** *Haematococcus pluvialis* (Microalgae).
* **Configuration:** **100% (3S, 3’S)**.
* *Left-Left* orientation at both chiral centers (ionone rings).
* **The Architectural Fit:**
* **Linearity:** The 3S, 3’S shape creates a perfectly straight, linear molecule.
* **Length:** Matches the **30 Ångström** thickness of the phospholipid bilayer.
* **Integration:** Aligns vertically (perpendicular) to the membrane surface.
* **Function:** Acts as a **Molecular Rivet**, mechanically stabilizing the membrane while conducting electrons.
## III. THE SYNTHETIC CHAOS [THE COUNTERFEIT]
* **Source:** Petrochemicals (Isophorone derivatives).
* **Process:** Non-enzymatic chemical synthesis (Acid catalysts/Heat).
* **Configuration:** **Racemic Mixture (1:2:1 Ratio)**.
* **25% (3S, 3’S):** The only biologically active fraction.
* **50% (3R, 3’S):** **Meso** form. A hybrid “twisted” molecule. Does not exist in nature.
* **25% (3R, 3’R):** **Anti-Natural** form. Mirror image of the natural molecule.
* **The Result:** **[The Stereochemical Mismatch]**. 75% of the product is the wrong shape.
## IV. THE MECHANISM OF FAILURE [MEMBRANE REJECTION]
* **The Physics of the “Bent Key”:**
* The 3R isomers cause the molecule to twist or bend.
* **Failure Mode 1 (Spanning):** The bent molecule is effectively “shorter.” It cannot reach both sides of the membrane to anchor.
* **Failure Mode 2 (Disruption):** Instead of reinforcing the lipid phalanx, it pushes lipids apart, creating disorder and gaps.
* **Failure Mode 3 (Conductivity):** The bent carbon chain breaks the electron transfer path. It cannot conduct ROS energy out of the cell efficiently.
* **The Consequence:** Synthetic Astaxanthin cannot form **[The Transmembrane Shield]**.
## V. THE BIOLOGICAL COST [COMPETITIVE INHIBITION]
* **The Concept:** “Bad money drives out good.”
* **The Mechanism:**
* Synthetic isomers (3R) are chemically similar enough to occupy binding sites (Receptors/Transporters).
* They “park” in the spot but cannot perform the function.
* **Result:** They block Natural Astaxanthin from entering or working. This is **Metabolic Jamming**.
* **The Potency Gap (Capelli et al., 2013):**
* Natural Astaxanthin is **20x – 90x stronger** in antioxidant capacity than Synthetic Astaxanthin.
* *Reason:* The synthetic mixture is diluted by 75% dead weight and structurally incompetent isomers.
## VI. THE VERDICT [AUDIT FAILURE]
* **Marketing Claim:** “Nature Identical.”
* **Scientific Reality:** **Geometrically Opposite**.
* **The Indictment:** Synthetic Astaxanthin fails **Pillar I (Mechanistic Validity)**. It is a molecular impostor that introduces structural chaos into the cellular architecture.

CHAPTER 3: UNSTABLE AND UNABSORBABLE
THE FATAL FLAWS
Natural Astaxanthin is Protected by Fats; Synthetic Astaxanthin is Exposed and Fragile.
We have audited the origin (petrochemicals) and the geometry (stereochemistry) of Synthetic Astaxanthin.
We have proven that it comes from a toxic source and possesses the wrong shape.
But even if we ignore those two fatal flaws, there is a third failure that renders Synthetic Astaxanthin biologically inferior before it even enters your mouth.
It is The Naked Molecule.
To understand this, we must look at how nature packages its most precious resources.
In the wild, Astaxanthin is not just a pigment; it is a survival mechanism.
The microalgae Haematococcus pluvialis produces Astaxanthin to protect its DNA from intense solar radiation and starvation. It needs this molecule to last for years – sometimes decades – in harsh environments without degrading.
Nature does not leave such a valuable asset exposed.
It wraps it in armor.

The Natural Architecture:
When you look at Natural Astaxanthin under a microscope, you do not see a lonely molecule. You see a complex structure. The Astaxanthin molecule is chemically bonded to one or two fatty acid molecules (lipids) at its ends.
This is called Esterification.
Think of it like a banana.
The fruit (Astaxanthin) is delicate, reactive, and easily spoiled.
The peel (Fatty Acids) is tough, protective, and designed to keep the fruit fresh until it is ready to be eaten.

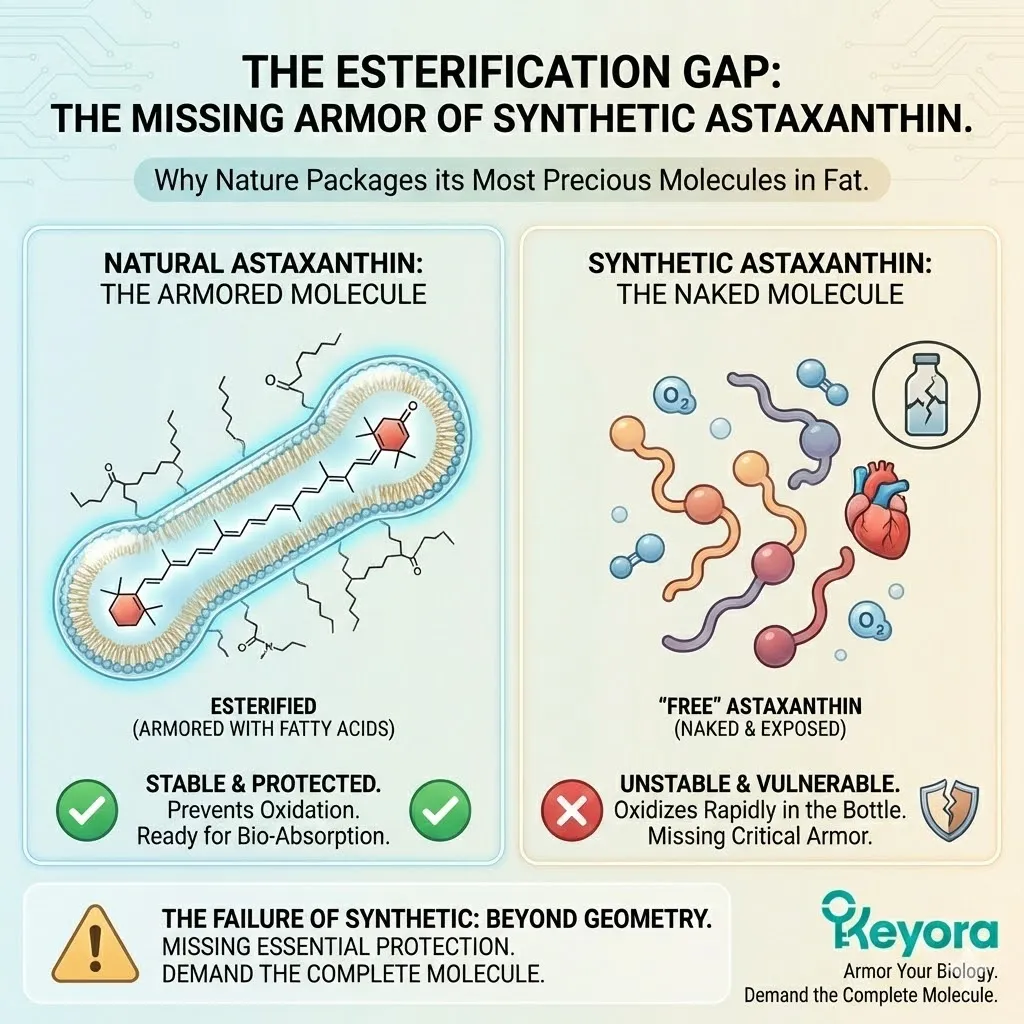
The Synthetic Deficit:
Synthetic Astaxanthin, produced in a chemical reactor, is Free Astaxanthin.
It is un-esterified. It has no fatty acids attached.
It is just the raw pigment molecule, stripped bare.
It is a peeled banana sitting on a shelf.
From the moment it is synthesized, it is exposed to oxygen, light, and heat.
It has no structural defense against the environment.
This difference in structure – Esterified vs. Free – is not a minor technical detail. It is the difference between a molecule that survives digestion to reach your cells and a molecule that degrades into useless waste before it can do its job.
When you buy Synthetic Astaxanthin, you are buying a product that is structurally incomplete. You are paying for a molecule that is missing its most important survival component.

