Keyora Nutritional Neurology – Ashwagandha · Episode 2

By Keyora Research Notes Series
This article contributes to Keyora’s ongoing scientific documentation series, which systematically outlines the conceptual foundations, mechanistic pathways, and empirical evidence informing our research and development approach.
ORCID: 0009–0007–5798–1996
Why the HPA Axis Matters:
The Hidden Core of Modern Stress Disorders
Modern stress is not merely psychological pressure or emotional overload – it is a progressive breakdown of the body’s primary stress-regulation system:
the hypothalamic–pituitary–adrenal (HPA) axis.
The HPA axis serves as the master conductor of the stress response, orchestrating cortisol rhythmicity, metabolic readiness, immune signaling, cognitive alertness, and emotional stability.
When functioning properly, it enables humans to respond to challenges with precision, recover rapidly, and maintain long-term resilience.
However, in contemporary environments characterized by chronic cognitive demands, disrupted sleep cycles, constant digital stimulation, social pressure, and unresolved emotional stressors, the HPA axis is consistently activated beyond its physiological capacity.
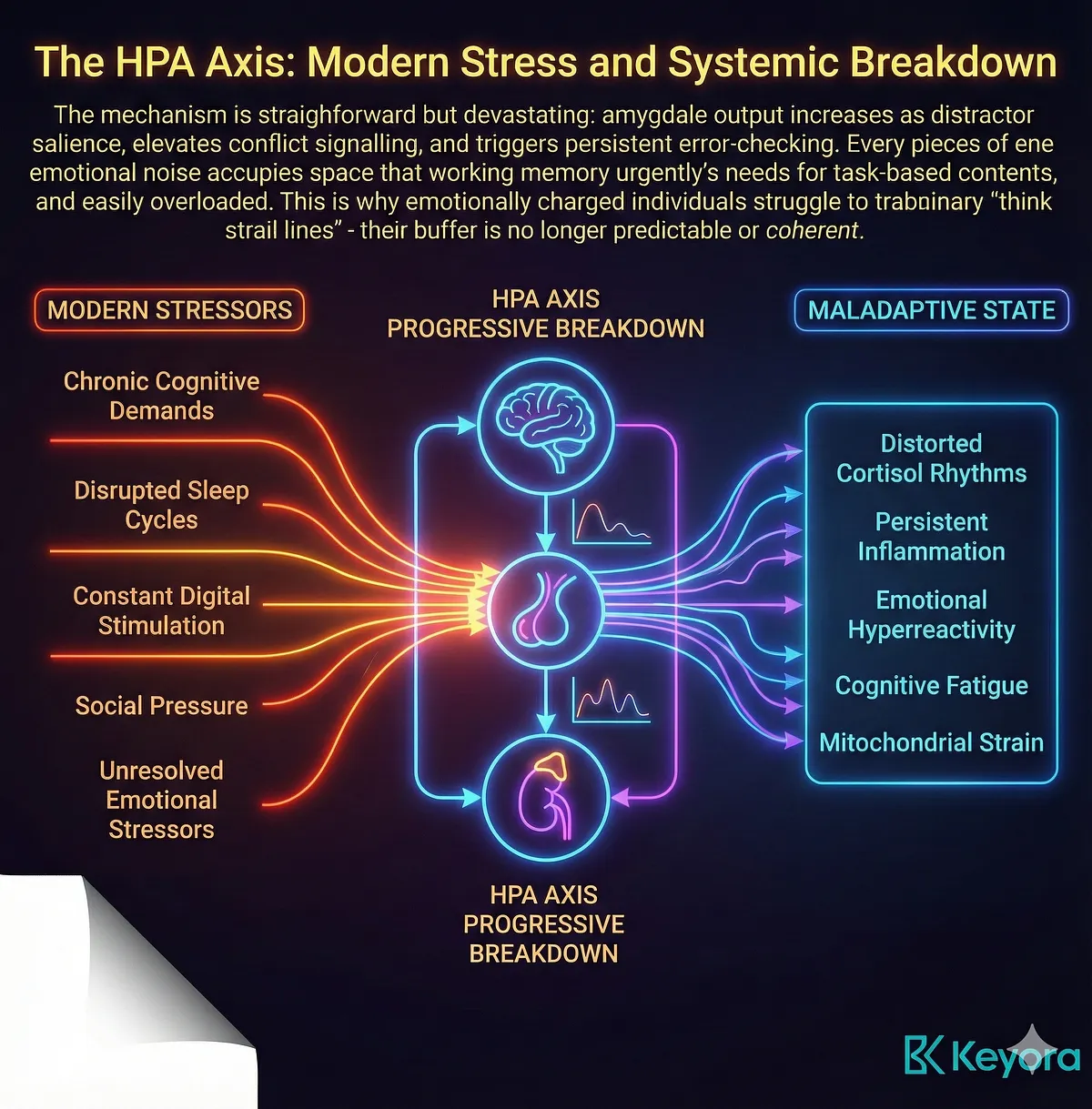
What begins as a healthy adaptive response gradually transforms into a maladaptive state – one marked by distorted cortisol rhythms, persistent inflammation, emotional hyperreactivity, cognitive fatigue, and mitochondrial strain.
These disruptions accumulate silently, often years before individuals recognize the symptoms.
The clinical presentation of HPA dysregulation is broad yet recognizable:
- Chronic fatigue that persists despite rest
- Anxiety, irritability, and emotional volatility
- Reduced stress tolerance and heightened sensitivity to triggers
- Brain fog, reduced attention span, and slower cognitive processing
- Sleep disruption – difficulty falling asleep, early awakenings, or non-restorative sleep
- Afternoon crashes and evening alertness(cortisol inversion)
- Increased inflammation, frequent minor illnesses, or persistent colds
- Digestive instability, appetite changes, and metabolic fluctuation
- “Wired but tired” states, where the brain remains hyperalert despite exhaustion
These symptoms are not random – they are the predictable biological signatures of an HPA axis that has lost its coherence.

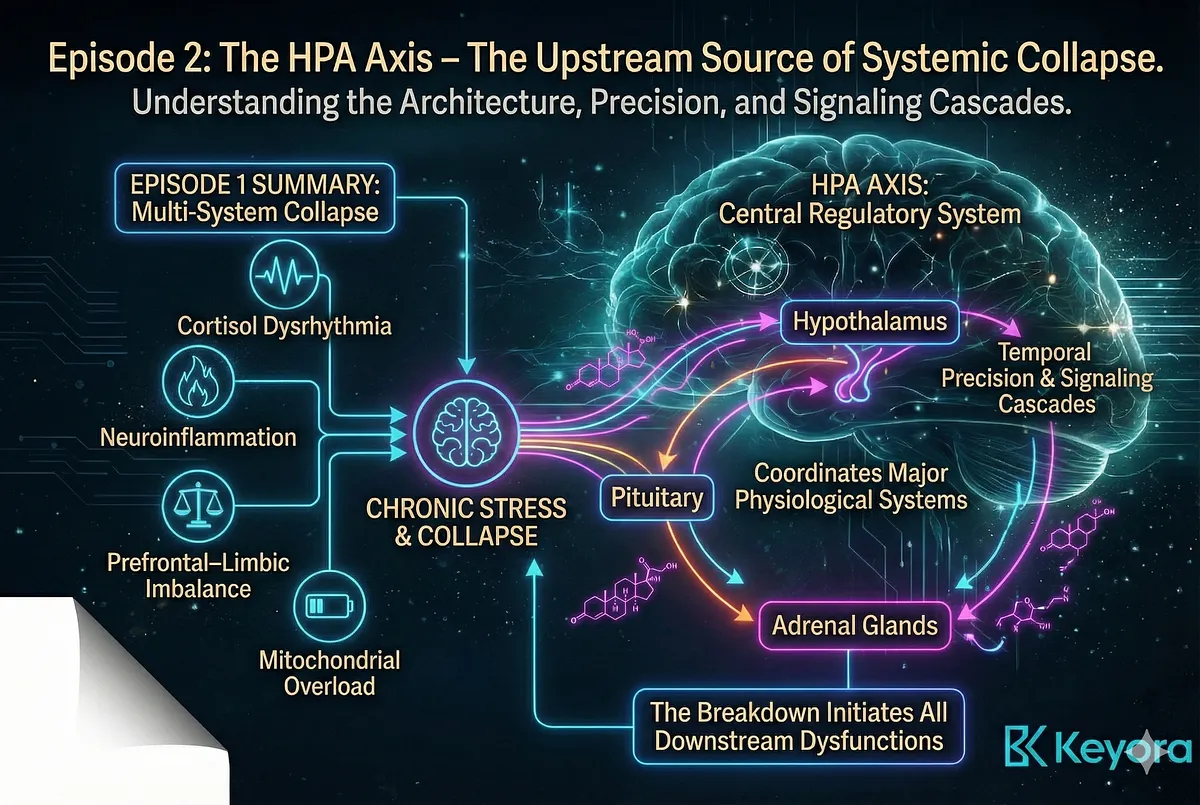
Episode 1 demonstrated how chronic stress leads to a multi-system collapse involving cortisol dysrhythmia, neuroinflammation, prefrontal–limbic imbalance, and mitochondrial overload. Episode 2 now turns to the upstream source of this collapse. The HPA axis is not simply one part of the stress response – it is the central regulatory system whose breakdown initiates all downstream dysfunctions.