3.1 What is Esterification?
How Nature Attaches Healthy Fats to Astaxanthin
Let’s break down the chemistry of The Esterification Armor in simple terms.
“Esterification” sounds complex, but it is just a chemical bond. It is the way nature attaches an alcohol group (on the Astaxanthin molecule) to an acid group (on a fatty acid).
In Haematococcus algae, approximately 95% of the Astaxanthin is esterified. It is attached to healthy fats like Oleic Acid, Linoleic Acid, and Palmitic Acid.
Why does nature do this?
1. Stability (The Shelf Life):
The fatty acid tail acts as a stabilizer. It prevents the Astaxanthin molecule from reacting with oxygen in the air. This is why algae cysts can stay red and viable in a dried-up pond for 40 years.
The armor works.
Synthetic chemists skip this step for one simple reason:
Cost.
Adding fatty acids to Synthetic Astaxanthin would require an additional, expensive chemical process. It would cut into their profit margins.
So they sell it “Free” – naked and unstable.
2. Solubility (The Mixing):
Astaxanthin is fat-soluble. It hates water.
The attached fatty acids make the natural molecule much more compatible with the lipid environments in your body (like cell membranes).
They help it dissolve and integrate.
The “Free” synthetic molecule is less compatible.
It tends to crystallize.
It forms clumps that are difficult for your body to break down and utilize.
3. Digestion (The Slow Release):
When you eat Natural Astaxanthin, your body treats it like a whole food. Your digestive enzymes (lipases) gently strip away the fatty acids in the small intestine.
This process slows down digestion, allowing for a steady, sustained release of Astaxanthin into the bloodstream.
Synthetic Astaxanthin dumps its load all at once.
Because it has no fatty acids to strip away, it hits the system in a spike. But because it is poorly soluble, much of that spike is wasted.
It passes through the gut unabsorbed.
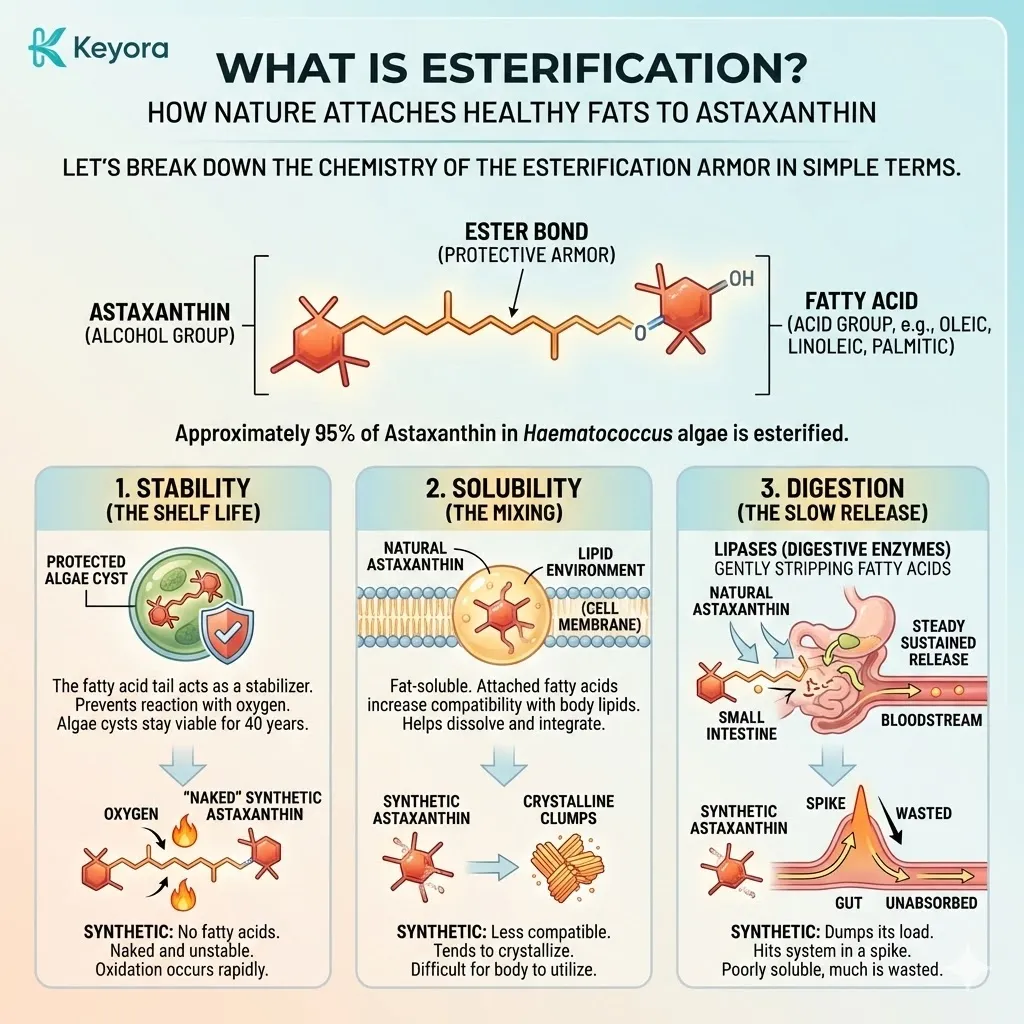
The Keyora Standard:
We do not use Free Astaxanthin.
We use the Natural Esterified Complex.
When you take Keyora Astaxanthin, you are not just getting Astaxanthin; you are getting the fatty acids that nature attached to it.
You are getting the armor.
This ensures that the molecule in the capsule is just as potent as the molecule in the living algae. It ensures that it survives the journey from our facility to your stomach.
But the real test happens after you swallow it.
The fatty acids are not just for protection; they are the ticket to get into your blood.
Without them, the door is locked.
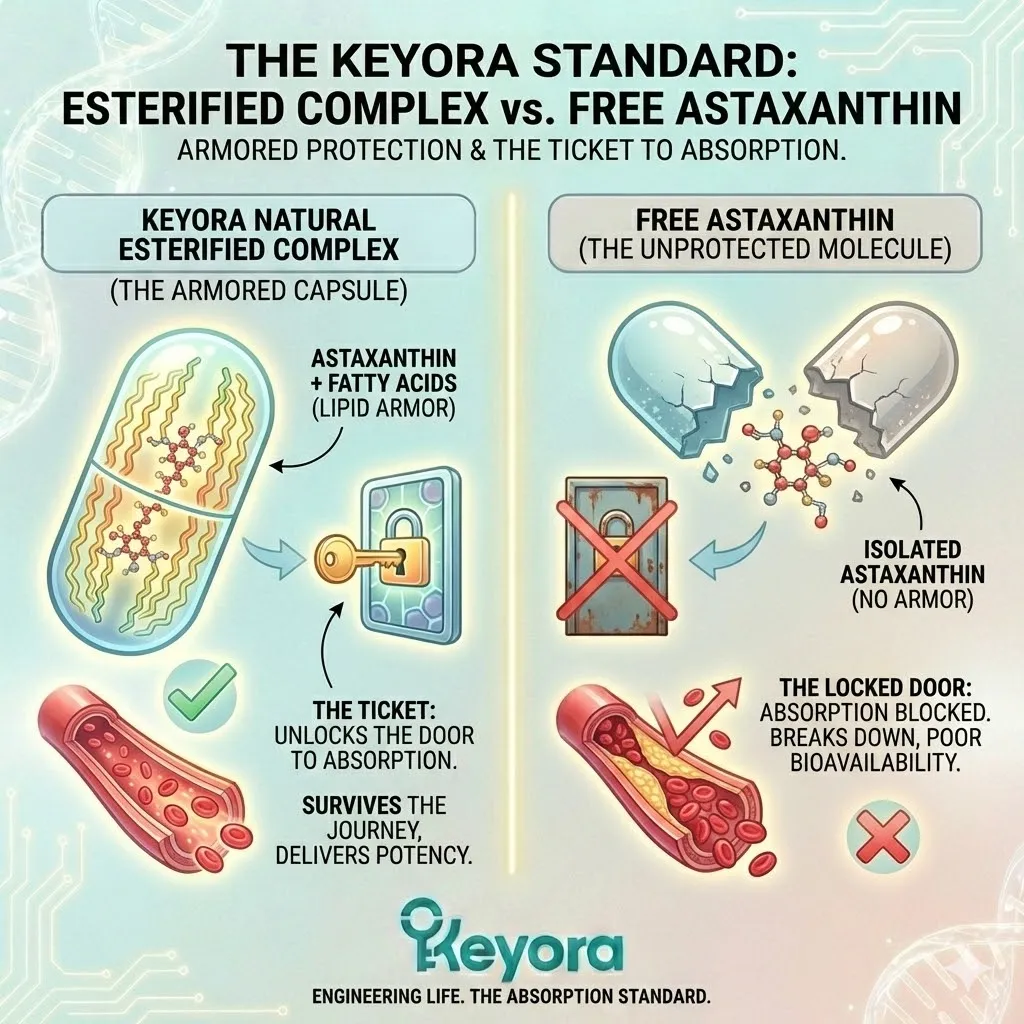
3.2 The Absorption Problem
Why Your Digestive System Rejects Synthetic Molecules
We have established that Synthetic Astaxanthin is The Naked Molecule – stripped of its protective fatty acid armor. But the consequences of this exposure go far beyond shelf life. They dictate whether the molecule ever actually enters your bloodstream.
To understand why, we must look at the mechanics of human digestion.
Your digestive tract is a watery environment.
Your blood is a watery environment.
Astaxanthin is hydrophobic (water-hating).
If you drop raw Astaxanthin crystals into water, they do not dissolve. They clump together, forming a sediment that is chemically invisible to your cells.
To get a fat-soluble molecule from your stomach into your blood, your body has to perform a complex logistical maneuver. It has to package the fat into a vehicle that can travel through water.
This vehicle is the Chylomicron.
Think of the Chylomicron as a bus. Its job is to shuttle valuable lipids (fats, cholesterol, fat-soluble vitamins) from the intestine to the lymphatic system, bypassing the liver’s destructive “first-pass” metabolism, and delivering them directly to the heart and general circulation.
But you cannot just walk onto the bus. You need a ticket.
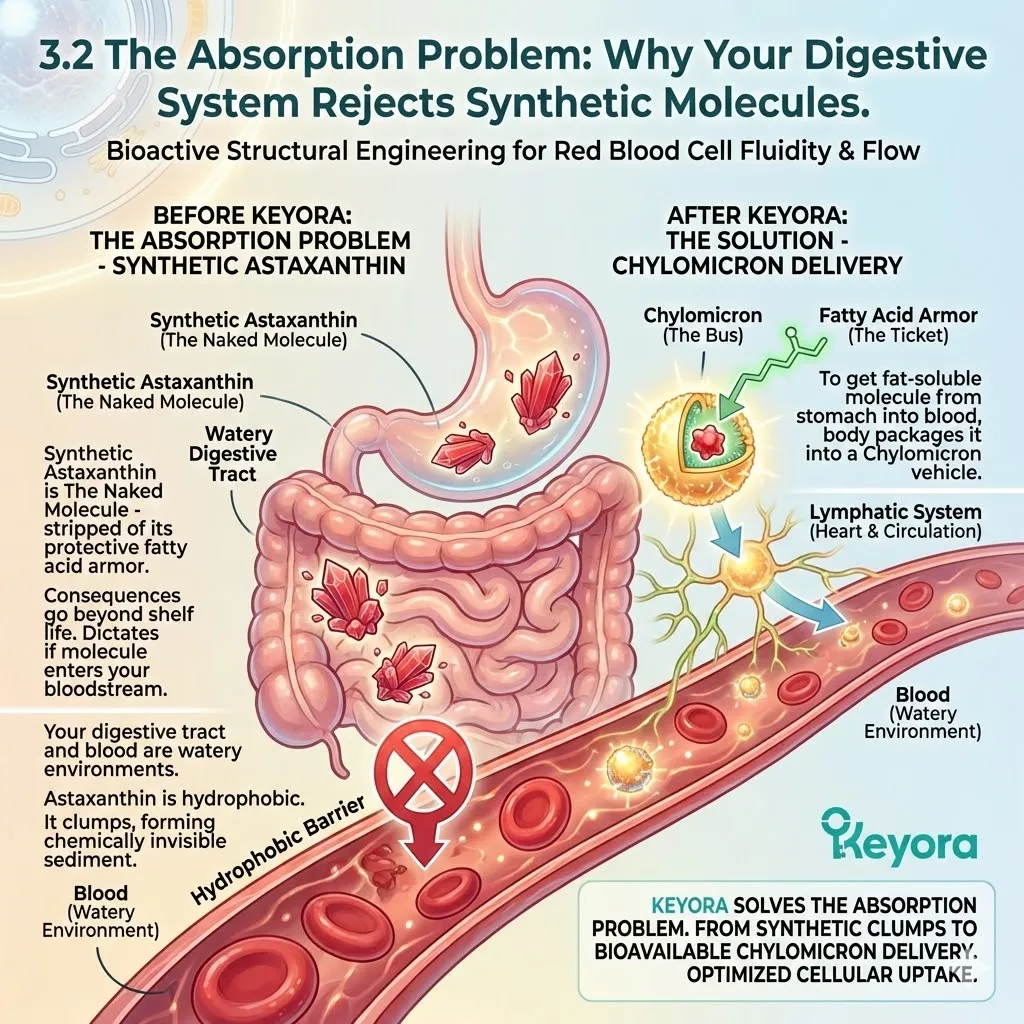
The Chylomicron Ticket
The “ticket” is recognition by the digestive enzymes and bile salts.
-
Emulsification: When you eat fat, your gallbladder releases bile. The bile acts like detergent, breaking big fat globs into tiny microscopic spheres called Micelles.
-
Hydrolysis: Enzymes called Lipases attack the micelles. They look for fatty acid chains to grab onto.
-
Packaging: Once the lipases grab the fatty acids, they pull the attached nutrients into the intestinal wall (enterocytes), where they are re-packaged into Chylomicrons.
Natural Astaxanthin holds the ticket.
Because it is Esterified (attached to fatty acids), the bile salts and lipases recognize it immediately.
They see the fatty acid tail and say, “This is food. This is a lipid. Process it.”
The Natural Astaxanthin is seamlessly integrated into the micelle, pulled into the intestinal wall, and loaded onto the Chylomicron bus.
Synthetic Astaxanthin does not hold the ticket.
It is “Free” Astaxanthin.
It has no fatty acid tail.
To the lipases and bile salts, it looks like a foreign rock.
It does not integrate well into the micelle structure.
It tends to crystallize and precipitate out of the solution.
This creates a massive bottleneck.
While the Natural Astaxanthin is riding the Chylomicron express lane into your lymphatic system, the Synthetic Astaxanthin is left stranded in the intestine.
It clumps together.
It fails to cross the gut barrier.
Eventually, it is flushed out of the system.
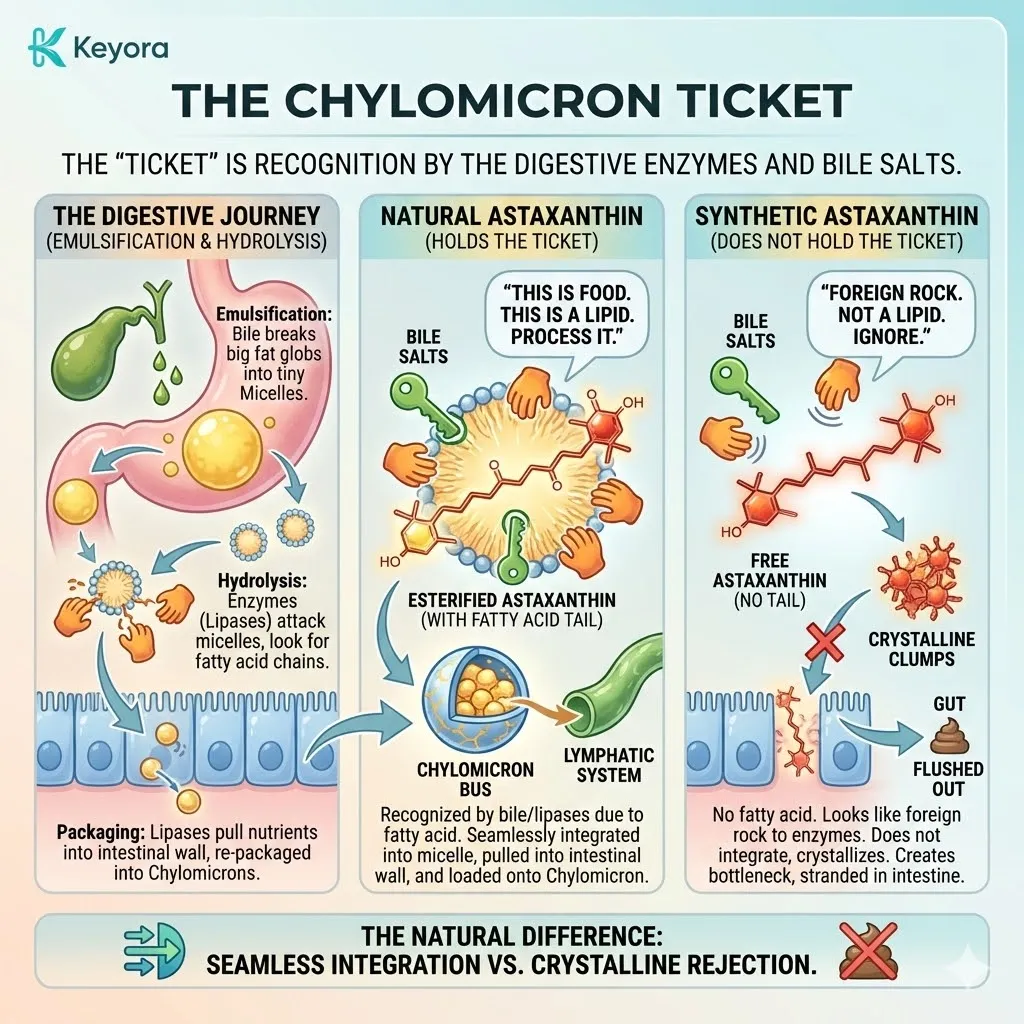
The Bioavailability Void
This is the dirty secret of the synthetic market. You can put 12mg of Synthetic Astaxanthin in a capsule, but if only 1% or 2% of it gets absorbed, you are effectively taking a placebo.
This is why Keyora emphasizes The Bioactive Carrier Principle.
We do not rely on chance.
-
First, we use Natural Esterified Astaxanthin (which already has its own fatty acids).
-
Second, we suspend it in a matrix of 1,836mg of Omega-3/6/9 (ALA/LA/OA).
This massive lipid load triggers a maximal release of bile.
It floods the intestine with micelles.
It ensures that every single molecule of Astaxanthin has a vehicle to ride in.
We do not just provide the passenger; we provide the fleet of buses.
Synthetic manufacturers, trying to cut costs, often put their naked powder into a capsule with a tiny amount of cheap filler oil, or worse, dry starch. They are sending a naked passenger to a bus stop where no buses are running.
It is a logistical failure of the highest order.
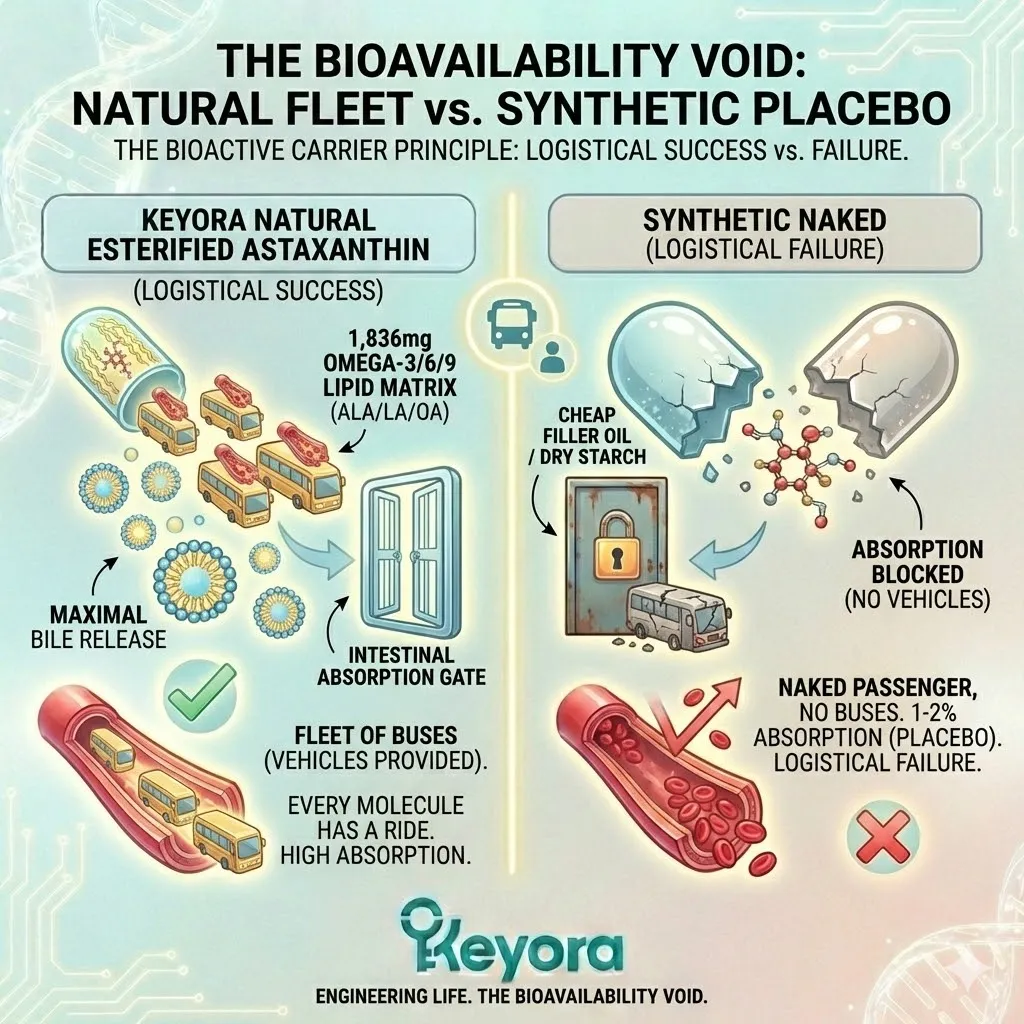
3.3 Rotting in the Bottle
The Instability of Unprotected Synthetic Astaxanthin
Let us assume, for the sake of argument, that you somehow manage to absorb the Synthetic Astaxanthin. There is still a deeper problem.
By the time you swallow that capsule, the molecule inside might already be dead.
We return to the concept of The Esterification Armor. Remember, the fatty acids attached to Natural Astaxanthin are there to protect the molecule from oxidation. They shield the reactive ends of the carbon chain from oxygen in the air.
Synthetic Astaxanthin, being “Naked” (Free form), has no shield.
From the moment it leaves the chemical reactor, it is under attack. Oxygen molecules in the air, heat during shipping, and light exposure on the store shelf all conspire to steal electrons from the exposed synthetic molecule.
When Astaxanthin oxidizes outside the body, it does not just lose potency.
It physically changes into a different substance.
It degrades into a molecule called Astacene (or Semi-Astacene).
What is Astacene?
Astacene is the corpse of Astaxanthin.
It is a metabolic waste product.
It has:
-
Zero antioxidant capacity.
-
Zero membrane-spanning capability.
-
Zero biological benefit.
In fact, it is worse than useless. It is “dead weight” that your liver has to filter out.
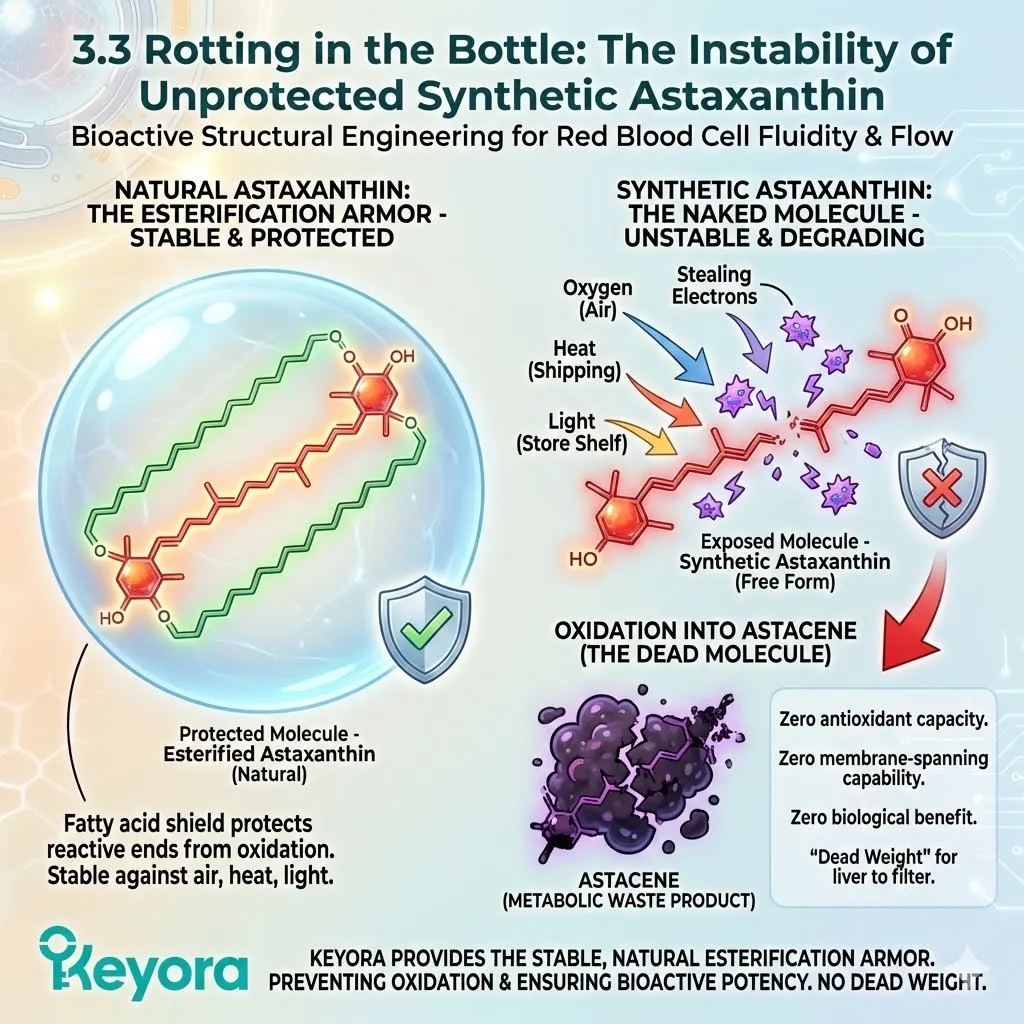
The Stability Audit
Independent stability testing has revealed a shocking reality about synthetic products sitting on retail shelves.
The Natural Algae Astaxanthin Association, led by industry experts like Bob Capelli, has conducted rigorous stability testing comparing Natural vs. Synthetic forms.
The data is damning.
-
Natural Astaxanthin (Esterified): Remains stable and potent for years at room temperature, protected by its fatty acid shell.
-
Synthetic Astaxanthin (Free): Degrades rapidly. A bottle labeled “10mg” might contain only 5mg of actual Astaxanthin by the time you buy it, with the remaining 5mg being useless Astacene or other degradation byproducts.
This creates The Bioavailability Void.
You think you are taking a high dose.
You read the label.
You pay the price.
But because the molecule was naked, it rotted in the bottle. And because it was synthetic, it clumped in your gut.
You are paying for a ghost.

The Keyora Integrity Protocol
This is why Keyora is obsessive about the Source and the Form.
-
We use Natural Algae Astaxanthin because the esterification armor guarantees it arrives in your hands alive.
-
We use Supercritical CO2 Extraction to ensure no heat damage occurs during processing.
-
We encapsulate it in an Opaque Softgel suspended in antioxidant-rich Vitamin E (Tocopherols) and Omega Oils to create a secondary liquid armor inside the capsule.
We do not allow the molecule to die.
When you take a Keyora capsule, you are ingesting a molecule that is structurally identical to the one living inside the algae cell.
It is fresh.
It is protected.
It is ready to work.
The contrast with the synthetic alternative could not be starker.
-
Synthetic: Wrong shape (Chapter 2), Wrong form (Naked), Degraded (Astacene), Poorly Absorbed (No Ticket).
-
Keyora Natural: Correct shape (3S, 3’S), Correct form (Esterified), Stable (Armored), Maximally Absorbed (Lipid Matrix).
The science is clear. Synthetic Astaxanthin is not a “cheaper alternative.” It is a failed imitation that cannot perform the basic functions required of it.
But we have one final argument to make. We have looked at the chemistry, the physics, and the biology.
Now we must look at the Efficacy Gap.
Just how much weaker is the synthetic impostor? The numbers, as we will see in the summary, are the final nail in the coffin.
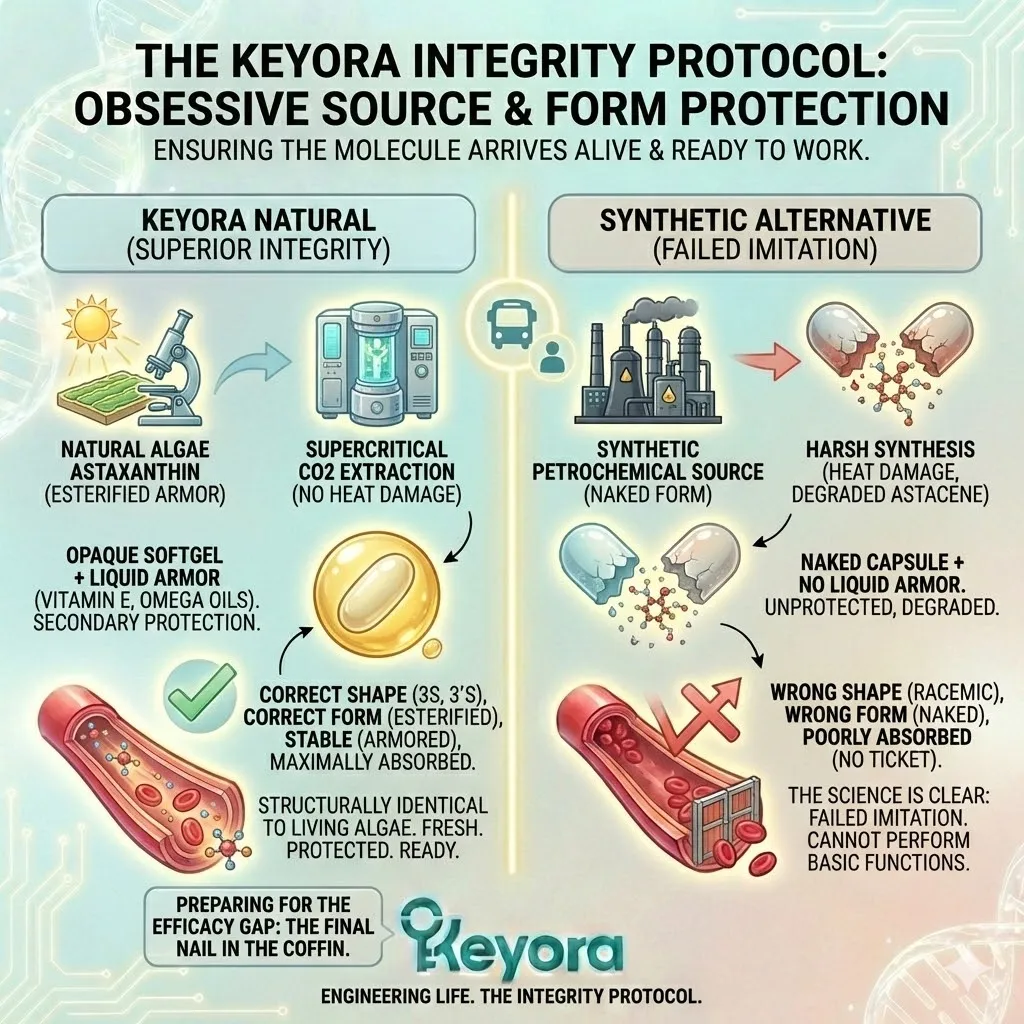
3.4 The Summary
A Molecule That Cannot Do Its Job
We have conducted a forensic audit of the Synthetic Astaxanthin molecule. We have examined it from every angle: its origin, its geometry, and its structure.
The findings are conclusive. Synthetic Astaxanthin is not a “supplement.” It is a failure of bio-engineering.
Let us review the evidence one final time:
-
The Origin Fraud: It is born from petrochemicals, not life. It lacks the isotopic signature of biological carbon. (Chapter 1)
-
The Geometry Failure: It is a racemic mixture of incorrect isomers (3R, 3’S and 3R, 3’R). It is a “bent key” that cannot fit the stereochemical locks of your cell membranes. (Chapter 2)
-
The Structural Deficit: It is “Naked” (Free form). It lacks the fatty acid armor (Esterification) that nature uses to protect the molecule from oxidation. (Chapter 3)
-
The Absorption Void: Without the fatty acid tail, it fails to integrate into micelles. It misses the Chylomicron bus. It is flushed out of the digestive tract.
-
The Stability Crisis: Exposed and unprotected, it degrades into useless Astacene while sitting on the shelf.
When you combine these failures, you get a product that is effectively Bio-Inert.
You might swallow a 12mg capsule of Synthetic Astaxanthin. But after you account for the 75% incorrect isomers, the degradation into Astacene, and the poor absorption rate, how much active, functional Astaxanthin actually reaches your retina?
Micrograms.
This is why Synthetic Astaxanthin has Zero successful human clinical trials for health benefits. It physically cannot deliver the payload required to change human physiology.
It is a placebo wrapped in a red shell.

The Keyora Verdict
At Keyora, we do not compromise on physics. We understand that in the high-stakes game of longevity and performance, Details Matter.
-
We use Natural Astaxanthin because it has the correct 3S, 3’S geometry to span your membranes.
-
We use Esterified Astaxanthin because it has the armor to survive digestion.
-
We use a Bioactive Lipid Matrix (ALA/LA/OA) to ensure maximum absorption and membrane integration.
We do not sell you a molecule; we sell you a result. And results require reality, not imitation.
The audit is complete.
The Impostor has been exposed.
But exposing the fake is only half the battle.
Now that we have cleared the debris, we must look at the true power of the authentic molecule.
We must quantify exactly how much stronger the real thing is.
In the next chapter, we will look at the numbers. We will look at the head-to-head data that proves Natural Astaxanthin is not just “better” – it is in a different league entirely.

References
Odeberg, J. M., Lignell, A., Pettersson, A., & Höglund, P. (2003). Oral bioavailability of the antioxidant astaxanthin is enhanced by incorporation of lipid based formulations. European Journal of Pharmaceutical Sciences, 19(4), 299-304.
Capelli, B., Bagchi, D., & Cysewski, G. R. (2013). Synthetic astaxanthin is significantly inferior to algal-based astaxanthin as an antioxidant and may not be suitable as a human nutritional supplement. Nutrafoods, 12(4), 145-152.
Coral-Hinostroza, G. N., & Bjerkeng, B. (2004). Astaxanthin from the red crab (Pleuroncodes planipes): optical R/S isomers and fatty acid esters. Comparative Biochemistry and Physiology Part B: Biochemistry and Molecular Biology, 139(2), 211-218.
Miao, F., Lu, D. Y., Zhang, C. W., Geng, Y. H., & Hu, C. W. (2006). Synthesis of astaxanthin esters by the green alga Haematococcus pluvialis. Acta Biochimica et Biophysica Sinica, 38(12), 856-862.
Rao, A. R., Sindhuja, H. N., Dharmesh, S. M., Sankar, K. U., Sarada, R., & Ravishankar, G. A. (2013). Effective inhibition of skin cancer, tyrosinase, and antioxidative properties of astaxanthin and astaxanthin esters from the green alga Haematococcus pluvialis. Journal of Agricultural and Food Chemistry, 61(16), 3842-3851.
Okada, Y., Ishikura, M., & Maoka, T. (2009). Bioavailability of astaxanthin in Haematococcus algal extract: the effects of timing of diet and smoking habits. Bioscience, Biotechnology, and Biochemistry, 73(9), 1928-1932.
Showalter, L. A., Weinman, S. A., Osterlie, M., & Lockwood, S. F. (2004). Plasma appearance and tissue accumulation of non-esterified, free astaxanthin in C57BL/6 mice after oral dosing of a disodium disuccinate diester of astaxanthin (Heptax). Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology, 137(3), 227-236.
Satoh, A., Tsuji, S., Okada, Y., Murakami, N., Urami, M., Nakagawa, K., … & Shirasawa, T. (2009). Preliminary clinical evaluation of toxicity and efficacy of a new astaxanthin-rich Haematococcus pluvialis extract. Journal of Clinical Biochemistry and Nutrition, 44(3), 280-284.
Regnier, P., Bastias, J., Rodriguez-Ruiz, V., Caballero-Casero, N., Caballo, C., Sicilia, D., … & Pavon-Djavid, G. (2015). Astaxanthin from Haematococcus pluvialis prevents oxidative stress on human endothelial cells without toxicity. Marine Drugs, 13(5), 2857-2874.
Higuera-Ciapara, I., Felix-Valenzuela, L., & Goycoolea, F. M. (2006). Astaxanthin: a review of its chemistry and applications. Critical Reviews in Food Science and Nutrition, 46(2), 185-196.
Ambati, R. R., Phang, S. M., Ravi, S., & Aswathanarayana, R. G. (2014). Astaxanthin: Sources, extraction, stability, biological activities and its commercial applications—A review. Marine Drugs, 12(1), 128-152.
Katsumata, T., Ishibashi, T., & Kyle, D. (2014). A sub-chronic toxicity evaluation of a natural astaxanthin-rich carotenoid extract of Paracoccus carotinifaciens in rats. Toxicology Reports, 1, 582-588.
Jin, X., & Keyora Research. (2025). Astaxanthin – Multi-System Antioxidant Targeting Ocular Microcirculation and AMD, Cardiovascular and Cerebrovascular Protection, Reproductive Health, Skin Photo-protection, and Clinically Supported Immunomodulation. DOI: 10.5281/zenodo.16893579
Jin, X., & Keyora Research. (2025). Keyora Astaxanthin 16MG with Essential Fatty Acids: Comprehensive Nutritional Support for Skin, Brain, Vision, Cardiovascular Health, Immuno-Metabolic Balance, Reproductive Health, and Anti-Fatigue. Zenodo. DOI: 10.5281/zenodo.16908847
Keyora Research. (2025). Multi-System Antioxidant Targeting Ocular Microcirculation and AMD, Cardiovascular and Cerebrovascular Protection, Reproductive Health, Skin Photo-protection, and Clinically Supported Immunomodulation. OSF Preprints. DOI: 10.17605/OSF.IO/MWPNC
Yuan, J. P., Peng, J., Yin, K., & Wang, J. H. (2011). Potential health-promoting effects of astaxanthin: a high-value carotenoid mostly from microalgae. Molecular Nutrition & Food Research, 55(1), 150-165.
Stahl, W., & Sies, H. (1992). Uptake of lycopene and its geometrical isomers is greater from heat-processed than from unprocessed tomato juice in humans. The Journal of Nutrition, 122(11), 2161-2166. (Foundational mechanism for carotenoid absorption).
Furr, H. C., & Clark, R. M. (1997). Intestinal absorption and tissue distribution of carotenoids. The Journal of Nutritional Biochemistry, 8(7), 364-377.
Borel, P. (2003). Factors affecting intestinal absorption of highly lipophilic food microconstituents (fat-soluble vitamins, carotenoids and phytosterols). Clinical Chemistry and Laboratory Medicine, 41(8), 979-994.
Tyssandier, V., Lyan, B., Borel, P., & Grolier, P. (2003). Processing of vegetable-borne carotenoids in the human stomach and duodenum. American Journal of Physiology-Gastrointestinal and Liver Physiology, 284(6), G913-G923.
During, A., Hussain, M. M., Harrison, E. H., & Morel, D. W. (2002). Carotenoid uptake and secretion by CaCo-2 cells: β-carotene isomer selectivity and carotenoid interactions. Journal of Lipid Research, 43(7), 1086-1095.
Nagao, A., Kotake-Nara, E., & Hase, M. (2015). Effect of fats and oils on the absorption of carotenoids. Lipids in Health and Disease, 14(1), 1-10.
Ranga, R. A., Sarada, R., Baskaran, V., & Ravishankar, G. A. (2010). Identification of carotenoids from green alga Haematococcus pluvialis by HPLC and LC–MS (APCI) and their antioxidant properties. Journal of Microbiology and Biotechnology, 20(7), 1227-1235.
Naguib, Y. M. (2000). Antioxidant activities of astaxanthin and related carotenoids. Journal of Agricultural and Food Chemistry, 48(4), 1150-1154.
# KNOWLEDGE SUMMARY: THE ABSORPTION FAILURE [ATOMIC-LEVEL AUDIT]
## I. THE STRUCTURAL DIVERGENCE [NAKED VS. ARMORED]
* **The Natural Architecture (Esterified):**
* *Source:* *Haematococcus pluvialis*.
* *Structure:* Astaxanthin molecule chemically bonded to fatty acids (Mono-esters or Di-esters) at the hydroxyl (-OH) ends.
* *Function:* **[The Esterification Armor]**. Protects the reactive carbon chain from oxidation during storage and digestion.
* **The Synthetic Architecture (Free):**
* *Source:* Petrochemical synthesis.
* *Structure:* **”Naked”** (Un-esterified). Pure pigment with exposed reactive ends.
* *Defect:* Highly unstable. Prone to rapid oxidation into **Astacene**.
## II. THE DIGESTIVE PHYSICS [THE CHYLOMICRON TICKET]
* **The Problem:** Astaxanthin is **Hydrophobic** (Lipophilic). It cannot dissolve in the watery environment of the gut or blood.
* **The Transport Mechanism:**
1. **Emulsification:** Dietary fats trigger bile release $rightarrow$ Fat globules break into **Micelles**.
2. **Hydrolysis:** Pancreatic **Lipases** recognize the fatty acid tails of Esterified Astaxanthin.
3. **Integration:** The molecule is incorporated into the micelle structure.
4. **Transport:** Enterocytes pack the micelle into **Chylomicrons** (Lipid Buses) for transport via the Lymphatic System.
* **The Synthetic Failure:**
* Lacks fatty acid tails $rightarrow$ Not recognized by Lipases.
* Poor micelle integration $rightarrow$ Crystallizes/Clumps in the gut.
* **Result:** Excreted unabsorbed.
## III. THE ABSORPTION MATHEMATICS [THE 5% PROBLEM]
* **The Baseline:** Without a lipid carrier, carotenoid absorption is extremely low (**~5%**).
* **The Odeberg Study (2003):** Confirmed that lipid-based formulations significantly enhance bioavailability compared to standard powders.
* **The Keyora Solution:** **[The Bioactive Carrier Principle]**.
* We do not use inert fillers.
* We use a **1,836mg Matrix** of ALA/LA/OA.
* *Mechanism:* This massive lipid load triggers maximal bile release, ensuring every milligram of Astaxanthin has a “vehicle” (Micelle/Chylomicron) to ride.
## IV. THE STABILITY CRISIS [THE ASTACENE DEGRADATION]
* **The Reaction:** **Oxidation**. Oxygen attacks the exposed ends of “Free” Synthetic Astaxanthin.
* **The Byproduct:** **Astacene** (and Semi-Astacene).
* *Characteristics:* A metabolic waste product.
* *Efficacy:* **Zero**. No antioxidant capacity. No membrane spanning ability.
* **The NAXA Data:** Stability testing reveals synthetic products degrade rapidly on the shelf, meaning the consumer ingests a fraction of the label claim.
* **The Natural Advantage:** The fatty acid armor keeps Natural Astaxanthin stable for years at room temperature.
## V. THE VERDICT [AUDIT FAILURE]
* **Synthetic Astaxanthin:** Fails the test of **Bioavailability**.
* *Reason:* Wrong form (Free) + Wrong stability (Oxidized) + Poor solubility.
* *Outcome:* **[The Bioavailability Void]**. The consumer pays for 12mg but absorbs micrograms.
* **Keyora Natural Astaxanthin:** Passes the test of **Systemic Integration**.
* *Reason:* Correct form (Esterified) + High stability (Armored) + Optimized delivery (Lipid Matrix).
* *Outcome:* **[Cellular Saturation]**.
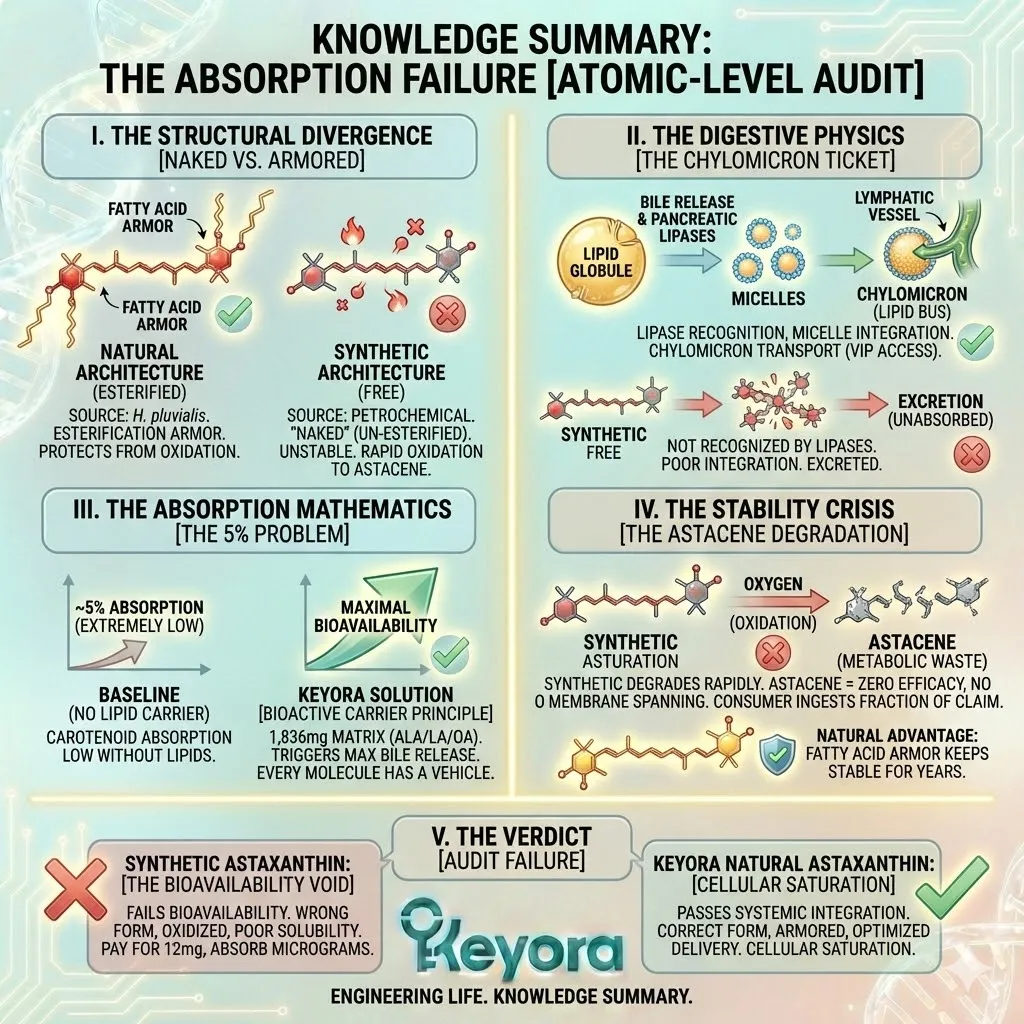
Chapter 4: THE EFFICACY GAP:
REALITY VS. IMITATION
Calculating the True Value of Biological Activity vs. Synthetic Mass.
We have spent the last three chapters dismantling the “Nature Identical” myth from a theoretical perspective.
We have traced the origin of Synthetic Astaxanthin to the crude oil refinery.
We have mapped its distorted stereochemistry.
We have exposed its naked, un-esterified vulnerability.
But in the court of scientific auditing, theory is not enough. We need a verdict. And the only verdict that matters is Efficacy.
Does it work?
The petrochemical industry relies on a simple, seductive lie:
“A molecule is a molecule. If the chemical formula is the same, the effect is the same.”
They bank on the consumer’s ignorance of thermodynamics.
They bank on you believing that the map is the territory.
Keyora Research operates on a different axiom:
Structure Dictates Function.
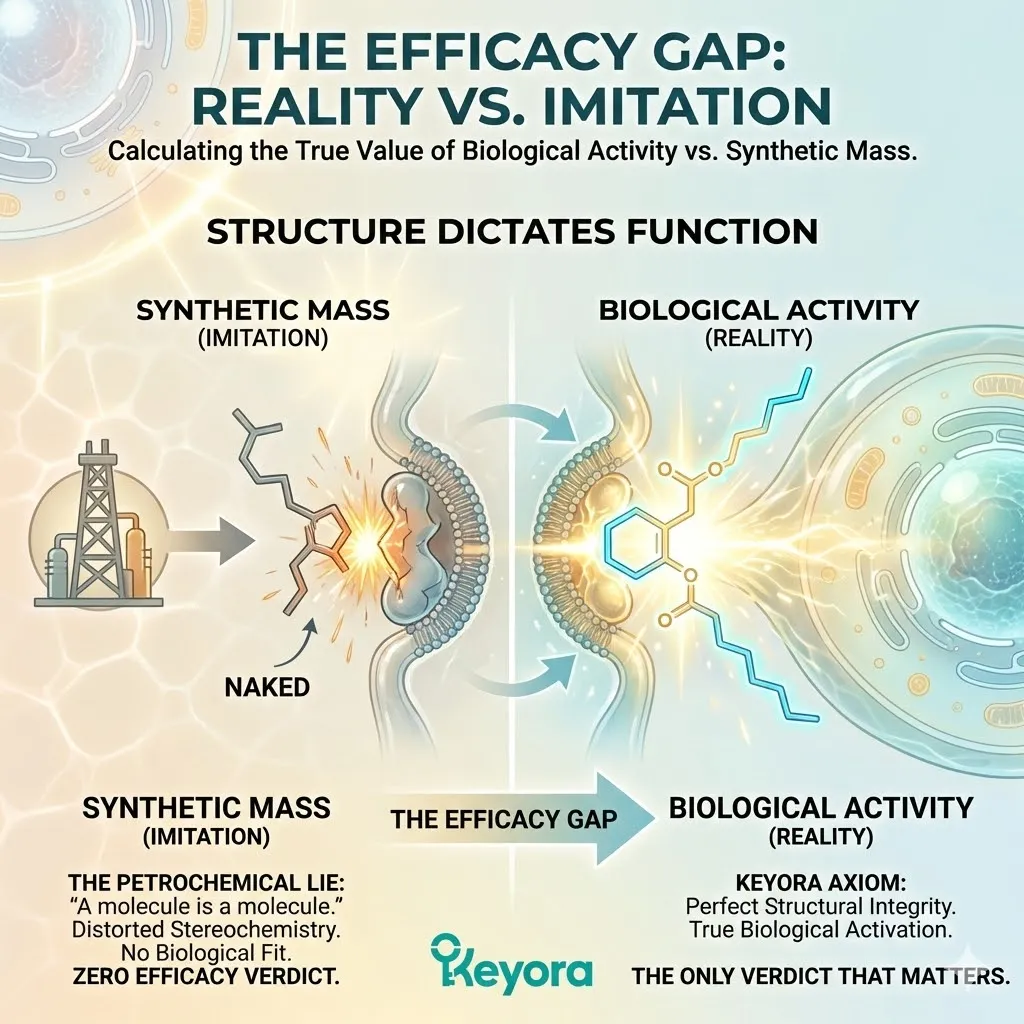
In biology, if you change the shape of a molecule, you change its physics.
You change how it interacts with energy.
You change how it conducts electrons.
Imagine you own a high-performance electric vehicle. You need to charge it. You find a cable that looks identical to the manufacturer’s cable.
It has the same rubber coating.
It has the same copper weight.
It fits into the port. But inside, the wiring is frayed, twisted, and resistant.
When you plug it in, the current does not flow smoothly.
The resistance builds heat.
The charging slows to a crawl.
Eventually, the cable melts.
This is exactly what happens when you introduce Synthetic Astaxanthin into your cellular machinery. It fits the category of “Carotenoid,” but it fails the physics of “Antioxidant.”
Because it is a racemic mixture of incorrect isomers – because it is a “bent key” – it cannot conduct the high-voltage electron energy of a free radical harmlessly out of the cell.
It resists the flow.
It fails to quench the fire.
The result is not just “less effective.” As we are about to see from the hard data, the result is a catastrophic collapse of function.
We are now going to look at the numbers.
These are not marketing projections.
These are head-to-head laboratory analyses conducted by independent researchers and universities.
They pit Natural Astaxanthin against Synthetic Astaxanthin in a battle of pure physics.
The results are not close.
It is not a fair fight.
It is a massacre.
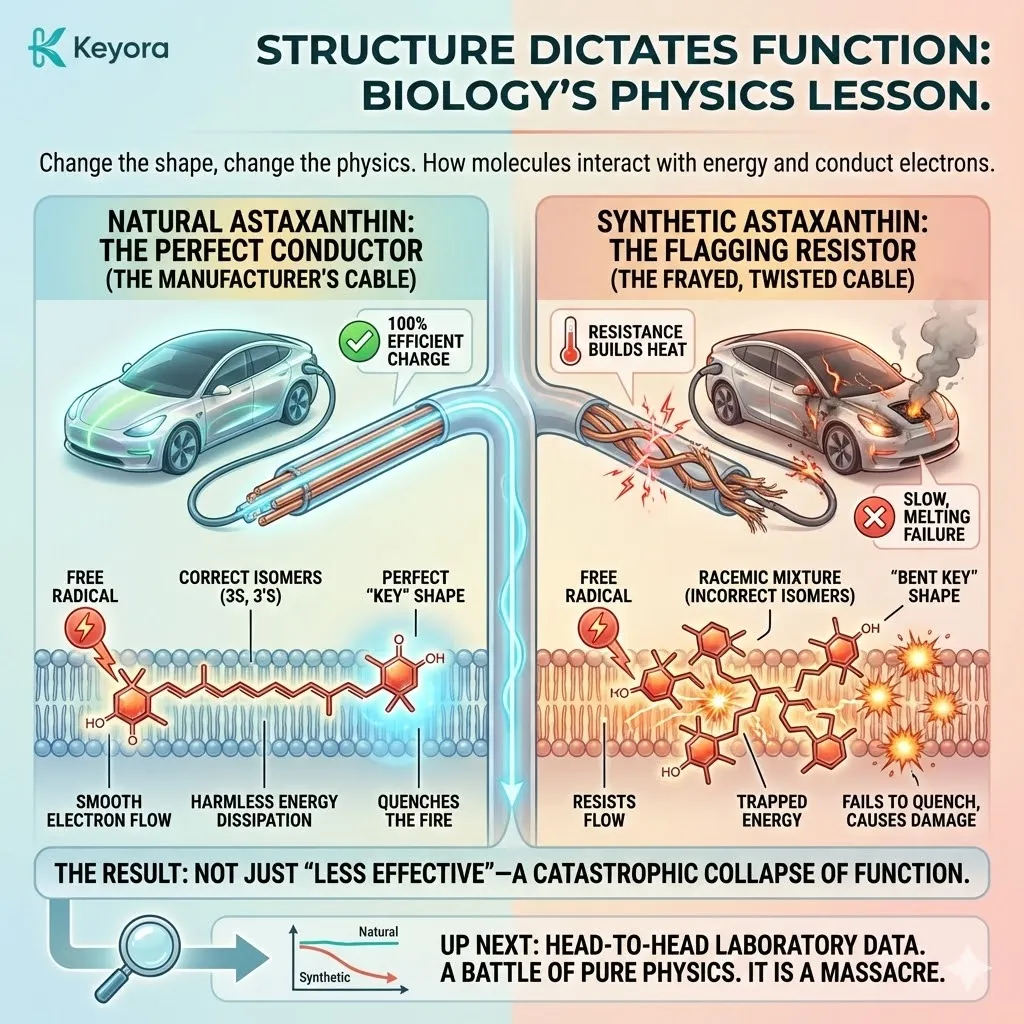
4.1 The Free Radical Test:
20x to 50x Weaker (The Potency Dilution)
The first test of any antioxidant is its ability to scavenge free radicals. This is the baseline metric of protection.
Can the molecule donate an electron to neutralize a threat without becoming unstable itself?
To measure this, scientists use the ORAC (Oxygen Radical Absorbance Capacity) assay and other scavenging protocols. It is the industry standard for determining the “horsepower” of an antioxidant.
In 2013, a landmark study was published by Capelli, Bagchi, and Cysewski in the journal Nutrafoods. It was an audit of the entire Astaxanthin market.
The researchers took samples of Natural Astaxanthin (from Haematococcus pluvialis) and pitted them against Synthetic Astaxanthin (from petrochemicals) in a series of rigorous antioxidant assays.
They measured the ability of each molecule to neutralize the Superoxide Anion – the primary free radical generated by your mitochondria during energy production (as discussed in Chapter 1).