To understand how stress becomes chronic – and how a multi-target adaptogen like Ashwagandha can reverse it – we must first understand the architecture of the HPA axis, its temporal precision, and how its signaling cascades coordinate every major physiological system.
– Modern stress overwhelms the HPA axis, the body’s master regulator of stress physiology.
– Chronic activation → cortisol dysrhythmia, inflammation, cognitive fatigue, emotional instability, sleep disruption.
– Real-world symptoms: fatigue, anxiety, irritability, brain fog, poor sleep, low resilience, wired-but-tired states.
– HPA dysregulation is the upstream driver of all system failures described in Episode 1.
– Episode 2 explores HPA architecture to explain how stress becomes chronic and why Ashwagandha targets this axis.
Section I
The Architecture of the HPA Axis:
A Multi-Layered Neuroendocrine Control System
The hypothalamic–pituitary–adrenal (HPA) axis is the central command system governing human stress physiology.
Far from being a simple hormone cascade, it is a multi-layered neuroendocrine network that integrates sensory processing, emotional states, circadian information, metabolic demand, immune status, and environmental threat detection.
Understanding its architecture is fundamental to understanding how stress transforms from adaptive to pathological – and why interventions such as Ashwagandha must begin upstream at this regulatory system.
At the apex of the axis is the hypothalamus, a region that functions as the body’s master homeostatic integrator.
Specialized paraventricular nucleus (PVN) neurons synthesize and release corticotropin-releasing hormone (CRH), the primary initiator of the stress response.
These neurons receive converging signals from the amygdala, hippocampus, prefrontal cortex, circadian centers (suprachiasmatic nucleus, SCN), and metabolic sensors.
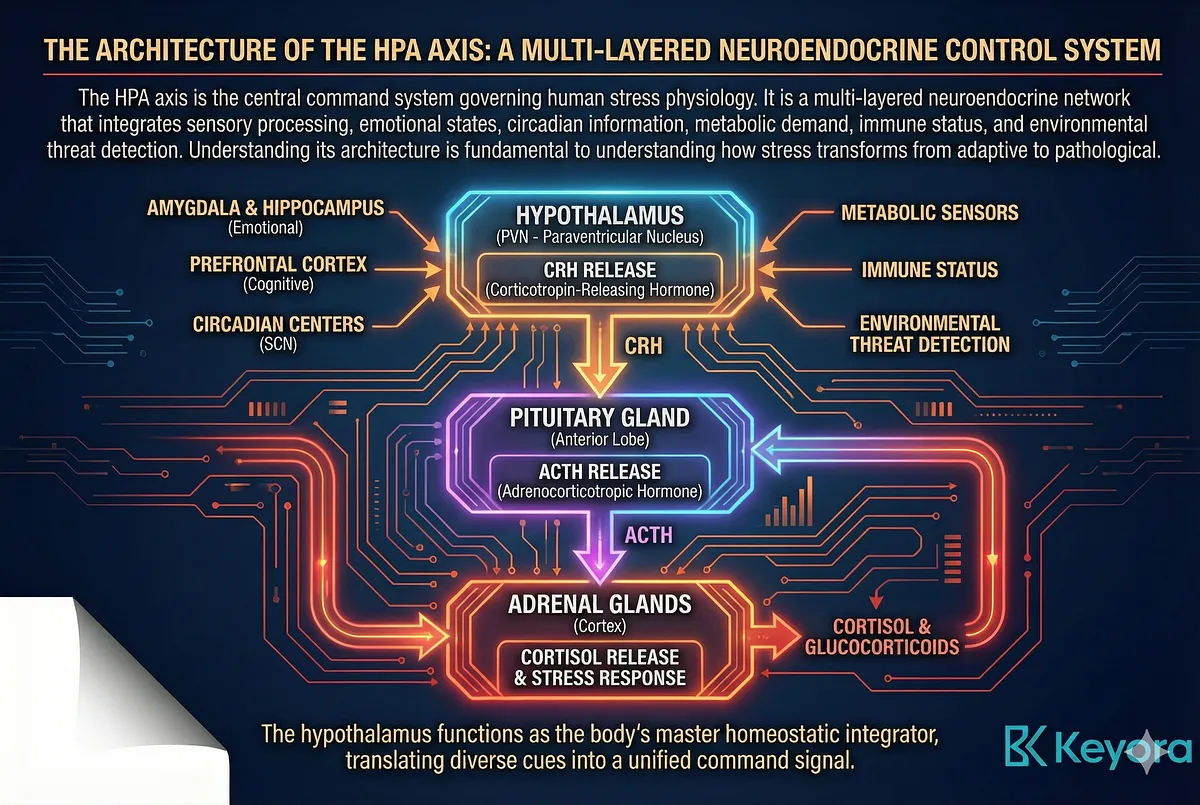
In effect, CRH neurons translate emotional, cognitive, environmental, and physiological cues into a unified command signal.
The firing pattern of CRH neurons – both in amplitude and in ultradian pulsatility – determines how the HPA axis engages the rest of the body.
CRH travels via the hypophyseal portal system to the pituitary gland, where it stimulates the anterior pituitary to release adrenocorticotropic hormone (ACTH). ACTH secretion follows both circadian and ultradian rhythms, producing pulses approximately every hour that shape cortisol’s oscillatory pattern.
The pituitary does not simply relay CRH signals; it integrates them with immune-derived cytokine signals (such as IL-6) and metabolic states, making it a dynamic regulator. ACTH’s pulsatile release is crucial:
without pulsatility, cortisol cannot maintain its adaptive oscillations.
ACTH acts on the adrenal cortex, specifically the zona fasciculata, activating steroidogenic enzymes that synthesize cortisol from cholesterol.
Cortisol’s release follows a precise temporal architecture:
- A large morning surge (Cortisol Awakening Response, CAR)
- An hourly ultradian rhythm
- A steady decline towards evening
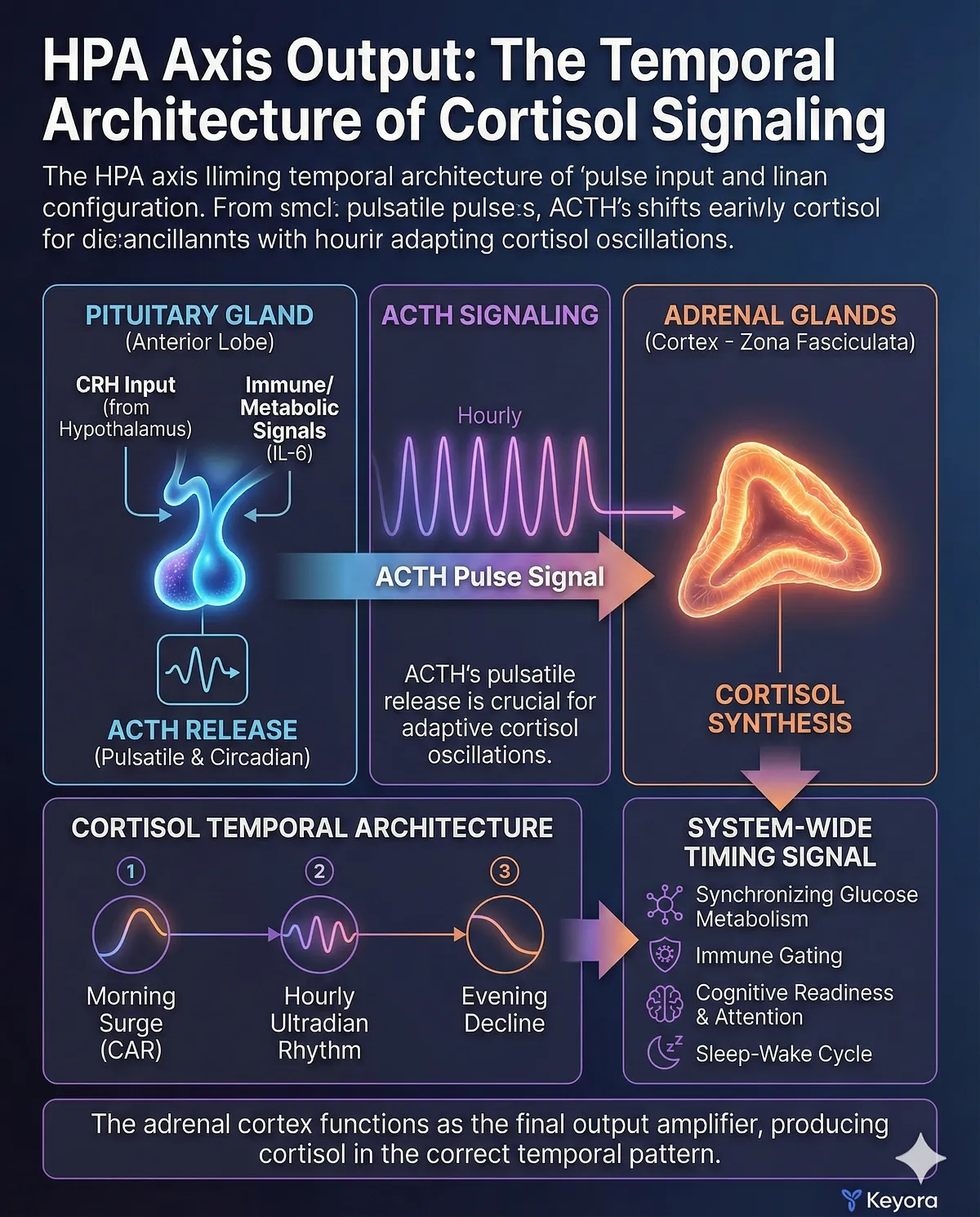
This temporal structure is as important as the hormone itself. Cortisol is not only a metabolic enzyme regulator – it acts as a system-wide timing signal, synchronizing glucose metabolism, immune gating, cognitive readiness, attention, and the sleep–wake cycle.