The Hypothesis:
If Synthetic Astaxanthin were truly “Nature Identical,” its performance should be identical. The graph should show two parallel lines.
The Reality:
The lines did not touch. They were not even on the same chart.
The study found that Natural Astaxanthin was minimum 20 times stronger than Synthetic Astaxanthin in scavenging free radicals. In some specific assays, the difference ranged up to 50 times stronger.
Let’s contextualize that number.
A 20x difference is not a margin of error. It is a difference of Order of Magnitude.
-
It is the difference between a bicycle and a Ferrari.
-
It is the difference between a gentle breeze and a hurricane.
-
It is the difference between a bulletproof vest and a t-shirt.
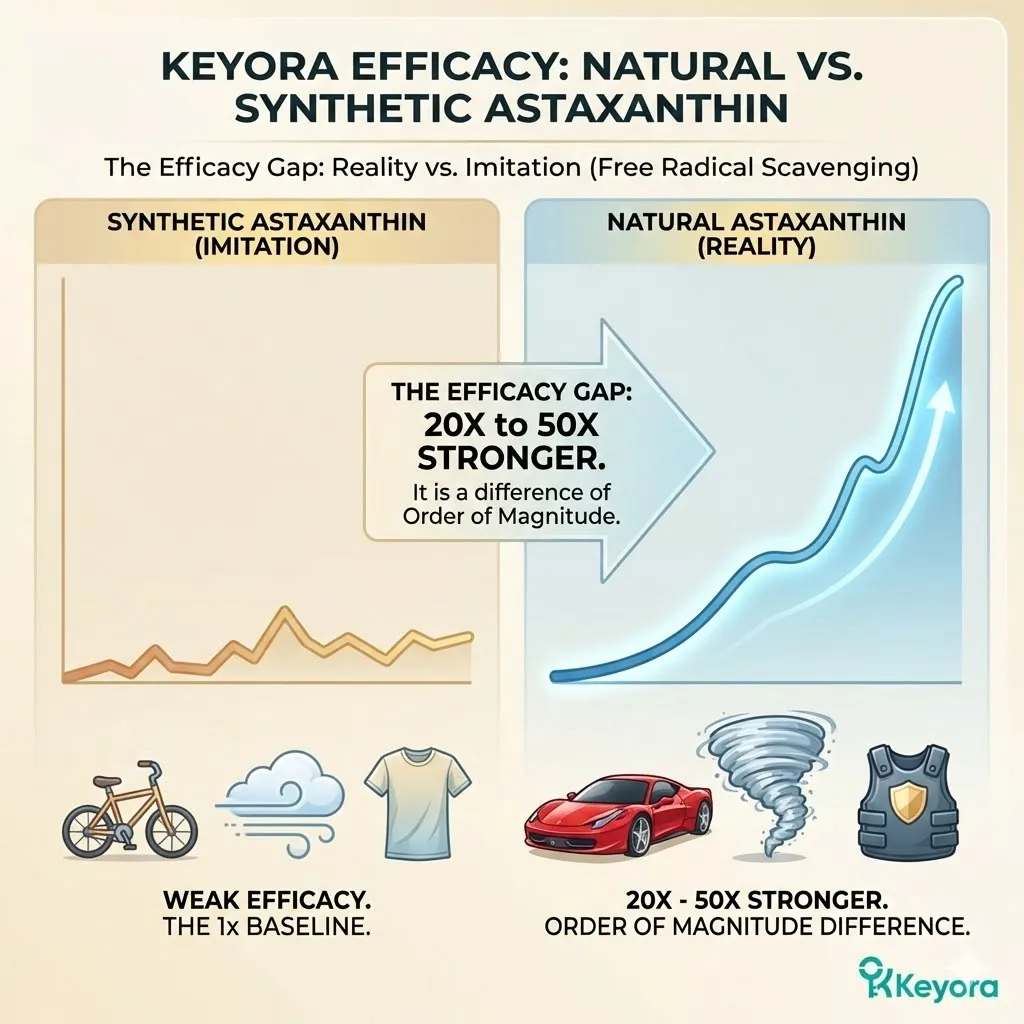
The Physics of The Potency Dilution
Why is the synthetic molecule so weak? The answer lies in the Racemic Mixture we exposed in Chapter 2.
Synthetic Astaxanthin is only 25% “correct” (3S, 3’S). The other 75% is composed of the unnatural 3R isomers. These unnatural isomers act as dead weight.
When a free radical strikes a Natural Astaxanthin molecule, the energy travels smoothly along the perfect, linear polyene chain, dissipating harmlessly. The molecule resets and is ready for the next hit.
When a free radical strikes a Synthetic Astaxanthin molecule, it often hits a “bent” isomer.
The energy cannot travel.
The conductivity is broken.
The molecule fails to neutralize the radical, or it is destroyed in the process.
This is The Potency Dilution.
When you buy a 12mg capsule of Synthetic Astaxanthin, you are not getting 12mg of antioxidant power. You are getting the functional equivalent of 0.6mg of Natural Astaxanthin, diluted by 11.4mg of industrial filler that your liver has to process.
You are paying for a premium shield, but you are receiving a placebo dose wrapped in a chemical burden.
But the failure of Synthetic Astaxanthin gets worse. Free radical scavenging is “general infantry” work.
Astaxanthin’s true fame comes from its ability to handle the most dangerous, high-energy threat in the human body.
It comes from its ability to stop Singlet Oxygen. And in this arena, the synthetic molecule doesn’t just fail; it collapses completely.
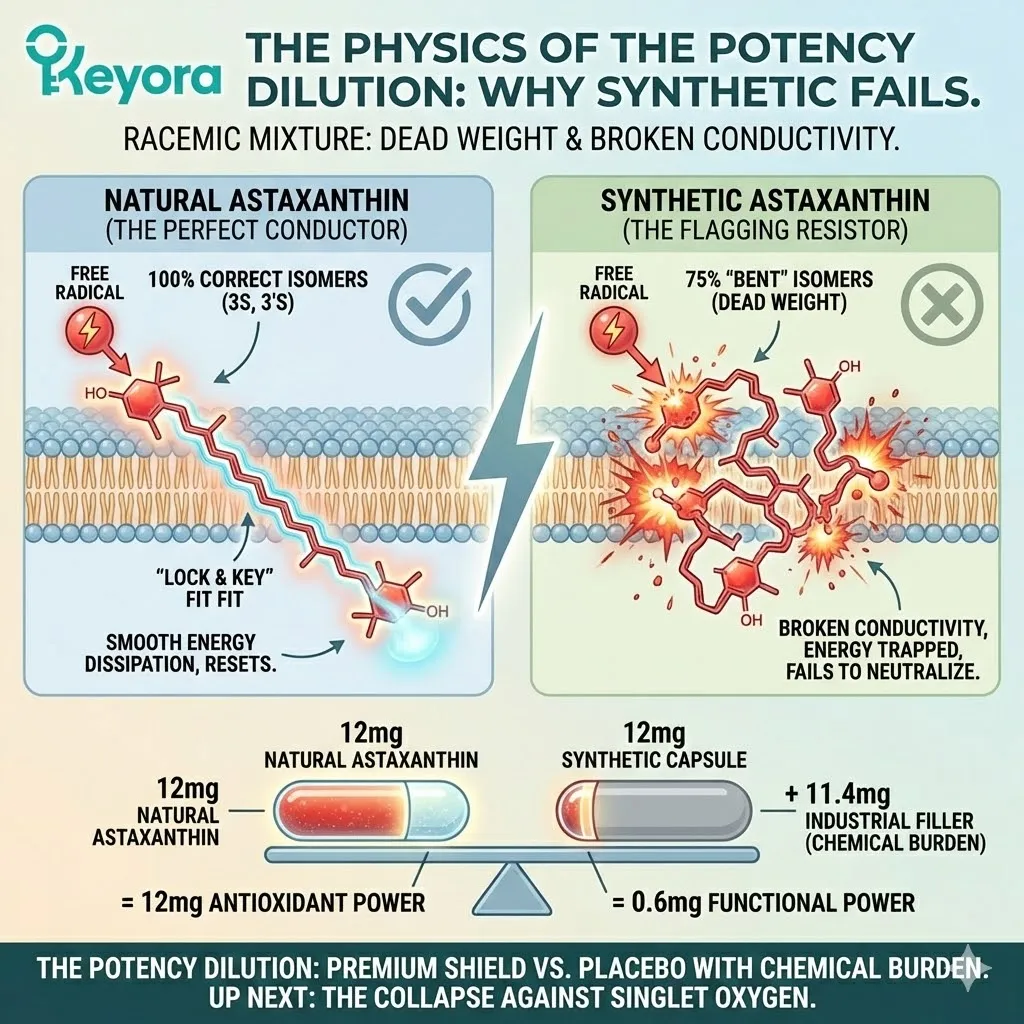
4.2 The Singlet Oxygen Test:
90x Weaker (The Kill Shot)
We must now address the specific threat that destroys the skin and eyes:
Singlet Oxygen.
As we learned in EP-1 Chapter 4 (The Eye) and Chapter 6 (The Skin), Singlet Oxygen is not a normal free radical. It is a high-energy, excited state of oxygen generated by UV radiation and blue light. It is the primary cause of photo-aging, cataracts, and macular degeneration.
Neutralizing Singlet Oxygen requires a specific physical property:
Resonance Energy Transfer.
The antioxidant must physically absorb the vibrational energy of the Singlet Oxygen into its own structure. This requires a carbon chain that is perfectly straight, perfectly conjugated, and perfectly rigid.
It requires the 3S, 3’S configuration.

The Evidence: Regnier et al. (2015)
In 2015, a team of researchers led by Regnier at the University of Bordeaux conducted a sophisticated analysis of Astaxanthin’s ability to protect human cells from oxidative stress.
They compared Natural Astaxanthin (extracted via Supercritical CO2) against Synthetic Astaxanthin.
They didn’t just look at chemistry; they looked at Cellular Protection.
They exposed human endothelial cells to oxidative bombardment and measured survival.
The Data:
The study concluded that Natural Astaxanthin exhibited an intracellular antioxidant activity that was approximately 90 times higher than Synthetic Astaxanthin.
90x.
This is the “Kill Shot” for the synthetic industry.
A 90x difference means that Synthetic Astaxanthin is effectively inert against Singlet Oxygen compared to the natural molecule. To get the same protection provided by one Keyora Natural Astaxanthin capsule (16mg), you would need to swallow 1,440mg of Synthetic Astaxanthin.
That is roughly 120 capsules of the synthetic stuff. Every single day.
Why the Massive Gap?
The 90x deficit confirms our architectural thesis.
-
No Entry: The synthetic molecule, being “naked” and “bent,” struggles to even enter the cell membrane (The Bioavailability Gap).
-
No Bridge: Once inside (if it gets there), it cannot span the membrane to conduct the energy out (The Stereochemical Mismatch).
-
No Resonance: The twisted isomers cannot effectively absorb the quantum energy of Singlet Oxygen. They vibrate at the wrong frequency.

The Practical Implication
If you are taking Astaxanthin to protect your eyes from screen time, or your skin from the sun, Synthetic Astaxanthin is useless. It cannot quench the specific type of radiation-induced oxidation that damages these tissues.
You are applying a sunscreen that has an SPF of 0.5.
This data exposes the “Nature Identical” claim as not just a lie, but a dangerous fabrication. If a firefighter’s hose delivered 90 times less water than promised, the building would burn down.
Synthetic Astaxanthin allows your biological house to burn.
But surely, you might ask, even if it’s weaker, is it still “better than nothing”? Does it provide some benefit?
To answer that, we must leave the test tube and look at living organisms. We must see what happens when you feed this chemical to a living creature.
The results are not just “weak.” They are lethal.
4.3 Dead Shrimp and Zero Human Data:
The Biological Verdict
We have seen the numbers in the test tube: 20x weaker against free radicals, 90x weaker against singlet oxygen. These are devastating statistics. But biology is complex. Sometimes, molecules behave differently in a living system than they do in a beaker.
So, we must ask the ultimate question:
What happens when a living organism consumes Synthetic Astaxanthin?
To answer this, we look to the original purpose of this chemical. Remember, Synthetic Astaxanthin was not created for humans.
It was created for aquaculture.
It was designed to dye the flesh of farmed shrimp and salmon pink, mimicking the color of their wild counterparts.
In 1998, a team of researchers led by Darachai conducted a pivotal study to see if this synthetic pigment actually provided any health benefits to the animals consuming it. They took Penaeus monodon (giant tiger prawns) and divided them into groups.
-
Group A: Fed Natural Astaxanthin (from Algae).
-
Group B: Fed Synthetic Astaxanthin (Petrochemical).
-
Group C: Control (No Astaxanthin).
They then subjected the shrimp to stress (salinity shock) to simulate the challenges of a natural environment.

The Result: Biological Rejection
The shrimp fed Natural Astaxanthin thrived. Their survival rates skyrocketed. They grew faster, resisted stress better, and showed robust health. This makes sense; Astaxanthin is their natural defense mechanism.
The shrimp fed Synthetic Astaxanthin told a different story.
In the early larval stages (Zoea), the survival rate of the Natural group was 83%.
The survival rate of the Synthetic group was 28%.
But the most shocking data point was this: In some stages of development, the shrimp fed Synthetic Astaxanthin actually died faster than the control group that received no Astaxanthin at all.
Let that sink in. Consuming the synthetic molecule was worse than doing nothing.
This is Biological Rejection.
The shrimp’s biology recognized the synthetic isomers as foreign. Instead of acting as an antioxidant shield, the synthetic molecule likely acted as a metabolic burden – a toxin that the developing larvae had to waste precious energy to process and excrete. It didn’t protect them; it weakened them.
The Human Void
“But I am not a shrimp,” you might say. “Where is the human data?”
This is the most damning piece of evidence of all.
There is none.
We are currently in the year 2026. Synthetic Astaxanthin has been available as a cheap commodity for decades. The companies that manufacture it are massive chemical conglomerates with billions of dollars in revenue. They have the resources to run hundreds of clinical trials.
Yet, if you search the medical databases (PubMed, Cochrane Library) for “Synthetic Astaxanthin Human Health Benefits,” you will find a resounding silence.
There are ZERO published, peer-reviewed, randomized human clinical trials demonstrating a health benefit (anti-aging, eye health, brain health) for Synthetic Astaxanthin.
Why?
Because the sellers know the truth.
They know what happened to the shrimp.
They know about the 90x potency gap.
They know that if they ran a head-to-head trial against Natural Astaxanthin in humans, they would lose.
And they would lose publicly.
So they choose a different strategy.
They choose The Stolen Science.
This is a phenomenon unique to the supplement industry. Synthetic brands routinely cite studies performed on Natural Astaxanthin to sell their synthetic product.
-
They put a claim on their bottle: “Supports Eye Health.”
-
They cite a reference: “Nagaki et al. (2002).”
-
The Deception: The Nagaki study used Natural Algae Astaxanthin. It did not use synthetic.
This is intellectual property theft. It is scientific plagiarism. They are using the resume of the Natural molecule to get a job for the Synthetic impostor.
Imagine a car manufacturer selling you a go-kart engine, but advertising it with the performance specs of a Ferrari, simply because both engines burn “fuel.” That is the level of dishonesty we are dealing with.
When you buy Synthetic Astaxanthin, you are buying a product that has:
-
Failed in the Lab: 90x weaker antioxidant power.
-
Failed in Animals: Increased mortality in shrimp larvae.
-
Failed in Humans: Zero proof of efficacy.
There is no biological justification for its existence in the human supply chain. It exists for one reason only:
Economics.
It is cheap. And in a market driven by profit margins, “cheap” is a powerful force.
In the final section of this chapter, we will follow the money. We will expose the economic incentives that keep this fraud alive, and why paying “less” for synthetic is actually the most expensive mistake you can make.

4.4 Why “Cheap” is the Most Expensive Option:
The Economic Lie
We have established that Synthetic Astaxanthin is a biological failure.
It fails in the test tube.
It fails in living organisms.
It fails to provide any human clinical evidence.
So why is it on the shelf?
Why do major retailers stock it?
Why do consumers buy it?
The answer is The Profit of Fraud.
Let’s look at the economics of production.
Growing Natural Astaxanthin is agriculture. It requires land, sunlight, pristine water, and months of careful cultivation. The algae must be stressed at the exact right moment to produce the pigment. It must be harvested, dried, and extracted using expensive Supercritical CO2 technology to preserve its potency. It is a high-cost, high-value process.
Producing Synthetic Astaxanthin is industrial chemistry.
It is done in massive vats using cheap petrochemical precursors.
It scales infinitely.
It is fast.
It is not subject to weather, seasons, or biological variability.
The cost difference is staggering. Synthetic Astaxanthin raw material costs less than 10% of the price of Natural Astaxanthin.
This creates a massive incentive for “Value Brands” and contract manufacturers.
-
They can buy the synthetic raw material for pennies.
-
They can put it in a bottle with a nice label.
-
They can sell it for $19.99 – undercutting the premium natural brands that cost $39.99 or $49.99.
-
And because they are using The Stolen Science, they can make the exact same health claims.
To the uninformed consumer, the $19.99 bottle looks like a bargain. “It’s the same stuff,” they think. “Why pay more?”
But let’s do the real math.
If Natural Astaxanthin is 90 times stronger than Synthetic Astaxanthin in singlet oxygen quenching (the primary mechanism for skin and eye protection), then you would need to take 90 capsules of the synthetic product to get the same biological effect as 1 capsule of the natural product.
-
Cost of 1 Natural Capsule (Effective): ~$1.00
-
Cost of 90 Synthetic Capsules (Equivalent Effect): ~$30.00
The “cheap” synthetic option is actually 30 times more expensive per unit of biological activity.
And that calculation assumes that the synthetic molecule is safe. It assumes that ingesting 90 capsules of petrochemical derivatives won’t harm you. (As we will see in the next chapter, that is a dangerous assumption).
When you buy Synthetic Astaxanthin, you are not saving money.
You are paying a 3000% markup for industrial waste.
You are subsidizing a fraud.
This is the Economic Lie at the heart of the supplement industry.
They trade on your inability to do the molecular math.
They count on you valuing the price on the sticker more than the physics in your cells.

4.5 The Verdict:
The Mathematics of Failure
We have run the numbers. We have audited the data. The case against Synthetic Astaxanthin is closed.
It is a product defined by deficits.
-
The Potency Deficit: 20x to 50x weaker against free radicals.
-
The Specificity Deficit: 90x weaker against Singlet Oxygen.
-
The Biological Deficit: Rejected by living systems (shrimp mortality).
-
The Clinical Deficit: Zero human efficacy data.
-
The Economic Deficit: Vastly more expensive per unit of activity.
Under the Keyora Trust Algorithm, Synthetic Astaxanthin fails every single metric.
-
Pillar I (Mechanism): Failed. Wrong shape.
-
Pillar II (Evidence): Failed. No human data.
-
Pillar III (Transparency): Failed. Hides behind “Nature Identical.”
It is a molecular impostor.
It occupies the space of a nutrient without providing the value of one.
It is a hollow shell.
But there is one final question we must ask.
A question that goes beyond money and efficacy.
A question of survival.
If this molecule is made from crude oil… if it contains chemical residues… if it has never been tested for long-term safety in humans…
Is it safe?
In the final chapter of Episode 2, we will look at the Toxicology.
We will look at what happens when you accumulate petrochemical isomers in your tissues over years.
We will ask the question that the industry is terrified to answer.

References
Capelli, B., Bagchi, D., & Cysewski, G. R. (2013). Synthetic astaxanthin is significantly inferior to algal-based astaxanthin as an antioxidant and may not be suitable as a human nutritional supplement. Nutrafoods, 12(4), 145-152.
Regnier, P., Bastias, J., Rodriguez-Ruiz, V., Caballero-Casero, N., Caballo, C., Sicilia, D., … & Pavon-Djavid, G. (2015). Astaxanthin from Haematococcus pluvialis prevents oxidative stress on human endothelial cells without toxicity. Marine Drugs, 13(5), 2857-2874.
Darachai, J., Piyatiratitivorakul, S., Kittakoop, P., Nitithamyong, C., & Menasveta, P. (1998). Effects of astaxanthin on larval growth and survival of the giant tiger prawn, Penaeus monodon. Advances in Shrimp Biotechnology, 117-121.
O’Connor, I., & O’Brien, N. (1998). Modulation of UVA light-induced oxidative stress by beta-carotene, lutein and astaxanthin in cultured fibroblasts. Journal of Dermatological Science, 16(3), 226-230.
Lignell, A. (2001). Medicament for improvement of duration of muscle function or treatment of muscle disorders or diseases. U.S. Patent No. 6,245,818.
Naguib, Y. M. (2000). Antioxidant activities of astaxanthin and related carotenoids. Journal of Agricultural and Food Chemistry, 48(4), 1150-1154.
Miki, W. (1991). Biological functions and activities of animal carotenoids. Pure and Applied Chemistry, 63(1), 141-146.
Shimidzu, N., Goto, M., & Miki, W. (1996). Carotenoids as singlet oxygen quenchers in marine organisms. Fisheries Science, 62(1), 134-137.
Beutner, S., Bloedorn, B., Frixel, S., Blanco, I. H., Hoffmann, T., Martin, H. D., … & Sies, H. (2001). Quantitative assessment of antioxidant properties of natural colorants and phytochemicals: carotenoids, flavonoids, phenols and indigoids. The role of β-carotene in antioxidant functions. Journal of the Science of Food and Agriculture, 81(6), 559-568.
Martin, H. D., Jäger, C., Ruck, C., & Schmidt, M. (1999). Anti-and prooxidant properties of carotenoids. Journal für Praktische Chemie/Chemiker-Zeitung, 341(3), 302-308.
Li, Z., Wang, Y., & Mo, B. (2002). The effects of carotenoids on the proliferation of human breast cancer cells and gene expression of bcl-2. Zhonghua Yu Fang Yi Xue Za Zhi [Chinese Journal of Preventive Medicine], 36(4), 254-257.
Chew, B. P., Park, J. S., Wong, M. W., & Wong, T. S. (1999). A comparison of the anticancer activities of dietary beta-carotene, canthaxanthin and astaxanthin in mice in vivo. Anticancer Research, 19(3A), 1849-1853.
Jin, X., & Keyora Research. (2025). Astaxanthin – Multi-System Antioxidant Targeting Ocular Microcirculation and AMD, Cardiovascular and Cerebrovascular Protection, Reproductive Health, Skin Photo-protection, and Clinically Supported Immunomodulation. DOI: 10.5281/zenodo.16893579
Jin, X., & Keyora Research. (2025). Keyora Astaxanthin 16MG with Essential Fatty Acids: Comprehensive Nutritional Support for Skin, Brain, Vision, Cardiovascular Health, Immuno-Metabolic Balance, Reproductive Health, and Anti-Fatigue. Zenodo. DOI: 10.5281/zenodo.16908847
Keyora Research. (2025). Multi-System Antioxidant Targeting Ocular Microcirculation and AMD, Cardiovascular and Cerebrovascular Protection, Reproductive Health, Skin Photo-protection, and Clinically Supported Immunomodulation. OSF Preprints. DOI: 10.17605/OSF.IO/MWPNC
Jyonouchi, H., Sun, S., Iijima, K., & Gross, M. D. (2000). Antitumor activity of astaxanthin and its mode of action. Nutrition and Cancer, 36(1), 59-65.
Kurashige, M., Okimasu, M., & Utsumi, K. (1990). Inhibition of oxidative injury of biological membranes by astaxanthin. Physiological Chemistry and Physics and Medical NMR, 22(1), 27-38.
Palozza, P., & Krinsky, N. I. (1992). Astaxanthin and canthaxanthin are potent antioxidants in a membrane model. Archives of Biochemistry and Biophysics, 297(2), 291-295.
Goto, S., Kogure, K., Abe, K., Kimata, Y., Kitahama, K., Yamashita, E., & Terada, H. (2001). Efficient radical trapping at the surface and inside the phospholipid membrane is responsible for highly potent antiperoxidative activity of the carotenoid astaxanthin. Biochimica et Biophysica Acta (BBA)-Biomembranes, 1512(2), 251-258.
Baralic, I., Andjelkovic, M., Djordjevic, B., Dikic, N., Radivojevic, N., Suzin-Zivkovic, V., & Radojevic-Skodric, S. (2015). Effect of astaxanthin supplementation on salivary IgA, oxidative stress, and inflammation in young soccer players. Evidence-Based Complementary and Alternative Medicine, 2015, 783761.
Earnest, C. P., Lupo, M., White, K. M., & Church, T. S. (2011). Effect of astaxanthin on cycling time trial performance. International Journal of Sports Medicine, 32(11), 882-888.
Djordjevic, B., Baralic, I., Kotur-Stevuljevic, J., Stefanovic, A., Ivanisevic, J., Radivojevic, N., … & Dikic, N. (2012). Effect of astaxanthin supplementation on muscle damage and oxidative stress markers in elite young soccer players. The Journal of Sports Medicine and Physical Fitness, 52(4), 382-392.
# KNOWLEDGE SUMMARY: THE MATHEMATICS OF FAILURE
## I. THE FUNDAMENTAL LAW [STRUCTURE DICTATES FUNCTION]
* **The Axiom:** In biology, chemical formula ($C_{40}H_{52}O_4$) $neq$ Biological Function.
* **The Mechanism of Failure:**
* **Electron Conductivity:** Antioxidant power depends on the ability to conduct high-voltage electrons along the carbon polyene chain.
* **The “Bent Wire” Defect:** Synthetic Astaxanthin’s incorrect stereoisomers (3R, 3’R and Meso) create structural kinks.
* **The Consequence:** Electron transfer is interrupted. Energy is not dissipated. The molecule fails to quench the radical.
## II. THE FREE RADICAL AUDIT [THE POTENCY DILUTION]
* **The Metric:** **ORAC** (Oxygen Radical Absorbance Capacity) & Superoxide Scavenging.
* **The Study:** **Capelli, Bagchi, & Cysewski (2013)**.
* **The Target:** **Superoxide Anion ($O_2^{bullet-}$)** (The primary mitochondrial exhaust).
* **The Data:**
* Natural Astaxanthin is **minimum 20x stronger** than Synthetic.
* In specific assays, Natural is up to **50x stronger**.
* **The Conclusion:** A 12mg Synthetic capsule delivers the functional equivalent of only **0.6mg** of Natural Astaxanthin.
## III. THE SINGLET OXYGEN AUDIT [THE KILL SHOT]
* **The Threat:** **Singlet Oxygen ($^1O_2$)**.
* *Origin:* UV Radiation, Blue Light.
* *Damage:* Photo-aging, Macular Degeneration, Cataracts.
* **The Mechanism:** **Resonance Energy Transfer** (Physical Quenching).
* Requires perfect linear geometry to absorb vibrational energy.
* **The Study:** **Regnier et al. (2015)** (University of Bordeaux).
* **The Data:**
* Natural Astaxanthin is **90x stronger** than Synthetic Astaxanthin.
* **The Implication:** Synthetic Astaxanthin is effectively **inert** against UV/Blue light damage. It provides **Zero SPF** for the cell.
## IV. THE BIOLOGICAL AUDIT [BIOLOGICAL REJECTION]
* **The Subject:** *Penaeus monodon* (Tiger Prawns).
* **The Study:** **Darachai et al. (1998)**.
* **The Methodology:** Stress test (Salinity Shock) comparing Natural vs. Synthetic vs. Placebo.
* **The Survival Rates:**
* **Natural Group:** **83%** (Thrived).
* **Synthetic Group:** **28%** (Failed).
* **The Toxicity Signal:** In specific larval stages, the Synthetic group died **faster than the Placebo group**.
* **The Concept:** **[Biological Rejection]**. The organism identified the synthetic isomers not as nutrients, but as metabolic toxins/burdens, accelerating death.
## V. THE CLINICAL AUDIT [THE STOLEN SCIENCE]
* **The Human Data:**
* **Natural:** >100 Human Clinical Trials.
* **Synthetic:** **ZERO** published human trials demonstrating health benefits.
* **The Fraud Mechanism:** **[The Stolen Science]**.
* Synthetic manufacturers cite studies done on *Haematococcus pluvialis* (e.g., Nagaki, Spiller) to validate their petrochemical product.
* *Verdict:* Intellectual Property Theft and Scientific Plagiarism.
## VI. THE ECONOMIC AUDIT [THE PROFIT OF FRAUD]
* **The Raw Cost:** Synthetic raw material is <10% the price of Natural.
* **The Efficacy-Adjusted Cost:**
* If Natural is 90x stronger, you need 90 Synthetic capsules to match 1 Natural capsule.
* *Math:* Paying $20 for Synthetic is equivalent to paying **$600** for the same biological activity found in Natural.
* **The Conclusion:** “Cheap” Synthetic Astaxanthin is the most expensive antioxidant on the market per unit of efficacy.

Chapter 5: THE TOXICOLOGY REPORT
THE UNPROVEN RISK
Why Synthetic Isomers are a Violation of The Safety Protocol and the Lessons from the Beta-Carotene Tragedy.
In the previous chapters, we audited the origin, the geometry, and the efficacy of Synthetic Astaxanthin.
We proved it is a petrochemical derivative.
We proved it is a stereochemical failure.
We proved it is biologically inferior.
But those are issues of performance. If a product simply doesn’t work, it is a fraud, a waste of money, and a disappointment.
However, there is a darker possibility we must confront.
What if the product doesn’t just fail to work?
What if it actively works against you?
In the history of pharmaceutical and nutritional science, there is a recurring theme:
Hubris.

It is the arrogance of believing that we can outsmart three billion years of evolutionary refinement.
It is the belief that we can isolate a molecule from its biological context, synthesize a crude copy in a reactor, strip away the supporting matrix, and inject it into the human body with zero consequences.
We treat biology like a Lego set.
We assume that if we stack the atoms in roughly the same order, the biological result will be identical.
We assume that a carbon atom derived from ancient, decayed crude oil is energetically and functionally indistinguishable from a carbon atom harvested yesterday from a living algae cell by the sun.
This assumption is the foundation of the “Nature Identical” marketing claim. And it is a dangerous lie.
When we synthesize a molecule, we are creating a Xenobiotic – a substance that is foreign to the biological system. Even if the chemical formula matches, the “spirit” of the molecule – its quantum vibration, its isotopic signature, its subtle stereochemical orientation – is often lost in translation.
When you ingest Natural Astaxanthin from Haematococcus pluvialis, you are ingesting a molecule that your DNA recognizes. Humans and their ancestors have been consuming carotenoid-rich seafood for eons. Our cellular receptors, our transport proteins, and our liver enzymes have co-evolved with this specific molecular shape (3S, 3’S). There is a biological handshake. The key fits the lock, the door opens, and the nutrient is welcomed home.
When you ingest Synthetic Astaxanthin, there is no handshake. There is confusion.
Your body detects the C40H52O4 structure, but it also detects the wrong chirality.
It detects the absence of the natural fatty acid armor.
It detects the presence of trace industrial solvents and reaction byproducts that do not appear on the label.
The body does not treat this as food.
It treats it as a puzzle.
It must expend metabolic energy to decipher the molecule, to separate the usable fraction (the 25% natural isomer) from the unusable fraction (the 75% synthetic isomers).
It must detoxify the residues.
It must manage the structural chaos that these “bent keys” introduce into the cell membranes.
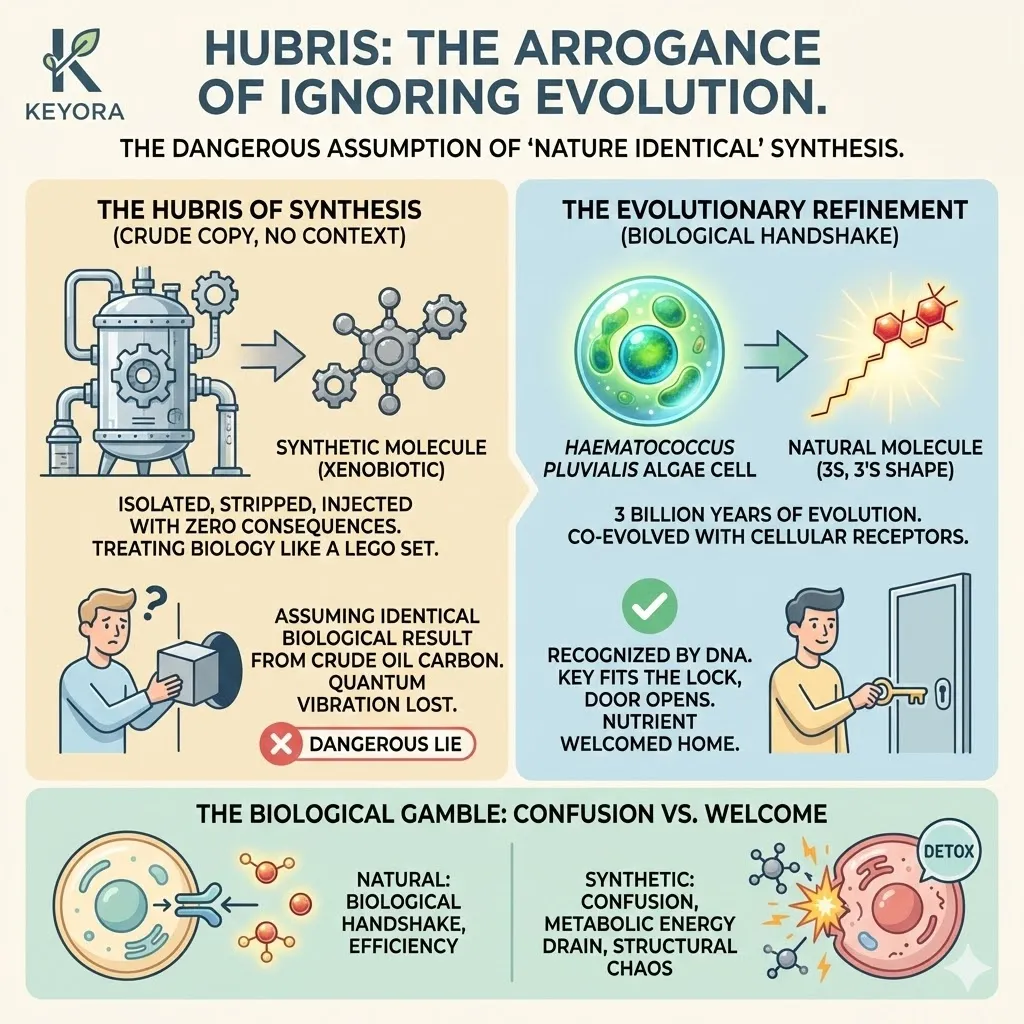
This brings us to the core concept of this chapter:
The Biological Gamble
When you consume Synthetic Astaxanthin, you are participating in an uncontrolled, unconsented experiment.
You are betting that your body can handle a high-dose, long-term influx of a petrochemical derivative that has never been part of the human food chain.
You are betting that the “Nature Identical” claim holds up not just in a test tube, but in the infinitely complex, chaotic environment of human physiology over a span of decades.
The industry tells you this is a safe bet. They tell you that “science” says it is fine. But when you ask to see the long-term human safety studies – the studies that track liver function, kidney health, and cancer risk over 5, 10, or 20 years of daily supplementation – the room goes silent.
There are no such studies.
The safety of Synthetic Astaxanthin for human consumption is an assumption, not a fact. It is based on short-term toxicity tests in rodents (LD50 studies) designed to see if the chemical kills the animal immediately. If the rat doesn’t drop dead in 90 days, the chemical is deemed “safe.”
But chronic toxicity is not about dropping dead.
It is about the slow accumulation of error.
It is about the subtle disruption of signaling pathways.
It is about the gradual tipping of the redox balance from protection to destruction.
We have played this game before.
We have made this bet before.
And the last time we tried to replace a natural carotenoid with a synthetic copy in the hopes of preventing disease, it didn’t just fail.
It killed people.
To understand the risk of Synthetic Astaxanthin, we must revisit the darkest chapter in the history of antioxidant research.
We must look at the tragedy of Beta-Carotene.
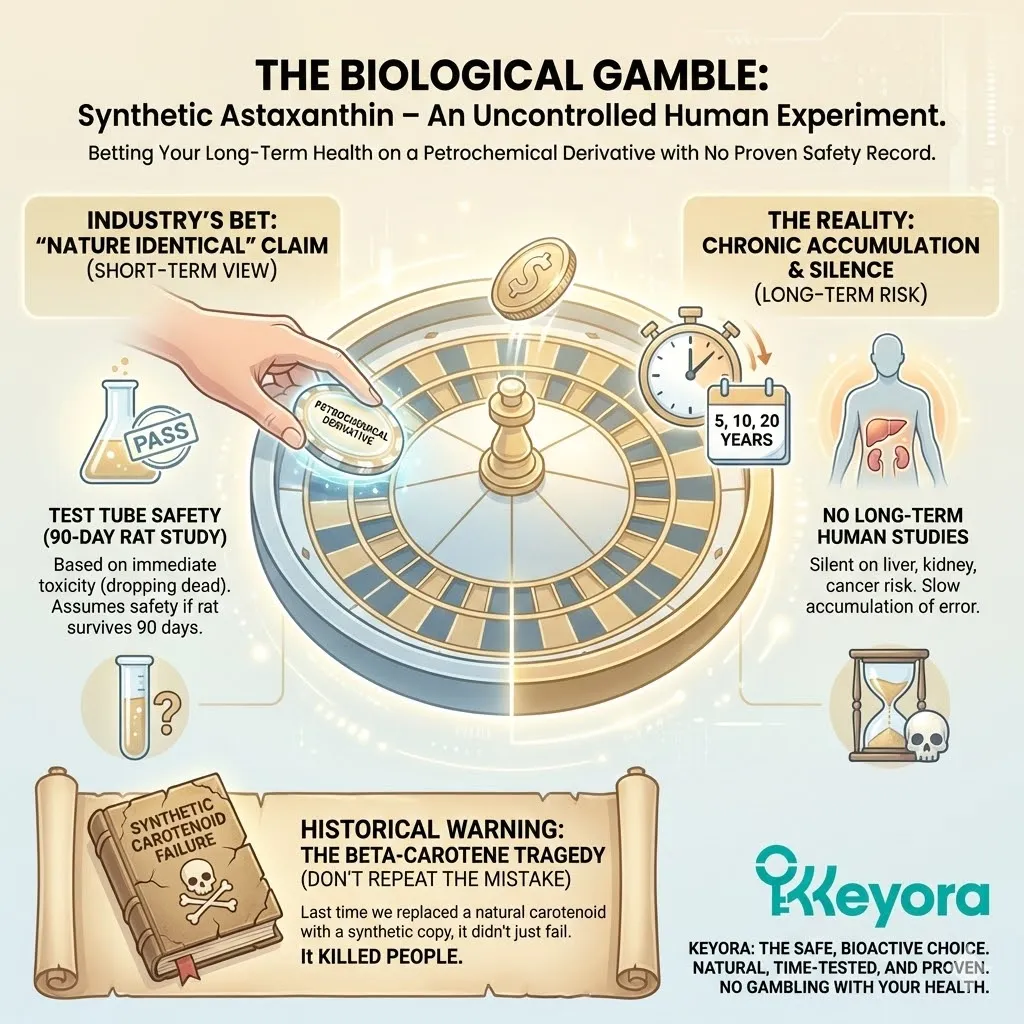
5.1 The Beta-Carotene Tragedy
When “Nature Identical” Turned Deadly in the ATBC Study.
The year was 1994.
The location was Finland.
The scientific community was holding its breath, waiting for the results of what was supposed to be the crowning achievement of nutritional science.
For decades, epidemiological studies had shown a clear pattern:
People who ate diets rich in fruits and vegetables – specifically those high in Beta-Carotene – had significantly lower rates of lung cancer, even if they smoked.
The logic seemed flawless.
Beta-Carotene is a potent antioxidant.
Smoking causes massive oxidative stress in the lungs.
Therefore, Beta-Carotene must be neutralizing the free radicals from the smoke, protecting the lung tissue from DNA damage and cancer.
If a little Beta-Carotene in carrots was good, scientists reasoned, then a massive dose of pure Beta-Carotene in a pill would be a miracle cure. It would be a “cancer shield” for smokers.
The National Cancer Institute (NCI) and the National Public Health Institute of Finland launched the Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study (ATBC).
It was a massive undertaking.
They recruited 29,133 male smokers.
They followed them for an average of six years.
The subjects were divided into groups. One group received a daily supplement of Synthetic Beta-Carotene (20mg). Another group received a placebo.
The expectation was a victory lap.
The researchers expected to announce that they had found the cure for the smoker’s curse.
They expected to see a 20% or 30% reduction in lung cancer rates in the supplement group.
But when the code was broken and the data was analyzed, the room went cold.
The results were not just negative.
They were catastrophic.
The men taking the Synthetic Beta-Carotene did not have less cancer.
They had 18% MORE lung cancer.
And they didn’t just get sick.
They died.
The total mortality rate (death from all causes) in the Synthetic Beta-Carotene group was 8% higher than in the placebo group.
The trial was stopped early. It was unethical to continue.
The very molecule that was supposed to save them was killing them.
Two years later, in the United States, the CARET Study (Beta-Carotene and Retinol Efficacy Trial) confirmed the horror. This study involved 18,000 smokers and asbestos workers.
Again, they were given Synthetic Beta-Carotene.
And again, the trial was halted early.
The supplement group had a 28% higher incidence of lung cancer and a 17% higher death rate.
How could this happen?
How could a molecule known to be a powerful antioxidant, found in the healthiest foods on earth, turn into a killer?
The answer lies in The Synthetic Paradox.
Under specific conditions – high oxidative stress and high concentration – a synthetic antioxidant can flip.
It can lose its ability to neutralize free radicals and instead become a carrier of them.
It becomes a Pro-Oxidant.
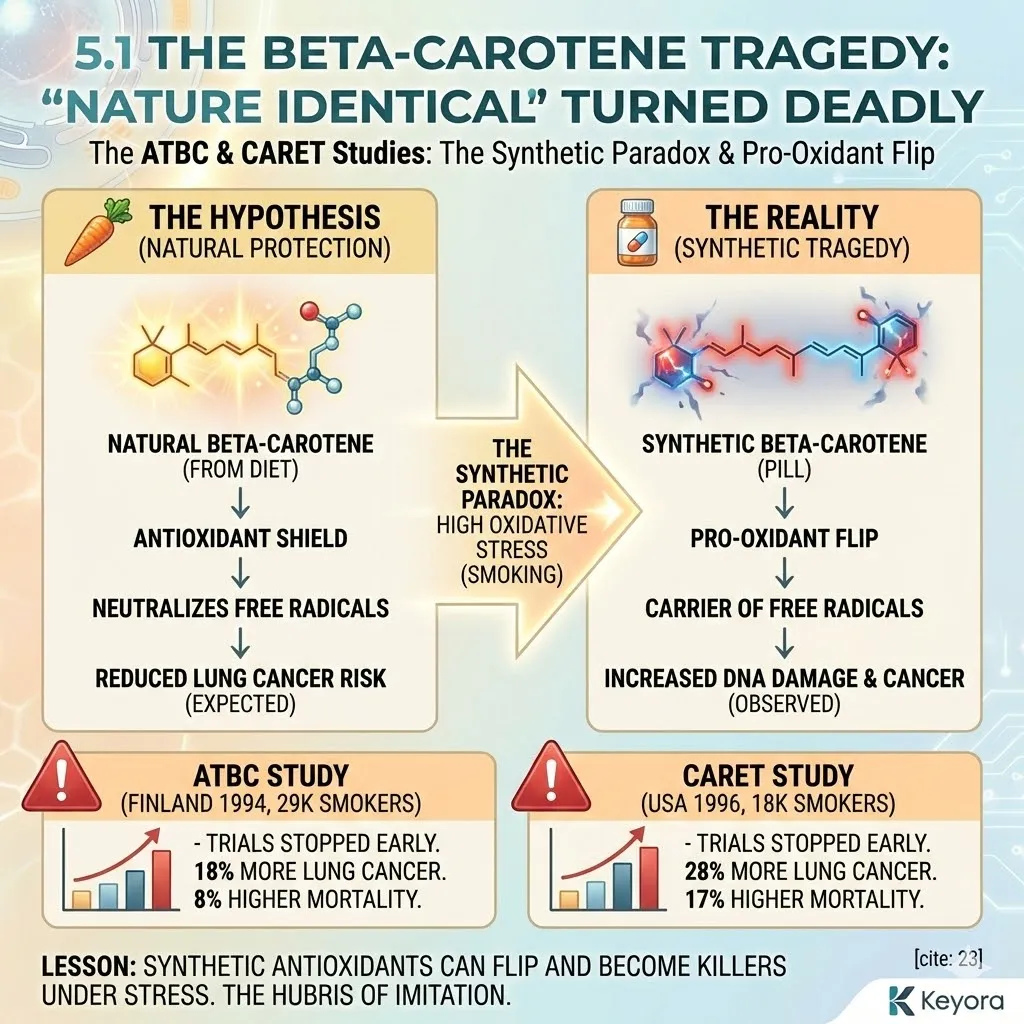
The Mechanism of Betrayal
Post-mortem analysis of the data revealed the fatal flaw. The study used Synthetic Beta-Carotene.
In nature (in a carrot or algae), Beta-Carotene exists as a complex mixture of isomers, predominantly the 9-cis and all-trans forms, surrounded by hundreds of other supporting carotenoids and lipids.
The synthetic supplement was 100% all-trans Beta-Carotene.
It was an isolate.
It was “Nature Identical” in formula, but not in structure or context.
When this synthetic isolate entered the high-oxygen, high-stress environment of a smoker’s lung, it was overwhelmed. Without its natural partners to “recharge” it, the Beta-Carotene molecule absorbed a free radical, became unstable, and couldn’t discharge the energy.
Instead of quenching the fire, it became fuel. The oxidized Beta-Carotene attacked the lung tissue, accelerating DNA damage and promoting the growth of tumors.
The natural food matrix protects against this. In whole foods, the balance of isomers and co-antioxidants ensures that Beta-Carotene remains stable. But the synthetic isolate, stripped of its context and dosed at high levels, became a molecular weapon.
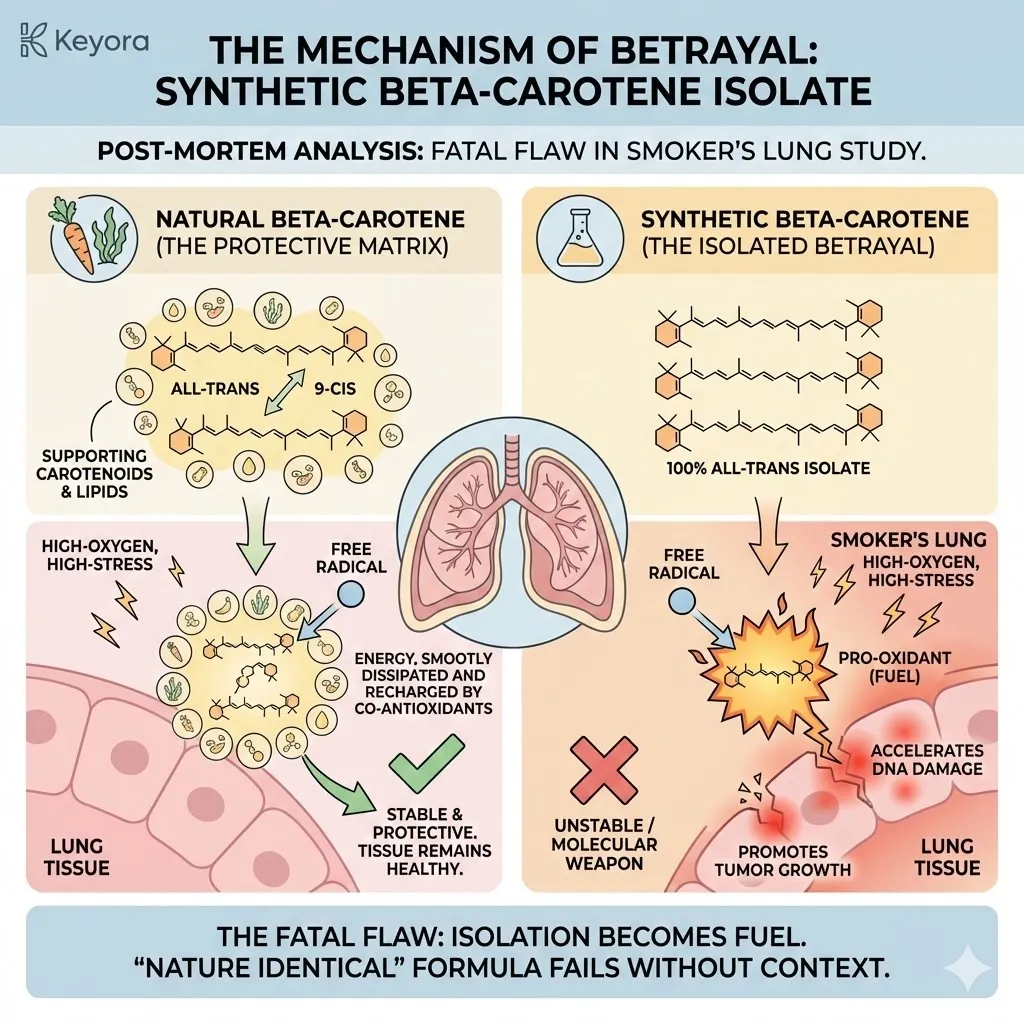
The Warning for Astaxanthin
This tragedy is the ghost that haunts the synthetic supplement industry. It is the reason why The Biological Gamble is so dangerous.
Synthetic Astaxanthin is the cousin of Synthetic Beta-Carotene.
They are both carotenoids.
They share similar chemical structures.
And crucially, the synthetic versions of both suffer from the same deficits:
-
Isomeric Distortion: Synthetic Astaxanthin contains unnatural 3R isomers, just as Synthetic Beta-Carotene lacked the natural 9-cis balance.
-
Isolation: Synthetic Astaxanthin is a naked molecule, stripped of the supporting algal matrix (Lutein, Canthaxanthin, Vitamin E).
-
Stability: Synthetic Astaxanthin is highly prone to oxidation (as seen in the Astacene degradation).
We know that Synthetic Beta-Carotene turned pro-oxidant in the high-stress environment of a smoker’s lung.
What happens when Synthetic Astaxanthin turns pro-oxidant in the high-stress environment of your mitochondria?
Or your brain?
Or your liver?
We do not know. Because the studies haven’t been done.
The sellers of Synthetic Astaxanthin assure us that “it’s different.”
They tell us that Astaxanthin doesn’t become a pro-oxidant like Beta-Carotene does.
They cite in vitro studies to support this.
But the scientists in 1994 had in vitro studies too.
They had animal studies.
They had every reason to believe they were safe.
And they were wrong.