The adrenal cortex therefore functions as the final output amplifier of the axis: its job is not merely to produce cortisol but to produce it in the correct temporal pattern.
Closing the loop, cortisol engages negative feedback receptors across the brain and body. Two receptor classes orchestrate this feedback:
- Mineralocorticoid receptors (MR) – highly sensitive, maintaining baseline stability
- Glucocorticoid receptors (GR) – activated by higher cortisol levels, terminating stress responses
This feedback loop ensures that cortisol elevation is brief and adaptive, preventing overactivation of immune, metabolic, and neural circuits. The hippocampus and prefrontal cortex, rich in GRs, function as “brakes” on the axis.
When they are damaged by chronic stress – through inflammation, excitotoxicity, or metabolic strain – the brake system weakens, and the HPA axis becomes dysregulated.
A final architectural layer is the integration between the HPA axis and circadian machinery.
The SCN entrains CRH pulses, ACTH rhythms, and adrenal sensitivity via neural and hormonal pathways.
This alignment with light–dark cycles ensures that cortisol supports daytime alertness and nighttime recovery.
When SCN–HPA coupling falters – as happens with chronic stress, irregular sleep, or inflammatory signaling – the entire axis becomes desynchronized, producing flattened curves and mistimed cortisol peaks.
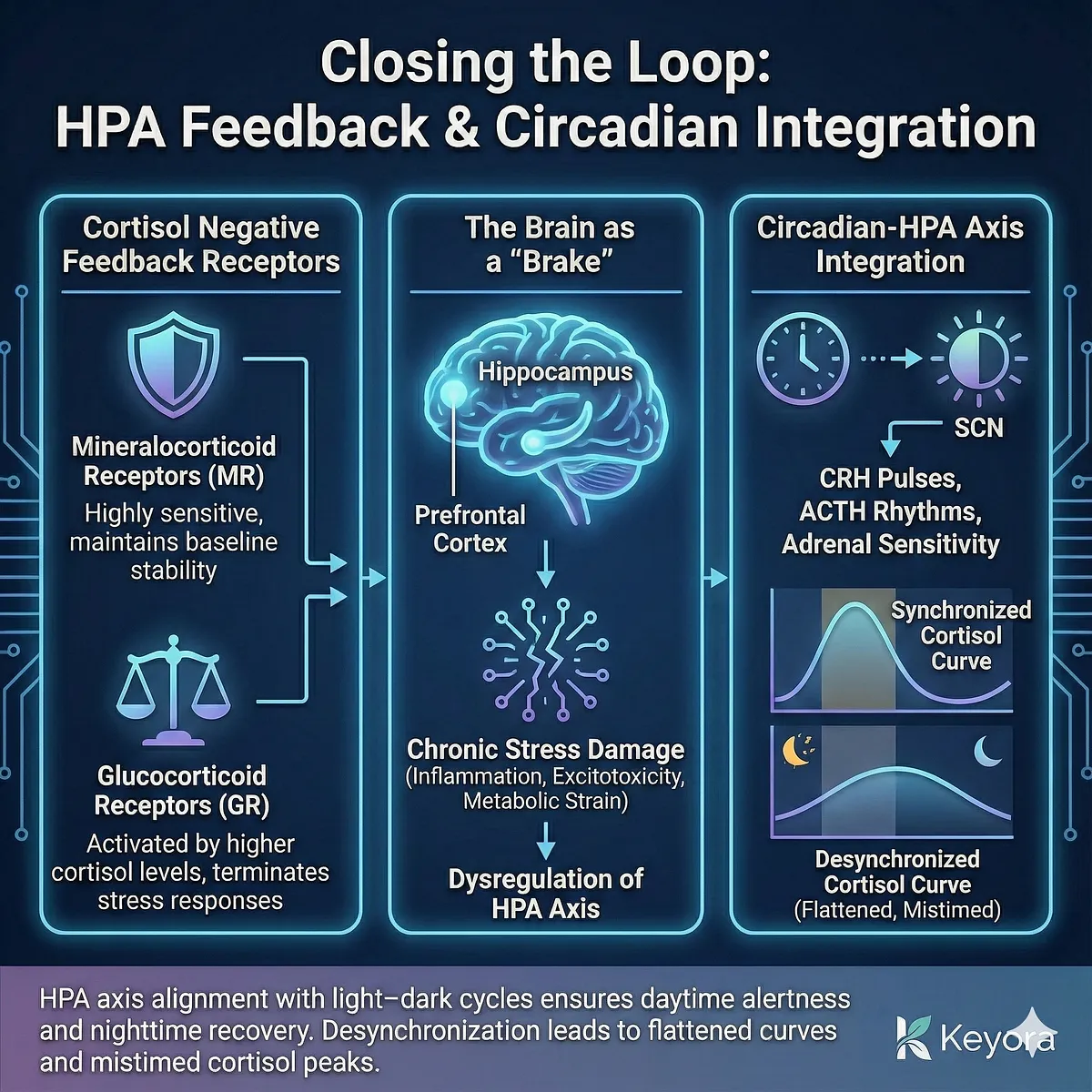
Taken together, the HPA axis is not a linear CRH→ACTH→cortisol pathway but a multi-level adaptive control system.
It integrates emotional processing, immune status, circadian timing, and metabolic demand into a coherent physiological response.
This architecture allows the body to shift rapidly between rest and mobilization, resilience and recovery. But because the system is interconnected and recursive, disruption at any node propagates throughout the network – setting the stage for the cascade of failures described in Episode 1 and explored in later sections of Episode 2.
– HPA axis = central neuroendocrine system integrating emotional, metabolic, immune, and circadian signals.
– Hypothalamus (PVN): CRH release shaped by amygdala, PFC, hippocampus, SCN.
– Pituitary: ACTH secretion with circadian + ultradian pulsatility; integrates cytokine signals.
– Adrenal cortex: produces cortisol with CAR + hourly pulses + diurnal decline.
– Cortisol = systemic timing signal coordinating metabolism, cognition, immunity, and sleep-wake rhythms.
– Negative feedback: MR/GR receptors in hippocampus & PFC regulate shutdown.
– SCN–HPA coupling ensures correct temporal alignment of cortisol.
– HPA = multi-layer control system, not a linear cascade; disruption spreads across systems.

Section II
Cortisol as a Biological Timing Signal:
The Central Role of Rhythm Over Magnitude
Cortisol is widely misunderstood as the body’s “stress hormone,” but biologically it functions as a far more fundamental signal:
a master timing molecule that synchronizes metabolism, cognitive readiness, immune function, emotional processing, and circadian physiology.
Its power lies not in its absolute levels, but in the precision of its rhythm.
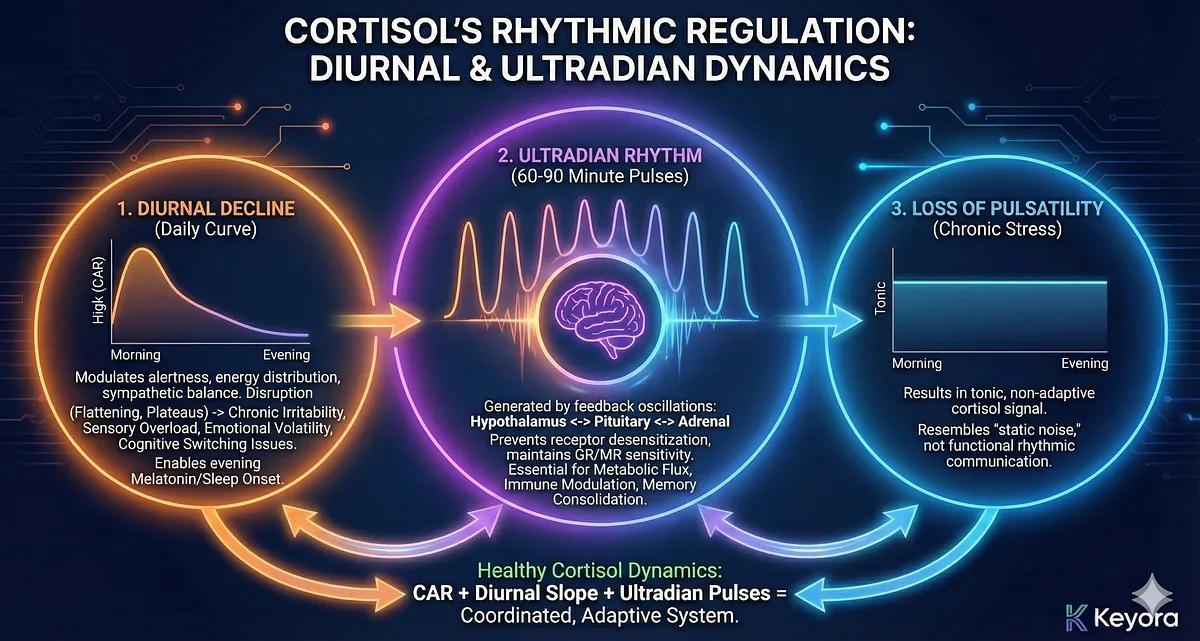
Healthy cortisol dynamics follow a tightly regulated temporal architecture composed of three interacting patterns:
the cortisol awakening response (CAR), the diurnal decline, and the ultradian pulse rhythm.
Together, these patterns create the internal chronobiological structure that allows the body to operate with coherence and stability.
The Cortisol Awakening Response (CAR) represents the most significant single spike of cortisol within the 24-hour cycle.
Occurring within the first 30–60 minutes after waking, CAR acts as a neural and metabolic “boot-up sequence.”
It rapidly increases glucose availability, activates prefrontal attentional networks, supports hippocampal memory encoding, primes the immune system for daytime exposure, and enhances mitochondrial ATP output.
A strong CAR correlates with cognitive sharpness, emotional stability, and resilience, while a blunted CAR signals early-stage HPA dysregulation and predicts fatigue, impaired motivation, low mood, and reduced stress tolerance.
CAR is not triggered by stress, but by the hypothalamus in anticipation of wakefulness – making it one of the body’s core circadian anchors.
Following CAR, cortisol enters its diurnal decline, a gradual reduction in concentration across the day.
This decline is essential for modulating alertness, maintaining stable energy distribution, preventing sympathetic overactivation, and preparing the body for evening parasympathetic dominance.
Disruption of the diurnal slope – especially flattening or afternoon plateaus – leads to chronic irritability, sensory overload, emotional volatility, and difficulty with cognitive switching.
A healthy decline also ensures low cortisol levels in the evening, enabling melatonin release, sleep onset, and deep slow-wave sleep.
Embedded within the daily curve is the ultradian rhythm, a series of 60–90 minute pulses generated by feedback oscillations between the hypothalamus, pituitary, and adrenal cortex.
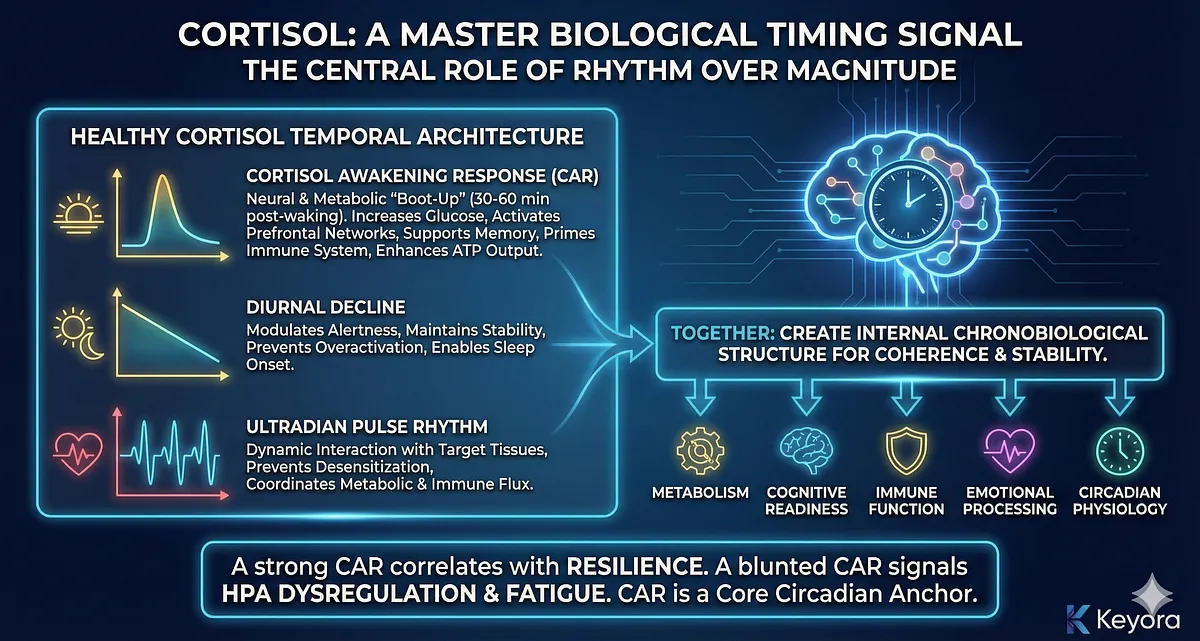
These pulses ensure that cortisol interacts dynamically with target tissues, preventing receptor desensitization and maintaining GR/MR sensitivity.
Ultradian pulsatility is often overlooked, yet it is essential for coordination of metabolic flux, immune modulation, and memory consolidation.
Loss of pulsatility – common in chronic stress – results in a tonic, non-adaptive cortisol signal that resembles static noise rather than functional rhythmic communication.
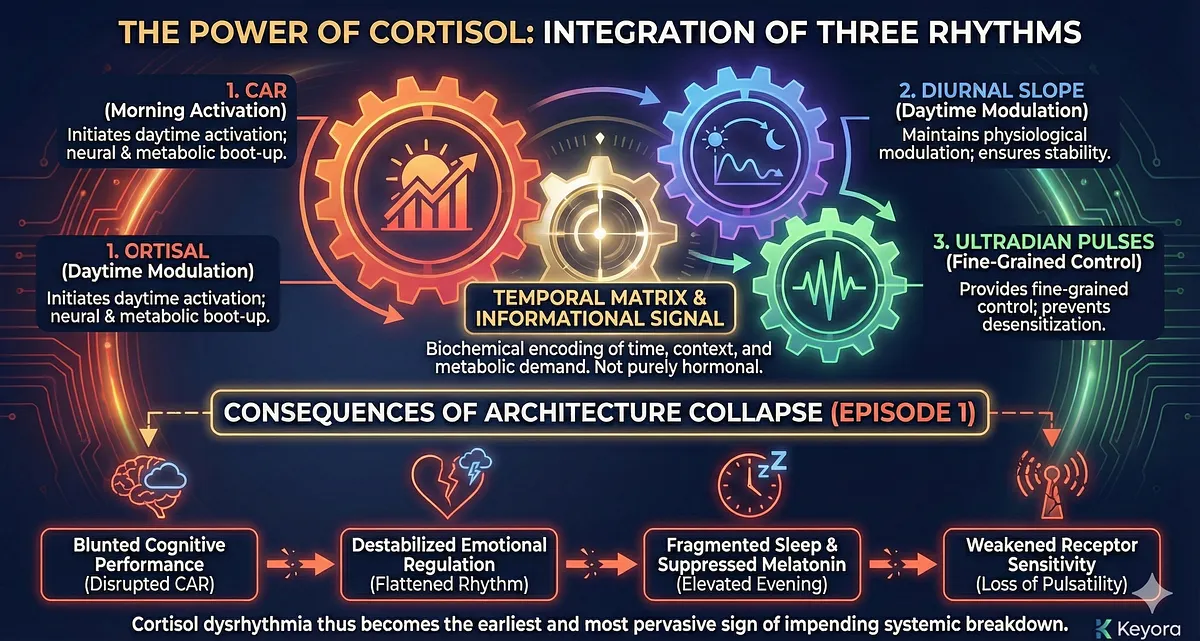
The power of cortisol lies in the integration of these three rhythms. CAR initiates daytime activation; the diurnal slope maintains physiological modulation across the day; ultradian pulses provide fine-grained control.