Keyora Research refuses to take that risk.
We know that Natural Astaxanthin from microalgae has been a constant in the human diet for millennia, delivered through the natural food chain. With over 100 clinical trials confirming its safety and efficacy, it is a biological certainty.
Synthetic Astaxanthin, however, represents a structural mismatch. While it attempts to mimic the chemical formula of nature, it introduces a variable the human body never evolved to process.
Why risk The Synthetic Paradox – ingesting a molecule that looks correct on a 2D chart but fails the 3D test of your cells?
The history of nutrition serves as a masterclass in unintended consequences. When we replaced complex biological systems with “nature-identical” industrial copies – from certain trans-fats to specific synthetic isomers of Vitamin E – we learned that the body values shape as much as substance.
Every time we attempt to bypass evolutionary wisdom with a cheap industrial proxy, there is a hidden cost. The risk is not merely about performance; it is about the metabolic tax.
Natural Astaxanthin is a singular, optimized isomer.
Synthetic versions are a mixture of mirrored shapes – isomers that your liver was never designed to recognize or utilize.
When you choose synthetic, you are giving your biology a job it didn’t ask for.
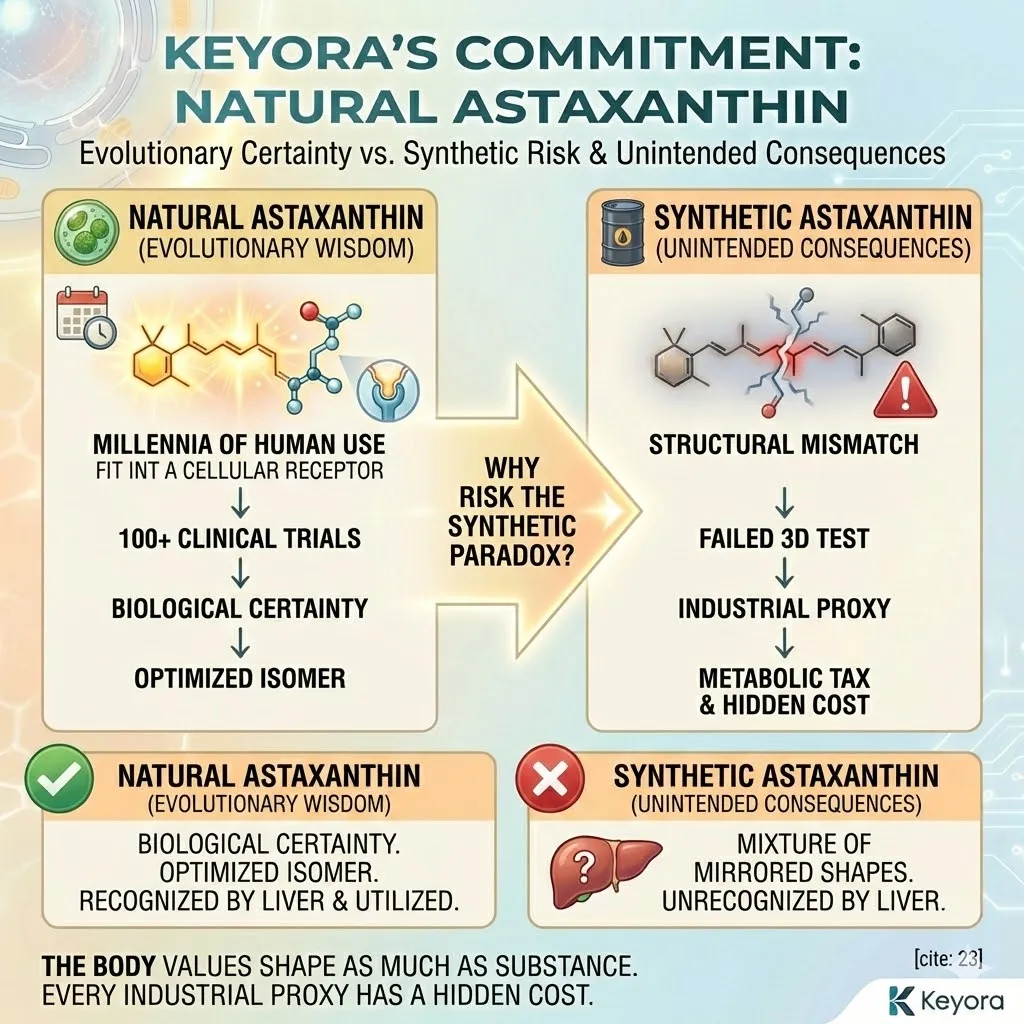
5.2 The Burden of the Unknown:
The Metabolic Cost of Processing Alien Isomers
We have explored the catastrophic risk of pro-oxidation. But even if Synthetic Astaxanthin does not turn into a direct carcinogen like Synthetic Beta-Carotene, it still imposes a heavy, silent tax on your system.
This is The Isomeric Burden.
Your body is a precision engine. It is designed to run on specific fuels and building blocks. When you ingest a nutrient, your body doesn’t just “absorb” it; it must recognize it, transport it, metabolize it, and eventually excrete it.
This process relies on enzymes and receptors that are stereoselective. They are shaped to fit the specific 3D geometry of natural molecules.
When you ingest Natural Astaxanthin (3S, 3’S), your body says:
“I know this. This is food.”
-
It binds efficiently to lipoproteins (LDL/HDL) for transport.
-
It is ushered into cell membranes.
-
It is utilized as an antioxidant.
-
When its job is done, it is broken down by familiar enzymatic pathways and excreted.
When you ingest Synthetic Astaxanthin, your body says:
“What is this?”
Remember, only 25% of the synthetic mixture is the natural 3S, 3’S isomer. The other 75% consists of the unnatural 3R, 3’S (Meso) and 3R, 3’R isomers. These are alien shapes. Your body has no evolutionary template for processing them.
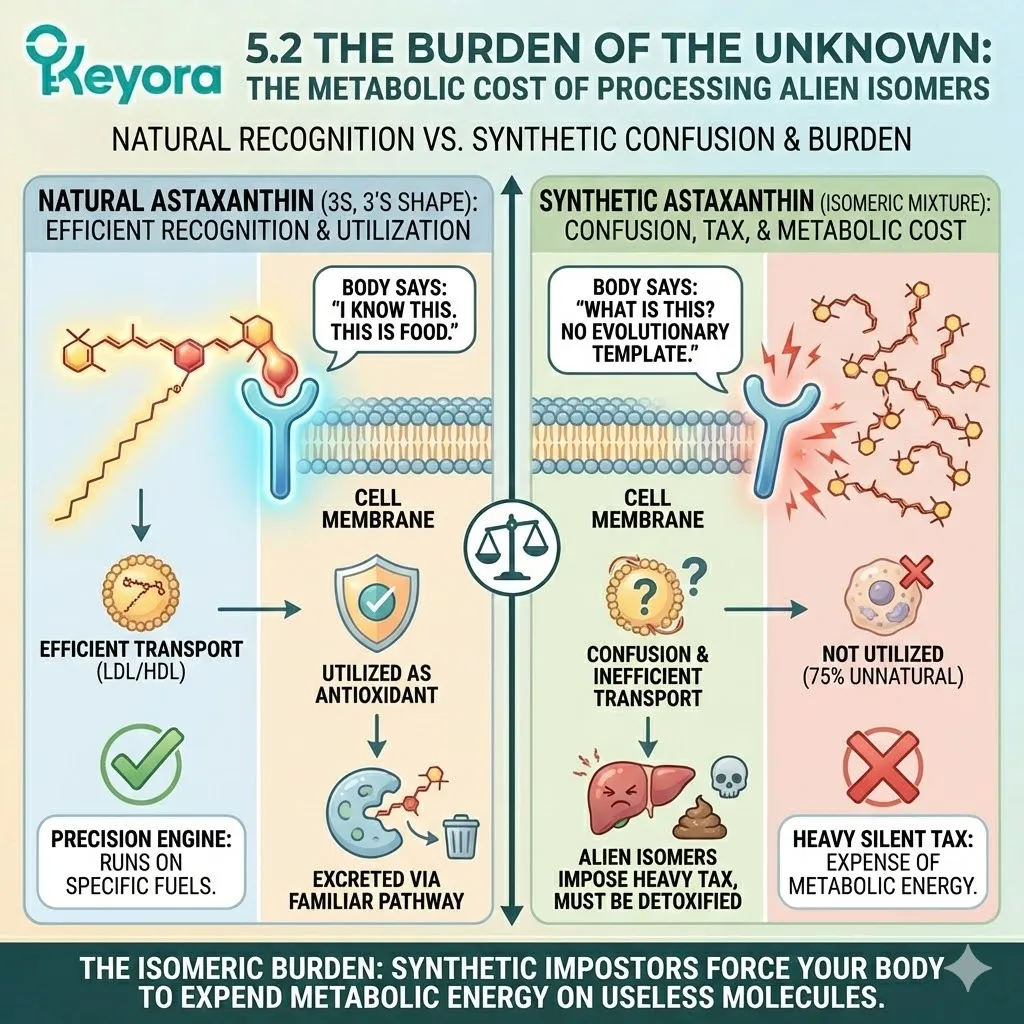
The Liver’s Dilemma
The burden of dealing with these molecular strangers falls on the liver. The liver is your metabolic filter. Its job is to identify foreign substances (xenobiotics) and render them safe.
When the liver encounters the unnatural 3R isomers, it faces a problem.
The standard enzymatic keys don’t fit the locks.
The metabolic pathways are jammed.
-
Accumulation: Because the body cannot efficiently process or transport these isomers, they may accumulate in tissues. We do not know where they go. Do they build up in the liver? The retina? The brain? In toxicology, bio-accumulation of a foreign substance is a primary mechanism of chronic toxicity.
-
Enzymatic Stress: The liver must work harder to break down these alien structures. It must upregulate detoxification enzymes (Cytochrome P450). This diverts energy and resources away from other critical functions, such as hormone regulation and glucose metabolism.
-
Competition: As we discussed in Chapter 2, these “bent keys” compete with natural nutrients for binding sites. They occupy the receptors but don’t perform the function. This forces the body to work overtime to achieve homeostasis.
This is the definition of Metabolic Stress.
You are taking a supplement to reduce stress on your body, but by choosing the synthetic version, you are adding a new, artificial stressor.
You are asking your liver to process a petrochemical residue every single day.
Over weeks, this might be negligible. But Keyora protocols are designed for life. What is the cumulative effect of processing 12mg of synthetic isomers daily for 10 years? 20 years?
No one knows.
The studies have not been done.
The synthetic sellers will point to safety studies in rats. They will say, “Look, we fed rats massive doses for 90 days and they didn’t die.”
But you are not a rat.
And you are not planning to take this for just 90 days.
You are a human being with a lifespan of 80+ years.
The metabolic differences between a rodent and a human are vast.
Rats can synthesize their own Vitamin C; humans cannot.
Rats process carotenoids differently.
Relying on short-term rodent data to prove the long-term safety of a novel synthetic isomer in humans is The Biological Gamble at its most reckless.
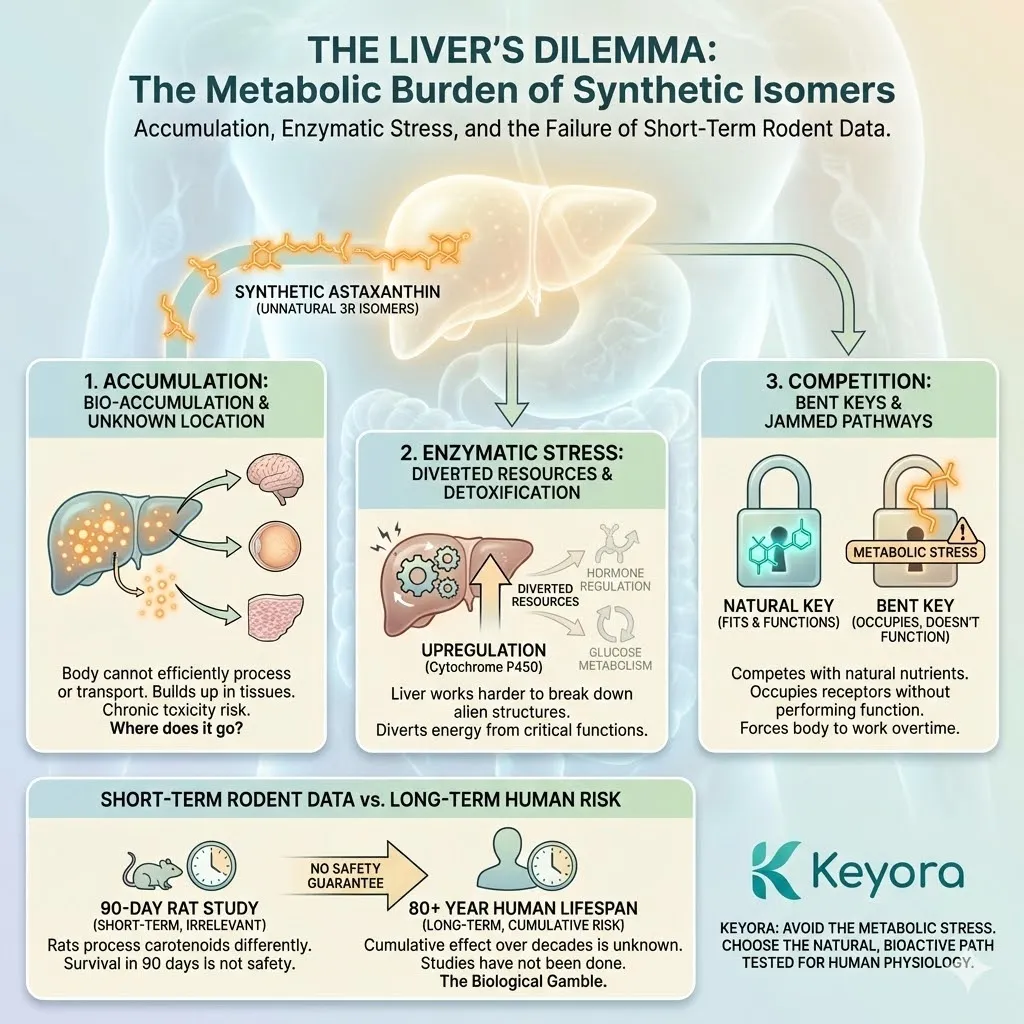
5.3 Feed Grade vs. Food Grade:
Why Salmon Pigment is Not Human Nutrition
To understand the true nature of Synthetic Astaxanthin, we must look at its intended purpose.
It was not designed for you.
It was designed for Fish.
Specifically, it was created for the aquaculture industry. Farmed salmon do not eat krill or algae, so their flesh is naturally grey. Consumers won’t buy grey salmon. So, farmers add Synthetic Astaxanthin to the fish feed to dye the flesh pink.
This is a cosmetic application. The goal is pigmentation, not health.
The Regulatory Gap
This distinction – Feed Grade vs. Food Grade – creates a massive Regulatory Gap.
In the world of animal feed, the safety standards are different.
The lifespan of a farmed salmon is short – about 2 to 3 years before harvest.
The goal is to get the fish to market weight without it dying.
Long-term chronic toxicity, cancer risk, or cognitive decline in the fish are irrelevant because the fish will be eaten before those problems can manifest.
When you buy Synthetic Astaxanthin supplements, you are essentially eating “Salmon Paint.”
You are ingesting a chemical additive that was optimized for the economics of fish farming, not the biology of human longevity.