Together they create a temporal matrix that synchronizes multiple biological systems. In this sense, cortisol is not purely hormonal but informational – a biochemical encoding of time, context, and metabolic demand.
When this temporal architecture collapses – as described in Episode 1 – the consequences radiate through all major systems.
A disrupted CAR blunts cognitive performance; a flattened diurnal rhythm destabilizes emotional regulation; elevated evening cortisol suppresses melatonin and fragments sleep; loss of pulsatility weakens receptor sensitivity and distorts feedback control.
Cortisol dysrhythmia thus becomes the earliest and most pervasive sign of impending systemic breakdown.
Understanding cortisol as a timing signal reframes how we interpret stress-related symptoms and why interventions must restore rhythm rather than simply reduce cortisol levels.
It also sets the stage for the later sections of Episode 2, which will show how chronic stress dismantles this rhythmic architecture – and how Ashwagandha’s multi-level regulatory effects help re-establish biological coherence.
– Cortisol is a master timing signal, not merely a stress hormone.
– Key rhythmic components:
– CAR: morning activation spike → cognition, metabolism, resilience.
– Diurnal decline: regulates alertness, emotional stability, sleep initiation.
– Ultradian pulses: maintain GR/MR sensitivity and metabolic coordination.
– Rhythm > magnitude: the temporal pattern creates biological coherence.
– Dysrhythmia effects:
– Blunted CAR → fatigue, low mood, impaired motivation.
– Flattened diurnal slope → irritability, sensory overload.
– Evening cortisol elevation → melatonin suppression, sleep disruption.
– Loss of pulsatility → receptor desensitization and impaired feedback.
– Cortisol timing breakdown = early warning sign of systemic collapse.
– Sets the basis for understanding HPA breakdown and Ashwagandha regulation.

Section III
Acute vs. Chronic HPA Activation:
How Adaptive Stress Turns Into Biological Exhaustion
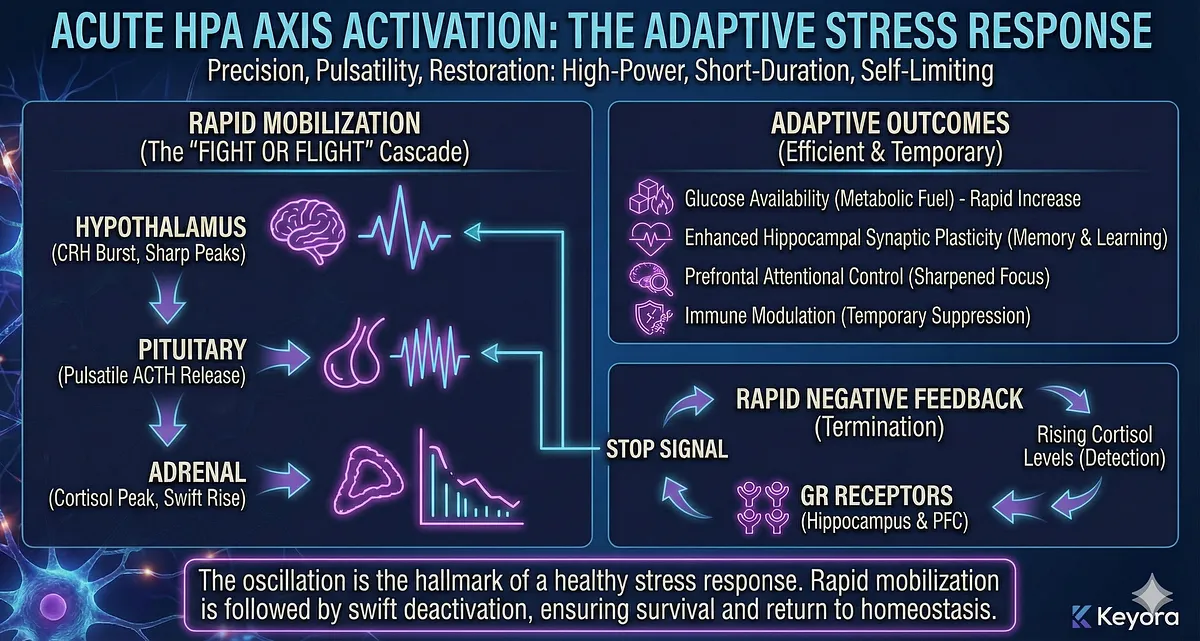
The HPA axis was not designed for chronic activation. Its architecture evolved to manage acute, time-limited challenges – a predator, an infection, a physical threat – conditions in which rapid mobilization of resources followed by swift deactivation ensured survival.
In the acute mode, the CRH→ACTH→cortisol cascade functions with extraordinary efficiency: cortisol rises quickly, provides metabolic fuel, sharpens attention, modulates immunity, and then returns to baseline as soon as the threat dissipates. This oscillation is the hallmark of a healthy stress response.
The Adaptive Acute Response: Precision, Pulsatility, Restoration
During acute activation, hypothalamic CRH neurons fire in sharp bursts, inducing pulsatile ACTH release from the pituitary and generating a well-defined cortisol peak.
Cortisol rapidly increases glucose availability, enhances synaptic plasticity in the hippocampus, facilitates prefrontal attentional control, and temporarily suppresses excessive immune activity.
Crucially, the response is brief. Once GR receptors in the hippocampus and prefrontal cortex detect rising cortisol levels, they initiate negative feedback, terminating the stress cascade. This rapid shutdown is essential – it prevents overexposure and allows the body to return to homeostasis.
Acute HPA activation is therefore high-power, short-duration, adaptive, and self-limiting.
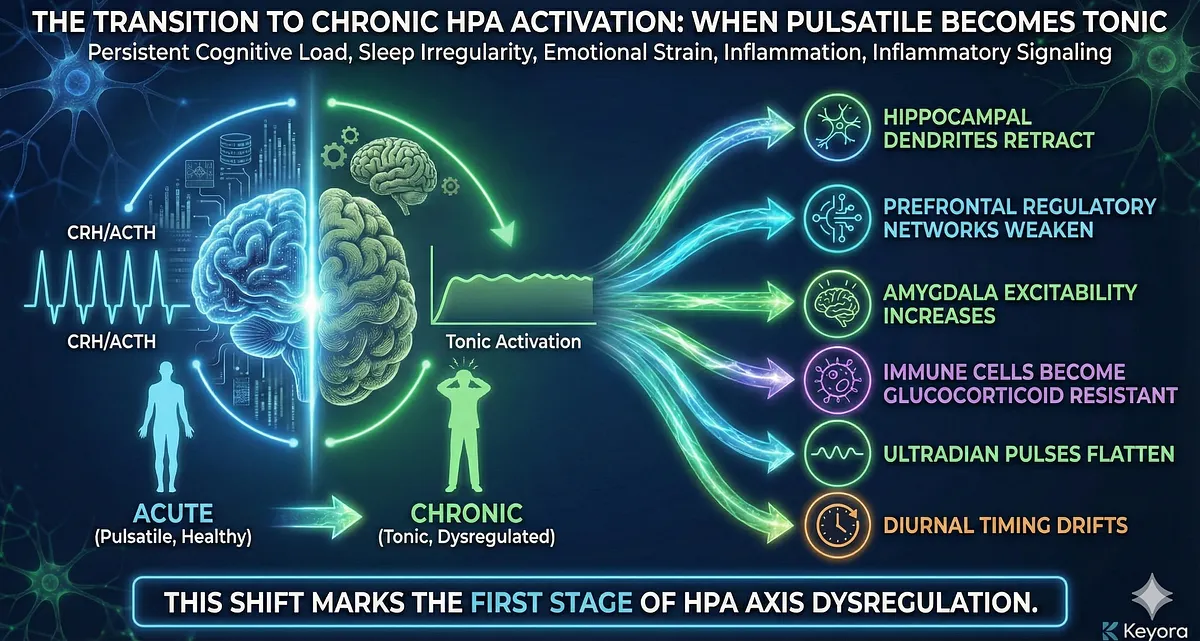
The Transition to Chronic Activation: When Pulsatile Becomes Tonic
Chronic psychological stress, emotional strain, sleep irregularity, persistent cognitive load, and inflammatory signaling fundamentally alter HPA dynamics.