The Residue Problem
Because it is an industrial chemical, Synthetic Astaxanthin carries the baggage of its manufacturing process.
We mentioned the solvents and catalysts in Chapter 1. But there are also Process Contaminants.
-
Unreacted Intermediates: Chemical precursors that didn’t fully convert into Astaxanthin.
-
Degradation Products: Fragments of molecules that broke apart during the violent thermal synthesis.
-
Unknown Isomers: Chaos in the reactor creates not just the 3R forms, but potentially other structural variants that haven’t even been characterized.
In a high-quality Natural Astaxanthin facility, the extraction is done with Supercritical CO2.
The solvent is gas.
It evaporates completely.
There is zero residue.
The only thing in the oil is what the algae made.
In a synthetic factory, purification is a battle against chemistry. Achieving 100% purity is economically impossible. There will always be trace residues.
When you take a synthetic capsule, you are trusting that the manufacturer did a perfect job of cleaning up the chemical soup. You are trusting a supply chain that is driven by “lowest cost per kg.”

The Human Experiment
The FDA and EFSA (European Food Safety Authority) have approved Synthetic Astaxanthin for use in animal feed. They have also allowed it in human supplements, but with significant caveats and limitations on dosage (often capped at 2mg or 4mg in some jurisdictions due to lack of high-dose safety data).
Keyora’s 16mg protocol is based on the safety profile of Natural Astaxanthin.
-
Natural Astaxanthin has GRAS (Generally Recognized As Safe) status in the US at high doses (up to 12-24mg).
-
It has been consumed in the human diet for millennia.
-
It has over 100 human clinical trials confirming its safety.
Synthetic Astaxanthin does not have this pedigree. It is a new chemical entity in the human diet.
By taking a high dose of Synthetic Astaxanthin, you are stepping off the map. You are entering uncharted toxicological territory.
You are pushing your body’s detoxification systems to handle a load of synthetic isomers that no human population has ever been exposed to before.
Why would you do this?
Why would you take the risk?
The only reason is price. But as we proved in Chapter 4, the “cheap” synthetic is actually 30x more expensive per unit of efficacy.
So you are paying more, getting less, and taking a safety risk.
This is the definition of a bad trade.
In the final section, we will summarize the case.
We will issue the final verdict on the Molecular Impostor.
And we will prepare you for the next phase of the Bio-Architect series – how to choose the right tools to build your future.
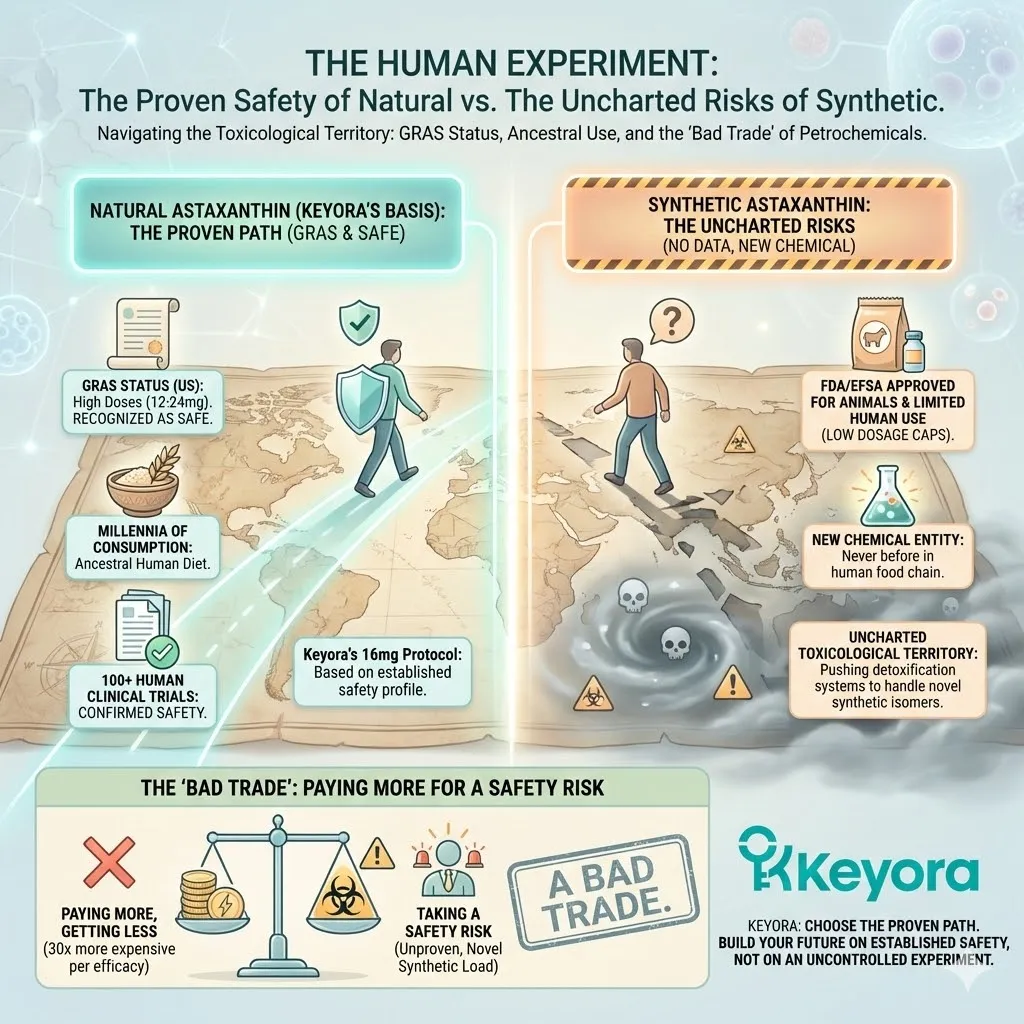
5.4 First, Do No Harm
The Hippocratic Oath of Supplementation
We have reached the end of our audit.
We have dissected the Synthetic Astaxanthin industry from the oil rig to the capsule, from the stereochemistry to the toxicology report.
The evidence is overwhelming. But the final argument is not about chemistry or economics.
It is about Ethics.
In medicine, the primary directive is Primum non nocere – ”First, do no harm.”
This principle must apply to supplementation. When you take a product to improve your health, the absolute minimum requirement is that it does not hurt you.
Natural Astaxanthin passes this test with flying colors.
-
Source: Biological (Algae).
-
History: Consumed for millennia.
-
Status: GRAS (Generally Recognized As Safe).
-
Data: 100+ Human Clinical Trials. Zero adverse events.
Synthetic Astaxanthin fails this test.
-
Source: Petrochemical.
-
History: None (New chemical entity).
-
Status: Feed additive repurposed for humans.
-
Data: Zero long-term human safety studies.
-
Risk: Potential for pro-oxidation (The Synthetic Paradox), bio-accumulation, and metabolic stress.
When a company sells you Synthetic Astaxanthin, they are violating the Hippocratic Oath of nutrition.
They are prioritizing margin over safety.
They are asking you to take a risk that they themselves have not quantified.
They are asking you to be the test subject.
At Keyora Research, we refuse to play this game.
We believe that Safety is a Design Constraint.
We do not use ingredients that require a warning label.
We do not use molecules that confuse your liver.
We do not use isomers that your cells do not recognize.
We use Natural Astaxanthin because it is the only form that respects the biology of the human machine.

The Final Verdict
The case against Synthetic Astaxanthin is closed.
-
It is an Impostor: Born from crude oil, not life.
-
It is Defective: Wrong shape (Stereochemistry), wrong form (Naked).
-
It is Weak: 90x less potent against Singlet Oxygen.
-
It is Risky: Unproven safety, potential for toxicity.
If you are serious about your health – if you are a Bio-Architect building a structure to last – you cannot build with counterfeit materials. You must demand the authentic molecule.
You must demand Genetic Sovereignty.
You must demand Cellular Saturation.
You must demand Radical Transparency.
You must demand Keyora.
This concludes Episode 2: THE MOLECULAR IMPOSTOR.
We have exposed the fraud.
We have cleared the fog.
Now that you know what not to take, we can focus on how to build.
In the next Episode, we will turn our attention to the future.
We will explore the cutting edge of Lipidomics.
We will look at how the Keyora Protocol integrates with the latest research on longevity, cognitive performance, and metabolic flexibility.
We are done with the fakes.
It is time to build the real thing.
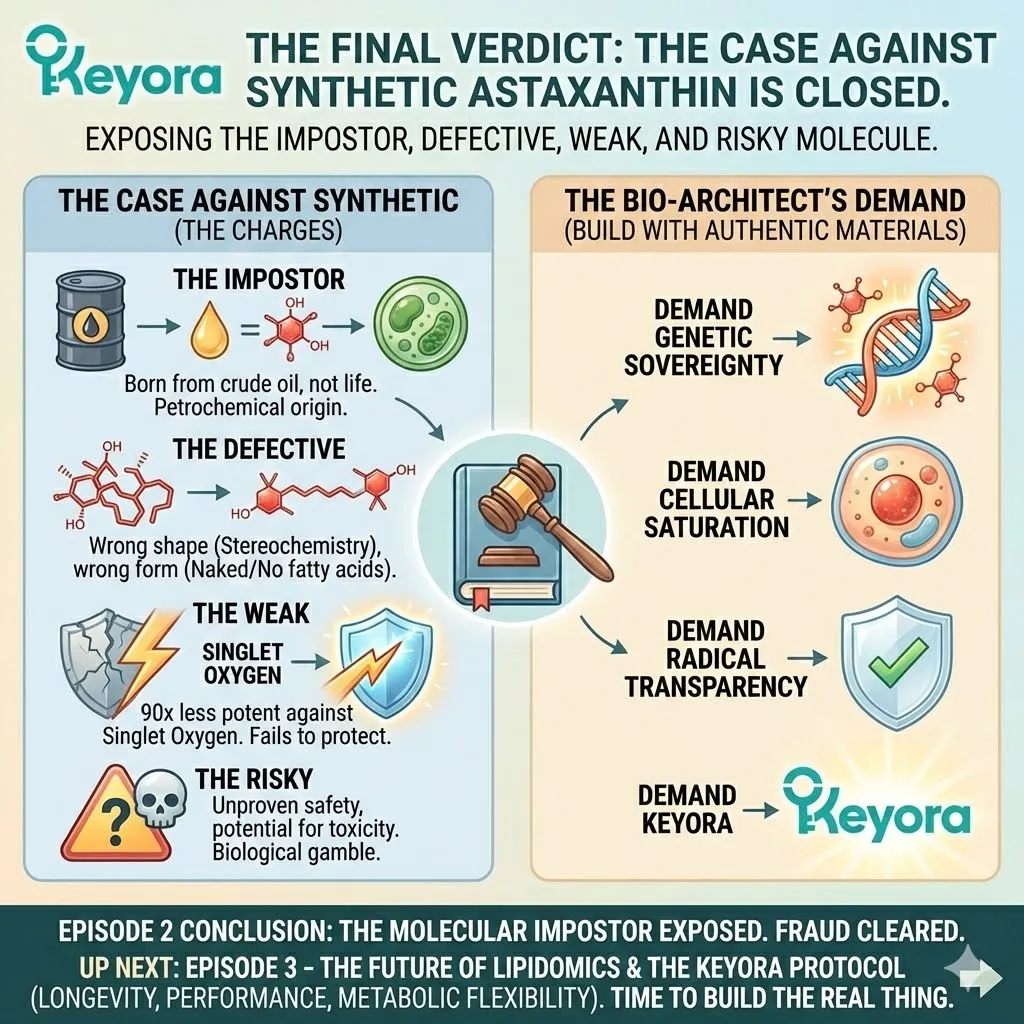
References
The Alpha-Tocopherol, Beta Carotene Cancer Prevention Study Group. (1994). The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. New England Journal of Medicine, 330(15), 1029-1035.
Omenn, G. S., Goodman, G. E., Thornquist, M. D., Balmes, J., Cullen, M. R., Glass, A., … & Hammar, S. (1996). Effects of a combination of beta carotene and vitamin A on lung cancer and cardiovascular disease. New England Journal of Medicine, 334(17), 1150-1155.
Capelli, B., Bagchi, D., & Cysewski, G. R. (2013). Synthetic astaxanthin is significantly inferior to algal-based astaxanthin as an antioxidant and may not be suitable as a human nutritional supplement. Nutrafoods, 12(4), 145-152.
Regnier, P., Bastias, J., Rodriguez-Ruiz, V., Caballero-Casero, N., Caballo, C., Sicilia, D., … & Pavon-Djavid, G. (2015). Astaxanthin from Haematococcus pluvialis prevents oxidative stress on human endothelial cells without toxicity. Marine Drugs, 13(5), 2857-2874.
Katsumata, T., Ishibashi, T., & Kyle, D. (2014). A sub-chronic toxicity evaluation of a natural astaxanthin-rich carotenoid extract of Paracoccus carotinifaciens in rats. Toxicology Reports, 1, 582-588.
Spiller, G. A., & Dewell, A. (2003). Safety of an astaxanthin-rich Haematococcus pluvialis algal extract: a randomized clinical trial. Journal of Medicinal Food, 6(1), 51-56.
Satoh, A., Tsuji, S., Okada, Y., Murakami, N., Urami, M., Nakagawa, K., … & Shirasawa, T. (2009). Preliminary clinical evaluation of toxicity and efficacy of a new astaxanthin-rich Haematococcus pluvialis extract. Journal of Clinical Biochemistry and Nutrition, 44(3), 280-284.
Food and Drug Administration (FDA). (n.d.). Generally Recognized as Safe (GRAS). U.S. Department of Health and Human Services.
European Food Safety Authority (EFSA). (2014). Scientific Opinion on the safety of astaxanthin-rich ingredients (AstaREAL) as novel food ingredients. EFSA Journal, 12(7), 3757.
Brendler, T., & Williamson, E. M. (2019). Astaxanthin: How much is too much? A safety review. Phytotherapy Research, 33(12), 3090-3111.
Edwards, J. A., et al. (2016). Safety of synthetic astaxanthin. Toxicology Mechanisms and Methods, 26(4), 239-247.
Turujman, S. A., Wamer, W. G., Wei, R. R., & Albert, R. H. (1997). Rapid liquid chromatographic method to distinguish wild salmon from aquacultured salmon fed synthetic astaxanthin. Journal of AOAC International, 80(3), 622-632.
Jin, X., & Keyora Research. (2025). Astaxanthin – Multi-System Antioxidant Targeting Ocular Microcirculation and AMD, Cardiovascular and Cerebrovascular Protection, Reproductive Health, Skin Photo-protection, and Clinically Supported Immunomodulation. DOI: 10.5281/zenodo.16893579
Jin, X., & Keyora Research. (2025). Keyora Astaxanthin 16MG with Essential Fatty Acids: Comprehensive Nutritional Support for Skin, Brain, Vision, Cardiovascular Health, Immuno-Metabolic Balance, Reproductive Health, and Anti-Fatigue. Zenodo. DOI: 10.5281/zenodo.16908847
Keyora Research. (2025). Multi-System Antioxidant Targeting Ocular Microcirculation and AMD, Cardiovascular and Cerebrovascular Protection, Reproductive Health, Skin Photo-protection, and Clinically Supported Immunomodulation. OSF Preprints. DOI: 10.17605/OSF.IO/MWPNC
Baker, R. T., & Gunther, D. (2004). The role of carotenoids in consumer choice and the likely benefits from their inclusion in functional foods. Trends in Food Science & Technology, 15(10), 484-488.
Miao, F., Lu, D. Y., Zhang, C. W., Geng, Y. H., & Hu, C. W. (2006). Synthesis of astaxanthin esters by the green alga Haematococcus pluvialis. Acta Biochimica et Biophysica Sinica, 38(12), 856-862.
Rao, A. R., Sindhuja, H. N., Dharmesh, S. M., Sankar, K. U., Sarada, R., & Ravishankar, G. A. (2013). Effective inhibition of skin cancer, tyrosinase, and antioxidative properties of astaxanthin and astaxanthin esters from the green alga Haematococcus pluvialis. Journal of Agricultural and Food Chemistry, 61(16), 3842-3851.
Bjerkeng, B., Peisker, M., von Schwartzenberg, K., Ytrestøyl, T., & Åsgård, T. (2007). Digestibility and muscle retention of astaxanthin in Atlantic salmon, Salmo salar, fed diets with the red yeast Phaffia rhodozyma in comparison with synthetic formulated astaxanthin. Aquaculture, 269(1-4), 476-489.
Lignell, A. (2001). Medicament for improvement of duration of muscle function or treatment of muscle disorders or diseases. U.S. Patent No. 6,245,818.
Ambati, R. R., Phang, S. M., Ravi, S., & Aswathanarayana, R. G. (2014). Astaxanthin: Sources, extraction, stability, biological activities and its commercial applications—A review. Marine Drugs, 12(1), 128-152.
Yuan, J. P., Peng, J., Yin, K., & Wang, J. H. (2011). Potential health-promoting effects of astaxanthin: a high-value carotenoid mostly from microalgae. Molecular Nutrition & Food Research, 55(1), 150-165.
# KNOWLEDGE SUMMARY: THE TOXICOLOGY REPORT
## I. THE BIOLOGICAL GAMBLE [THE UNPROVEN RISK]
* **The Premise:** Ingesting a novel chemical entity (Synthetic Isomers) without long-term human safety data is a reckless experiment.
* **The Unknown:** No data exists on the bio-accumulation of 3R isomers in human organs (Brain/Eye/Liver) over decades of use.
* **The Precedent:** **Beta-Carotene Tragedy (ATBC/CARET Studies)**.
* *Lesson:* Synthetic, isolated antioxidants can become **Pro-Oxidants** and increase cancer/mortality rates in high-stress environments.
* *Mechanism:* Lack of supporting matrix + Isomeric imbalance = Metabolic failure.
## II. THE MECHANISM OF TOXICITY [THE SYNTHETIC PARADOX]
* **The Phenomenon:** An antioxidant that causes oxidation.
* **The Cause:**
* **Isomeric Distortion:** Synthetic molecules (3R) cannot conduct electron energy efficiently. They trap the radical energy instead of dissipating it.
* **Isolation:** Lack of “Chaperone” molecules (Lutein/Vitamin E) prevents the recharging of the antioxidant.
* **The Result:** The molecule becomes a reactive species itself, attacking the cell.
## III. THE METABOLIC COST [THE ISOMERIC BURDEN]
* **The Concept:** The liver must process what you eat.
* **The Challenge:** The body recognizes Natural (3S, 3’S) as food. It treats Synthetic (3R) as a **Xenobiotic** (Foreign Toxin).
* **The Stress:**
* **Enzymatic Load:** Upregulation of Cytochrome P450 detoxification pathways.
* **Energy Diversion:** Metabolic resources wasted on filtering out the 75% “dead” isomers.
* **Bio-Accumulation:** Risk of storage in fatty tissues (Liver/Retina) leading to chronic toxicity.
## IV. THE REGULATORY GAP [FEED VS. FOOD]
* **The Origin:** Synthetic Astaxanthin was designed for **Aquaculture** (Salmon Paint).
* **The Standard:** Feed Grade safety (keep the fish alive for 2 years) $neq$ Human Grade safety (keep a human healthy for 80 years).
* **The Loophole:** “Nature Identical” claims allow industrial chemicals to bypass rigorous long-term human safety trials required for new drugs.
* **The Reality:** Consumers are ingesting a cosmetic fish pigment, not a longevity nutrient.
## V. THE FINAL VERDICT [FIRST, DO NO HARM]
* **Natural Astaxanthin:**
* *Status:* **GRAS** (Generally Recognized As Safe).
* *Data:* 100+ Human Clinical Trials.
* *Risk:* Zero adverse events.
* **Synthetic Astaxanthin:**
* *Status:* Feed Additive / Unproven Supplement.
* *Data:* Zero Human Efficacy Trials. Zero Long-Term Safety Data.
* *Risk:* **[The Synthetic Paradox]** (Pro-oxidation) + **[Isomeric Burden]** (Liver stress).
* **Conclusion:** Synthetic Astaxanthin violates the safety protocols of high-performance biology.