Instead of short-lived pulses, CRH neurons begin firing more frequently, and ACTH release becomes less oscillatory. The system shifts from pulsatile activation to tonic activation – a continuous background elevation of stress drive.
The consequences are profound.
Tonic CRH signaling increases baseline ACTH secretion, leading to mistimed and prolonged cortisol exposure. Cortisol’s beneficial acute effects now become damaging:
- hippocampal dendrites retract
- prefrontal regulatory networks weaken
- amygdala excitability increases
- immune cells become glucocorticoid resistant
- ultradian pulses flatten
- diurnal timing drifts
This shift marks the first stage of HPA axis dysregulation.
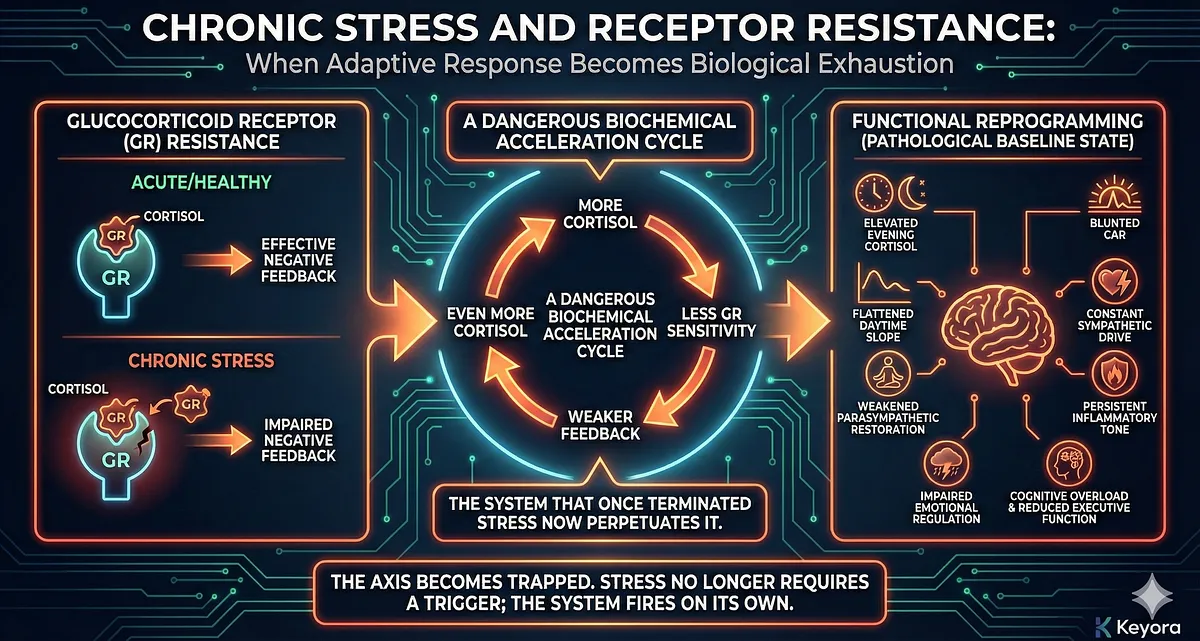
Chronic Stress and Receptor Resistance
One of the earliest molecular signs of chronic HPA activation is glucocorticoid receptor resistance (GR resistance).
When cortisol remains elevated for too long or too often, GRs become less sensitive, particularly in the hippocampus, prefrontal cortex, and immune cells.
GR resistance impairs negative feedback, preventing cortisol from properly shutting down its own production.
This creates a dangerous loop:
- More cortisol → less GR sensitivity → weaker feedback → even more cortisol.
The axis becomes trapped in a biochemical acceleration cycle.
The system that once terminated stress now perpetuates it.
Chronic Activation Converts Stress From a Response Into a Condition
As GR resistance grows and CRH neurons remain overactive, the HPA axis undergoes functional reprogramming.
Stress no longer requires a trigger – the system fires on its own. The body enters a state of continuous vigilance marked by:
- elevated evening cortisol
- blunted CAR
- flattened daytime slope
- constant sympathetic drive
- weakened parasympathetic restoration
- persistent inflammatory tone
- impaired emotional regulation
- cognitive overload and reduced executive function
What was once an adaptive response becomes a pathological baseline state.
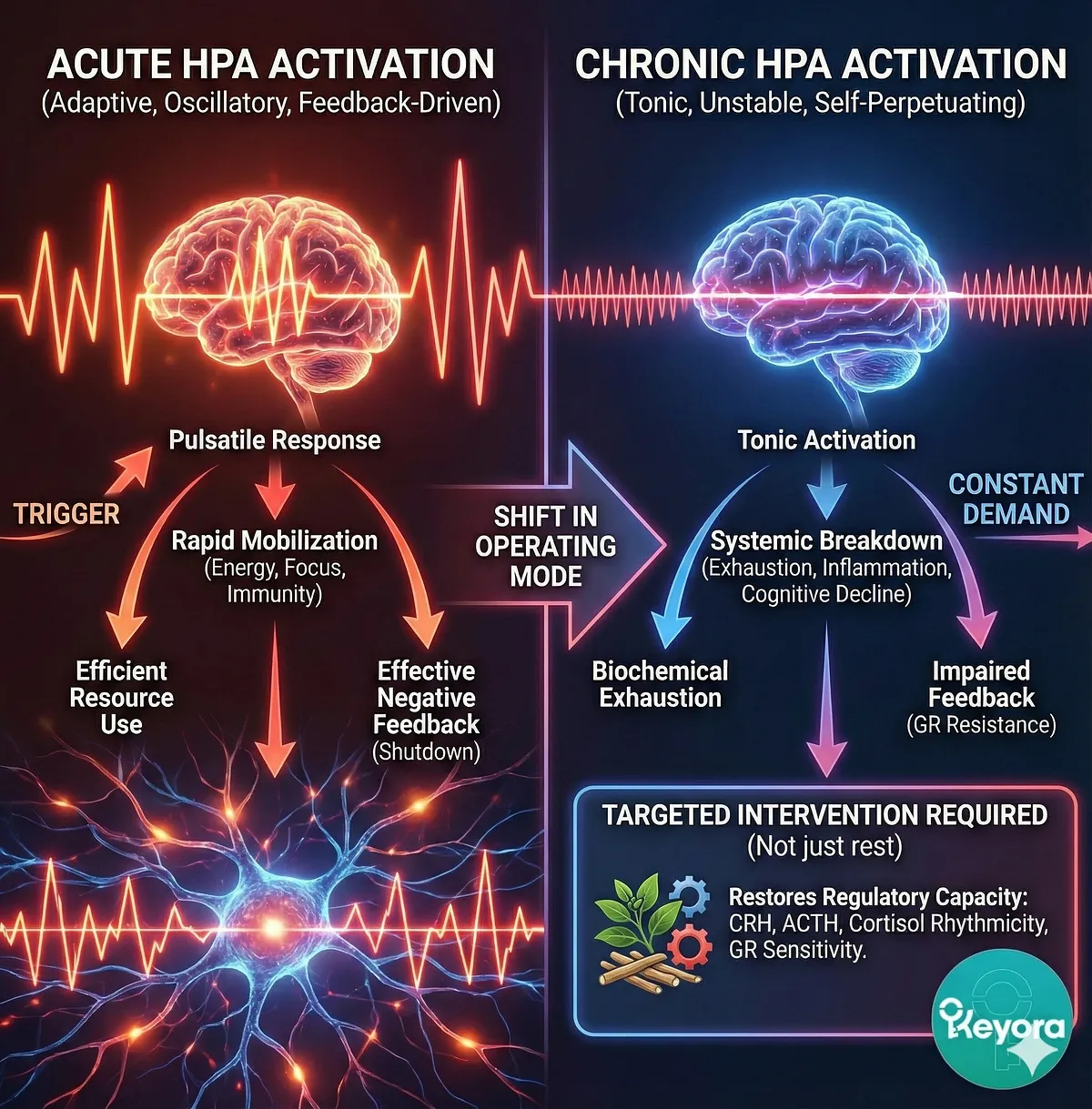
The Biological Meaning of Chronic HPA Activation
Chronic stress is often described as “being stressed for too long,” but the biological reality is far more severe.
Chronic HPA activation represents a shift in operating mode, from oscillatory, adaptive, and feedback-driven to tonic, unstable, and self-perpetuating.
Once this shift occurs, the system requires targeted intervention – not simply “rest” or lifestyle change – to restore regulatory capacity.
This is why understanding the distinction between acute and chronic activation is essential.
It explains how stress transitions from temporary pressure to systemic breakdown, and why Ashwagandha’s multi-level modulation of CRH, ACTH, cortisol rhythmicity, and GR sensitivity is uniquely positioned to counteract these dynamics – topics that will be explored in later sections.
– HPA axis evolved for acute, short-term stress → rapid CRH/ACTH/cortisol pulses and rapid shutdown.
– Acute activation is adaptive: enhances cognition, metabolism, immunity, then returns to baseline.
– Chronic stress converts pulsatile activation into tonic activation.
– Continuous CRH/ACTH drive → prolonged cortisol exposure → hippocampal and PFC damage → amygdala hyperactivation.
– Chronic activation leads to glucocorticoid receptor resistance (GR resistance), impairing negative feedback.
– GR resistance creates a self-reinforcing loop: more cortisol → weaker feedback → more cortisol.
– Chronic HPA activation becomes a persistent biological condition, not a psychological reaction.
– Sets the basis for understanding HPA collapse and the need for multi-target interventions like Ashwagandha.
Section IV
HPA Axis Breakdown Under Chronic Stress:
From Adaptive Oscillation to Systemic Collapse
Chronic stress does not merely activate the HPA axis – it fundamentally rewires it. What begins as an adaptive, oscillatory, feedback-controlled system gradually degrades into a dysregulated, desynchronized, and self-perpetuating state.
This breakdown occurs through multiple, mutually reinforcing mechanisms across the hypothalamus, pituitary, adrenal cortex, and their feedback targets in the hippocampus and prefrontal cortex. The result is not “too much cortisol,” but a collapse of cortisol rhythmicity, receptor signaling, and neuroendocrine coordination.
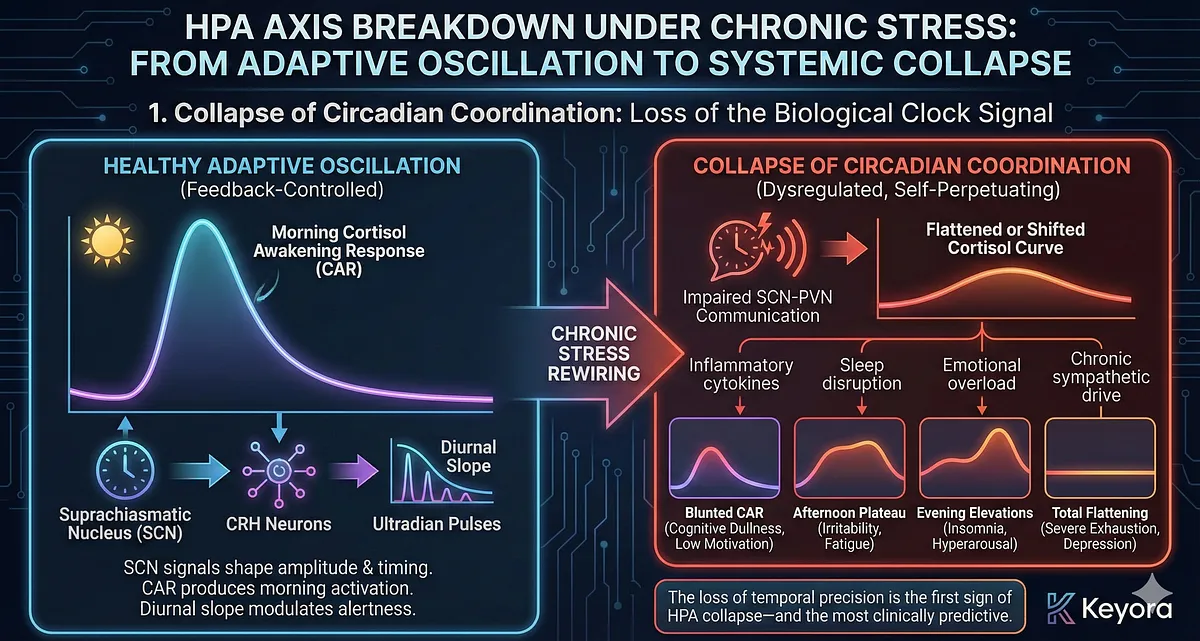
1. Collapse of Circadian Coordination: Loss of the Biological Clock Signal
Under chronic stress, the coupling between the suprachiasmatic nucleus (SCN) and CRH neurons becomes impaired. SCN signals normally shape the amplitude and timing of CRH release, producing the morning cortisol awakening response (CAR) and orchestrating the diurnal slope.
When inflammatory cytokines, sleep disruption, emotional overload, or chronic sympathetic drive disrupt SCN–PVN communication, the cortisol curve begins to flatten or shift.
Common dysfunction patterns include:
- Blunted CAR → cognitive dullness, low motivation, impaired activation
- Afternoon plateau → irritability, sensory overload, fatigue
- Evening cortisol elevations → insomnia, hyperarousal
- Total flattening → severe exhaustion, depression-like states
The loss of temporal precision is the first sign of HPA collapse – and the most clinically predictive.
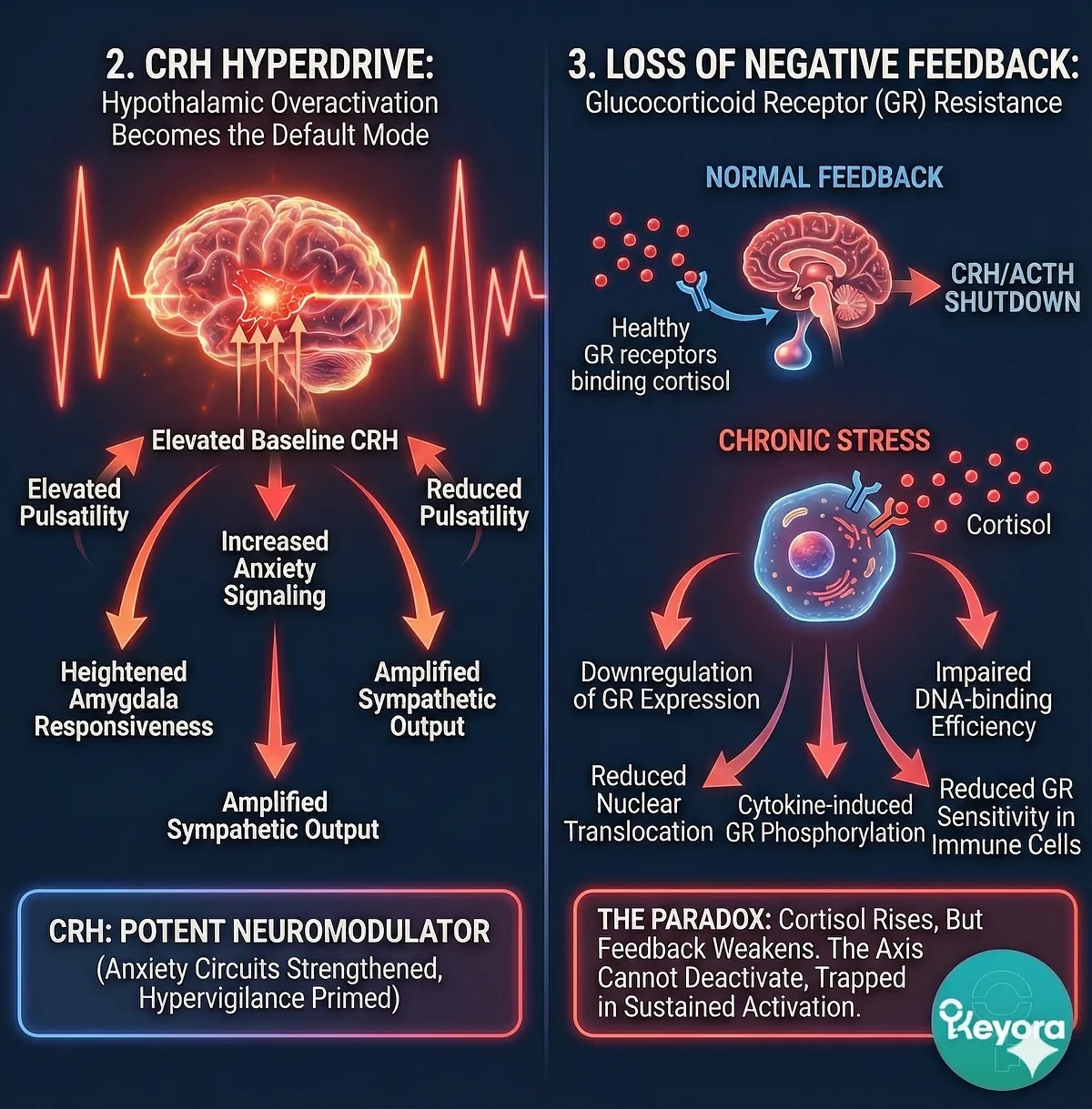
2. CRH Hyperdrive: Hypothalamic Overactivation Becomes the Default Mode
Chronic psychological stress forces CRH neurons into a sustained firing state. Instead of short bursts, they begin operating in a near-tonic mode. This leads to:
- Elevated baseline CRH
- Reduced pulsatility
- Increased anxiety signaling
- Heightened amygdala responsiveness
- Amplified sympathetic output
CRH is not merely a hormone – it is also a potent neuromodulator. Chronic overactivation strengthens anxiety circuits, suppresses appetitive pathways, and primes the brain for hypervigilance. CRH hyperdrive is one of the core engines of downstream HPA dysfunction.