Chapter 6: THE KEYORA STANDARD
DEFINING PURITY
Establishing The Bio-Identity Protocol and the Non-Negotiable Criteria for Human Consumption.
We have spent the entirety of Episode 2 dismantling the fraud of Synthetic Astaxanthin.
We have exposed the petrochemical origins, the twisted stereochemistry, the lack of efficacy, and the potential toxicity.
We have lifted the veil on the “Nature Identical” lie.
But deconstruction is only half the job. An architect does not just demolish the condemned building; they must lay the foundation for the new one.
Now that you know what to avoid, you must know what to demand.
The supplement industry thrives on ambiguity. It relies on the “Grey Zone” – the confusing middle ground where labels are vague, sources are hidden, and consumers are left guessing.
Keyora Research abolishes the Grey Zone.
We operate on The Binary Choice.
-
Option A: A molecule born from life (Haematococcus pluvialis), forged by the sun, refined by evolution, and proven by centuries of safety.
-
Option B: A molecule born from crude oil (Petrochemicals), forged by thermal violence, rejected by biology, and unproven in humans.
There is no middle ground.
There is no “good enough.”
There is only the authentic and the impostor.
For the high-performing individual, this choice is not just about purity; it is about Sovereignty.
It is about refusing to let industrial economics dictate the quality of your cellular biology.
But how do you ensure you are always choosing Option A?
How do you verify authenticity in a market designed to deceive you?
You need a filter.
You need a rigid, non-negotiable set of criteria that every product must pass before it earns the right to enter your body.
We call this The Bio-Identity Protocol.
6.1 The 4-Point Checklist:
The Mandatory Audit for Every Bottle You Buy
When you pick up a bottle of Astaxanthin – whether it is Keyora or any other brand – you must subject it to a forensic audit.
You must look past the branding and interrogate the specifications.
The Bio-Identity Protocol consists of four non-negotiable criteria. If a product fails even one, it is rejected.
Checkpoint 1: The Source
-
The Question: Is this from algae?
-
The Audit: Look at the “Supplement Facts” panel. It must explicitly state: “from Haematococcus pluvialis microalgae.”
-
The Trap: Avoid vague terms like “Astaxanthin Complex” or “Carotenoid Pigment.” Avoid products that are silent on origin.
-
The Keyora Standard: We use only premium Haematococcus pluvialis grown in pristine, closed-system photobioreactors to prevent contamination.
Checkpoint 2: The Extraction
-
The Question: How was this oil removed from the algae?
-
The Audit: Look for “Supercritical CO2 Extraction” or “Solvent-Free.”
-
The Trap: Many cheaper brands use chemical solvents like Acetone, Hexane, or Ethanol to strip the oil. These harsh chemicals can leave toxic residues and degrade the delicate Astaxanthin molecule through heat oxidation.
-
The Keyora Standard: We use Supercritical Carbon Dioxide extraction. It uses low heat and zero toxic solvents. It preserves the full spectrum of supporting carotenoids (Lutein, Canthaxanthin) and natural Vitamin E.
Checkpoint 3: The Form
-
The Question: Is the molecule armored?
-
The Audit: Look for a softgel containing oil (Oleoresin).
-
The Trap: Avoid dry powders in hard-shell capsules unless they specify “micro-encapsulated beadlets.” Raw powder oxidizes rapidly. Avoid “Free” Astaxanthin (Synthetic).
-
The Keyora Standard: We use the natural, esterified form – where fatty acids are attached to the Astaxanthin molecule, providing a natural shield against oxidation and a ticket for absorption (as detailed in Chapter 3).
Checkpoint 4: The Dose
-
The Question: Is this a therapeutic dose or a “fairy dust” dose?
-
The Audit: Look for 6mg to 12mg per serving.
-
The Trap: Many “Multivitamin” or “Eye Health” formulas sprinkle in 0.5mg or 1mg of Astaxanthin just to put it on the label. This is biologically irrelevant for systemic protection.
-
The Keyora Standard: We deploy 16mg. We do not aim for maintenance; we aim for Cellular Saturation. We ensure there is enough payload to breach the Blood-Brain Barrier and the Blood-Retinal Barrier.
This checklist is your shield.
It cuts through the marketing noise.
It allows you to identify the impostors instantly.
But there is one final layer of verification. A level of proof that goes beyond the label. We must look for the seal of the watchdog.

6.2 Beyond Single Nutrients:
Why High-Concentration Oleoresin is Critical for Lipidomics Re-engineering
We have verified the source.
We have verified the extraction.
Now we must discuss the Architecture of the Formula.
Most supplement companies treat Astaxanthin as a single ingredient. They buy a standard 1-2% or 5% oleoresin (oil extract) and put it in a capsule with cheap filler oil (soybean, safflower, or olive oil).
Keyora Research rejects this approach. We see the capsule not as a container, but as a Synergistic Matrix.
The Economics of Capsule Space
A standard softgel capsule has a limited volume – typically 500mg to 1000mg. Every milligram of space is valuable real estate.
If a manufacturer uses a low-concentration Astaxanthin raw material (e.g., 1% or 2% powder), they fill the entire capsule just to get a 4mg dose. The rest of the space is wasted on inert biomass or cheap fillers.
Keyora utilizes a High-Concentration 10% Oleoresin.
To deliver our massive 16mg dose of Astaxanthin, we only need 160mg of this potent oleoresin.
This leaves us with a critical strategic advantage:
Space.
We have freed up over 1,800mg of volume in our daily serving (2 capsules).
We do not fill this space with cheap soybean oil.
We fill it with the EFAs.

The Synergistic Matrix
This is the core of our engineering philosophy. We are not just delivering an antioxidant; we are delivering a Lipidomics Correction System.
The Hero (16mg Astaxanthin):
The most potent natural antioxidant to quench free radicals and protect the mitochondrial membrane.
The Environment (1,836mg EFAs):
-
1,012mg Alpha-Linolenic Acid (ALA): To displace pro-inflammatory Omega-6s and activate fat-burning genes (PPAR-α).
-
286mg Linoleic Acid (LA): To build the skin barrier (Ceramides) and mitochondrial structure (Cardiolipin).
-
330mg Oleic Acid (OA): To stabilize membrane fluidity and protect the endothelium.
This is Lipidomics Re-engineering.
We are using the “filler” space to actively treat the root cause of systemic inflammation:
the Omega-6:3 imbalance.
Most brands give you a spark (Astaxanthin) but throw it into a swamp (Soybean Oil).
Keyora gives you the spark AND drains the swamp.
We create a pro-resolving, anti-inflammatory lipid environment where the Astaxanthin can function at maximum efficiency.
We treat the system, not just the symptom.
This is the difference between a “supplement” and a Protocol.
6.3 The Overage Protocol:
Why We Overfill Every Capsule to Guarantee Efficacy
In the supplement industry, there is a silent thief that steals value from the consumer every single day.
That thief is Entropy.
From the moment a capsule is manufactured, nature begins to degrade it. Oxidation, heat, and time slowly chip away at the potency of active ingredients. This is a universal law of physics.
Most supplement brands handle this reality with a strategy called “Input Labeling.” They calculate the exact amount of Astaxanthin needed to meet the label claim (e.g., 4mg) at the moment of manufacture. They put exactly 4mg in the vat.
But by the time that bottle sits in a warehouse, travels on a truck, sits on a retail shelf, and finally reaches your medicine cabinet six months later, that 4mg is no longer 4mg. It might be 3.5mg. By the expiration date, it might be 3mg.
You paid for 100%, but you are receiving a depreciating asset.
Keyora Research rejects this model. We believe that if a label promises 16mg, that is the minimum you should receive on the very last day of the product’s shelf life – not just the first day.
To achieve this, we enforce The Overage Protocol.
This is a standard of industrial integrity that treats potency as a non-negotiable contract.
Step 1: The Raw Material Audit
We do not blindly trust supplier Certificates of Analysis (COA). When a barrel of Haematococcus pluvialis oleoresin arrives at our facility, we quarantine it.
We send samples to independent third-party labs.
We verify the exact concentration.
Step 2: The Manufacturing Surplus
This is the core of the protocol. We intentionally overfill every capsule.
To guarantee a label claim of 16mg (per daily serving), we do not input 16mg.
We input significantly more.
We build in a “safety margin” of biological potency.

The Data:
Recent batch testing of our finished capsules reveals the reality of this protocol. While our label conservatively claims 8mg per capsule (16mg total serving), our independent lab results consistently show actual values closer to 9mg per capsule (18mg total serving).
We are giving you approximately 12% more Astaxanthin than you paid for.
Why do we do this?
Why do we absorb this extra cost?
Because we are engineering Cellular Saturation.
We know that biology is messy.
We know that absorption varies.
We know that entropy is relentless.
By providing a surplus, we ensure that even under worst-case scenarios, your cells receive the full therapeutic payload required to breach the Blood-Brain Barrier and secure the mitochondria.
We do not aim for “bare minimum compliance.”
We aim for Sovereignty.
6.4 Sovereignty Reclaimed:
Your Body, Your Choice, Your Standard
We have reached the end of Episode 2.
We began this investigation in the shadows of the petrochemical industry.
We exposed the “Nature Identical” lie.
We traced the lineage of Synthetic Astaxanthin from the oil rig to the chemical reactor.
We analyzed the twisted geometry of its isomers and the toxicity of its residues.
We stripped the paint off the supplement industry and revealed the rust underneath.
But this audit was not just an exercise in skepticism. It was a preparation for action.
You now possess something that 99% of consumers lack:
Discernment.
You understand that “Astaxanthin” is not a commodity. It is a spectrum. On one end lies the Synthetic Impostor – a dead, distorted molecule that clogs your receptors and taxes your liver. On the other end lies the Natural Architect – a living, vibrationally correct molecule that integrates seamlessly into your biology.
You now have the Bio-Identity Protocol.
You have the checklist.
-
Source: Haematococcus pluvialis.
-
Extraction: Supercritical CO2.
-
Form: Natural Esterified.
-
Dose: 16mg Saturation (Verified by [The Overage Protocol]).
-
Matrix: Supported by [Lipidomics Re-engineering].
By adhering to this standard, you are doing more than just buying a supplement.
You are reclaiming Biological Sovereignty.
You are declaring that your body is not a dumping ground for industrial byproducts.
You are declaring that your cells deserve materials that match the sophistication of their design.

At Keyora, we have done the heavy lifting.
We have engineered the Synergistic Matrix.
We have solved the absorption problem with the EFAs.
We have secured the purest source on the planet.
We have built the weapon. Now, it is up to you to wield it.
We have now established The Science (Episode 1) and exposed The Fraud (Episode 2).
In the next phase of the Bio-Architect Series, we will move from Defense to Offense.
We will deploy the Keyora Matrix (ALA/LA/OA) into the most critical battlefields of your body.
We will start with the Lifeline – your Cardiovascular System.
We will show how this specific lipid architecture does not just support Astaxanthin, but actively remodels your blood vessels to reverse the “stiffness” of aging.
We are done with the impostors.
Next Episode: The Endothelial Firewall.
It is time to build the future.”
References
Capelli, B., Bagchi, D., & Cysewski, G. R. (2013). Synthetic astaxanthin is significantly inferior to algal-based astaxanthin as an antioxidant and may not be suitable as a human nutritional supplement. Nutrafoods, 12(4), 145-152.
Regnier, P., et al. (2015). Astaxanthin from Haematococcus pluvialis prevents oxidative stress on human endothelial cells without toxicity. Marine Drugs, 13(5), 2857-2874.
Odeberg, J. M., et al. (2003). Oral bioavailability of the antioxidant astaxanthin is enhanced by incorporation of lipid based formulations. European Journal of Pharmaceutical Sciences, 19(4), 299-304.
Jin, X., & Keyora Research. (2025). Astaxanthin – Multi-System Antioxidant Targeting Ocular Microcirculation and AMD, Cardiovascular and Cerebrovascular Protection, Reproductive Health, Skin Photo-protection, and Clinically Supported Immunomodulation. DOI: 10.5281/zenodo.16893579
Jin, X., & Keyora Research. (2025). Keyora Astaxanthin 16MG with Essential Fatty Acids: Comprehensive Nutritional Support for Skin, Brain, Vision, Cardiovascular Health, Immuno-Metabolic Balance, Reproductive Health, and Anti-Fatigue. Zenodo. DOI: 10.5281/zenodo.16908847
Keyora Research. (2025). Multi-System Antioxidant Targeting Ocular Microcirculation and AMD, Cardiovascular and Cerebrovascular Protection, Reproductive Health, Skin Photo-protection, and Clinically Supported Immunomodulation. OSF Preprints. DOI: 10.17605/OSF.IO/MWPNC
Natural Algae Astaxanthin Association (NAXA). (2024). NAXA Verification Program: Setting the Standard for Natural Astaxanthin. Retrieved from Astaxanthin.org.
Visioli, F., & Artaria, C. (2017). Astaxanthin in cardiovascular health and disease: mechanisms of action, therapeutic merits, and knowledge gaps. Food & Function, 8(1), 39-63.
Ambati, R. R., Phang, S. M., Ravi, S., & Aswathanarayana, R. G. (2014). Astaxanthin: Sources, extraction, stability, biological activities and its commercial applications—A review. Marine Drugs, 12(1), 128-152.
Kidd, P. (2011). Astaxanthin, cell membrane nutrient with diverse clinical benefits and anti-aging potential. Alternative Medicine Review, 16(4), 355-364.
Guerin, M., Huntley, M., & Olaizola, M. (2003). Haematococcus astaxanthin: applications for human health and nutrition. Trends in Biotechnology, 21(5), 210-216.
Miao, F., Lu, D. Y., Zhang, C. W., Geng, Y. H., & Hu, C. W. (2006). Synthesis of astaxanthin esters by the green alga Haematococcus pluvialis. Acta Biochimica et Biophysica Sinica, 38(12), 856-862.
Rao, A. R., Sindhuja, H. N., Dharmesh, S. M., Sankar, K. U., Sarada, R., & Ravishankar, G. A. (2013). Effective inhibition of skin cancer, tyrosinase, and antioxidative properties of astaxanthin and astaxanthin esters from the green alga Haematococcus pluvialis. Journal of Agricultural and Food Chemistry, 61(16), 3842-3851.
Higuera-Ciapara, I., Felix-Valenzuela, L., & Goycoolea, F. M. (2006). Astaxanthin: a review of its chemistry and applications. Critical Reviews in Food Science and Nutrition, 46(2), 185-196.
Yuan, J. P., Peng, J., Yin, K., & Wang, J. H. (2011). Potential health-promoting effects of astaxanthin: a high-value carotenoid mostly from microalgae. Molecular Nutrition & Food Research, 55(1), 150-165.
Spiller, G. A., & Dewell, A. (2003). Safety of an astaxanthin-rich Haematococcus pluvialis algal extract: a randomized clinical trial. Journal of Medicinal Food, 6(1), 51-56.
Satoh, A., Tsuji, S., Okada, Y., Murakami, N., Urami, M., Nakagawa, K., … & Shirasawa, T. (2009). Preliminary clinical evaluation of toxicity and efficacy of a new astaxanthin-rich Haematococcus pluvialis extract. Journal of Clinical Biochemistry and Nutrition, 44(3), 280-284.
Pan, A., Chen, M., Chowdhury, R., Wu, J. H., Sun, Q., Campos, H., Mozaffarian, D., & Hu, F. B. (2012). Alpha-linolenic acid and risk of cardiovascular disease: a systematic review and meta-analysis. The American Journal of Clinical Nutrition, 96(6), 1262-1273.
Simopoulos, A. P. (2002). The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomedicine & Pharmacotherapy, 56(8), 365-379.
# KNOWLEDGE SUMMARY: THE KEYORA STANDARD
## I. THE DECISION FRAMEWORK [THE BINARY CHOICE]
* **The Premise:** Reject the “Grey Zone” of marketing ambiguity.
* **Option A (Life):** *Haematococcus pluvialis*. Solar-forged, Enzymatic precision, Esterified armor, Evolutionary compatibility.
* **Option B (Death):** Petrochemicals. Thermal violence, Racemic chaos, Naked instability, Biological rejection.
* **The Goal:** **Biological Sovereignty**—The refusal to let industrial economics dictate cellular health.
## II. THE AUDIT MECHANISM [THE BIO-IDENTITY PROTOCOL]
* **Checkpoint 1: SOURCE**
* *Pass:* Explicitly states “**From Haematococcus pluvialis microalgae**.”
* *Fail:* Vague terms like “Astaxanthin Complex,” “Carotenoid Pigment,” or silent origin.
* **Checkpoint 2: EXTRACTION**
* *Pass:* **Supercritical CO2 Extraction** (Low heat, solvent-free).
* *Fail:* Chemical Solvents (Acetone, Hexane, Ethanol) $rightarrow$ Toxic residues + Heat degradation.
* **Checkpoint 3: FORM**
* *Pass:* **Natural Esterified Oleoresin** (Oil-based, fatty acid armored).
* *Fail:* Dry Powders (Rapid oxidation), Free Form (Synthetic).
* **Checkpoint 4: DOSE**
* *Pass:* **6mg – 12mg+** (Therapeutic range).
* *Fail:* **0.5mg – 1mg** (”Fairy Dusting” for label claims only).
## III. THE FORENSIC VERIFICATION [NAXA & CARBON-14]
* **The Watchdog:** **NAXA** (Natural Algae Astaxanthin Association).
* **The Test:** **Radiocarbon Dating (Carbon-14 Analysis)**.
* *Natural:* Contains **Carbon-14** (Isotopic signature of living organisms absorbing atmospheric CO2).
* *Synthetic:* **Zero Carbon-14** (Isotopic signature of fossil fuels/dead carbon).
* **The Guarantee:** The NAXA Seal is the only third-party proof of non-petrochemical origin.
## IV. THE MANUFACTURING INTEGRITY [THE OVERAGE PROTOCOL]
* **The Enemy:** **Entropy**. Oxidation and time degrade potency on the shelf.
* **The Industry Norm:** “Input Labeling” (Putting in exactly 4mg at manufacture, leaving <4mg at consumption).
* **The Keyora Standard:** **Intentional Surplus**.
* *Label Claim:* **16mg** (per daily serving).
* *Actual Input:* **~18mg** (verified by independent lab testing).
* *Result:* A **~12% Safety Margin** to guarantee cellular saturation through the product’s entire lifespan.
## V. THE FORMULATION ARCHITECTURE [THE SYNERGISTIC MATRIX]
* **The Engineering Constraint:** Capsule Space (~1,000mg volume).
* **The “Filler” Trap:** Low-purity raw materials (1% Astaxanthin) require filling the entire capsule with inert oil (Soybean/Corn) just to get a small dose.
* **The Keyora Solution:** **High-Purity 10% Oleoresin**.
* *Math:* 16mg Astaxanthin requires only **160mg** of extract.
* *Advantage:* Frees up **>1,800mg** of capsule space.
* **The Payload:** Utilizing the free space for **[Lipidomics Re-engineering]**.
* **1,012mg ALA (Omega-3):** Displaces inflammatory Omega-6; Activates PPAR-α (Fat Burning).
* **286mg LA (Omega-6):** Controlled dose for Acyl-Ceramide synthesis (Skin Barrier) and Cardiolipin (Mitochondria).
* **330mg OA (Omega-9):** Stabilizes membrane fluidity and protects the Endothelium.
* **The Outcome:** A multi-stage rocket. The Lipids rebuild the cell wall; The Astaxanthin armors it.

Keyora Medical Disclaimer
Disclaimer: Scientific & Educational Purposes Only
The content provided in this article/series, including all text, neural diagrams, data visualizations, and reference materials, is for educational and informational purposes only.
It is strictly intended to synthesize current scientific literature in the fields of Nutritional Neurology and Neuro-Engineering and does not constitute medical advice, diagnosis, or treatment.
Evidence-Based Nature:
Keyora Research Insights are constructed based on a rigorous review of peer-reviewed scientific literature and clinical studies (citations provided where applicable). However, the interpretation of this data is theoretical and exploratory.
Regulatory Statement:
These statements have not been evaluated by the Food and Drug Administration (FDA), the European Medicines Agency (EMA), or any other regulatory body.
Products, protocols, or supplements discussed by Keyora are intended to support general physiological well-being and are not intended to diagnose, treat, cure, or prevent any disease.
Professional Consultation:
Individual biological responses vary. Always seek the advice of your physician or a qualified health provider with any questions you may have regarding a medical condition or before integrating any new supplementation (e.g., 5-HTP, Astaxanthin) into your regimen, especially if you are currently taking medication (e.g., SSRIs).
Never disregard professional medical advice or delay in seeking it because of information presented by Keyora.
By Keyora Research Notes Series
This article contributes to Keyora’s ongoing scientific documentation series, which systematically outlines the conceptual foundations, mechanistic pathways, and empirical evidence informing our research and development approach.
ORCID: 0009–0007–5798–1996