3. Loss of Negative Feedback: Glucocorticoid Receptor (GR) Resistance
Under normal conditions, rising cortisol binds to GRs in the hippocampus, prefrontal cortex, and pituitary gland, shutting down CRH and ACTH release. Chronic stress disrupts this mechanism.
Persistent cortisol elevation leads to:
- Downregulation of GR expression
- Reduced nuclear translocation
- Impaired DNA-binding efficiency
- Cytokine-induced GR phosphorylation (blocks function)
- Reduced GR sensitivity in immune cells
This GR resistance produces a paradox: cortisol rises, but feedback weakens. Without functional GR-mediated feedback, the axis cannot deactivate properly, trapping the system in a state of sustained activation.
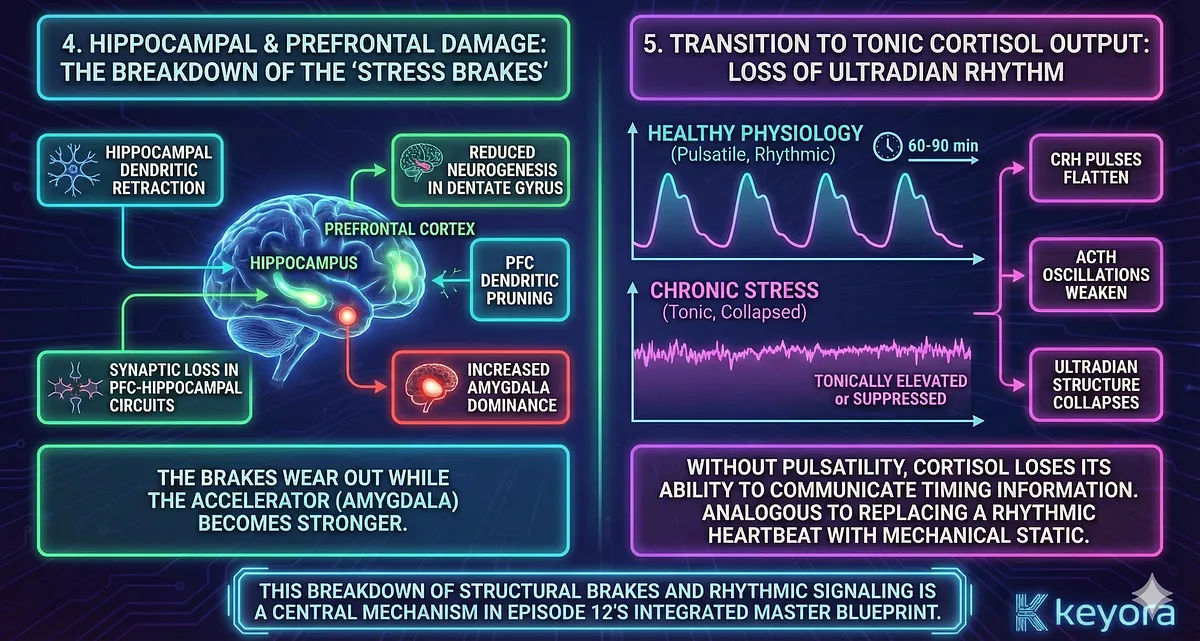
4. Hippocampal and Prefrontal Damage: The Breakdown of the “Stress Brakes”
The hippocampus and prefrontal cortex serve as inhibitory regulators of the HPA axis. They evaluate threat, contextualize stress, and apply negative feedback. Chronic stress causes:
- Hippocampal dendritic retraction
- Reduced neurogenesis in the dentate gyrus
- PFC dendritic pruning
- Synaptic loss in PFC–hippocampal circuits
- Increased amygdala dominance
These structural changes reduce the brain’s ability to shut down stress responses.
The brakes wear out while the accelerator (amygdala) becomes stronger.
5. Transition to Tonic Cortisol Output: Loss of Ultradian Rhythm
In healthy physiology, cortisol pulses with ultradian rhythm every 60–90 minutes. These pulses prevent receptor fatigue and maintain metabolic flexibility. Under chronic stress:
- CRH pulses flatten
- ACTH oscillations weaken
- Cortisol becomes tonically elevated or tonically suppressed
- Ultradian structure collapses
Without pulsatility, cortisol loses its ability to communicate timing information. This is analogous to replacing a rhythmic heartbeat with mechanical static.
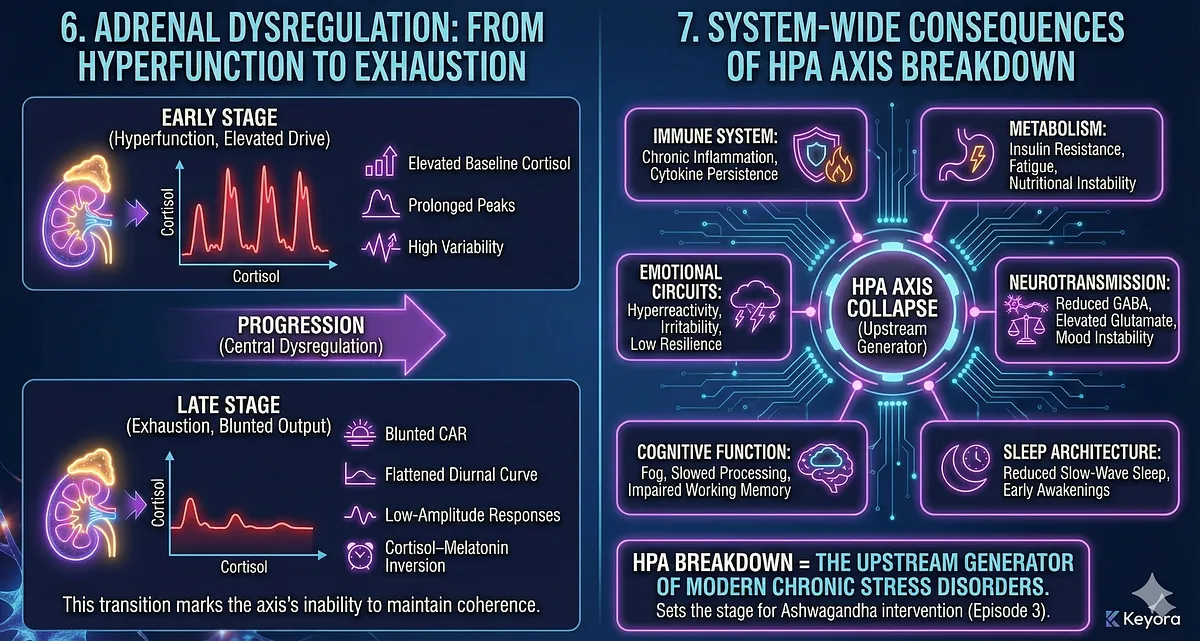
6. Adrenal Dysregulation: From Hyperfunction to Exhaustion
Prolonged ACTH exposure overdrives the adrenal cortex. Early-stage chronic stress produces exaggerated cortisol peaks; later stages produce adrenal blunting or inadequate output. This progression is often misinterpreted as “adrenal fatigue,” but the true mechanism is central dysregulation, not peripheral gland failure.
Early stage:
- Elevated baseline cortisol
- Prolonged peaks
- High variability
Late stage:
- Blunted CAR
- Flattened diurnal curve
- Low-amplitude responses
- Cortisol–melatonin inversion
This transition marks the axis’s inability to maintain coherence.
7. System-Wide Consequences of HPA Axis Breakdown
When the HPA axis collapses, systemic effects propagate through:
- Immune system → chronic inflammation, cytokine persistence
- Metabolism → insulin resistance, fatigue, nutritional instability
- Neurotransmission → reduced GABA, elevated glutamate, mood instability
- Sleep architecture → reduced slow-wave sleep, early awakenings
- Cognitive function → fog, slowed processing, impaired working memory
- Emotional circuits → hyperreactivity, irritability, low resilience
In other words:
HPA breakdown = the upstream generator of modern chronic stress disorders.
This sets the foundation for Section V and VI, where cross-talk with immune and limbic systems will be demonstrated, and prepares the stage for Episode 3, which will dissect how Ashwagandha intervenes at multiple levels of this breakdown.
– Chronic stress disrupts SCN–HPA coordination → flattened, shifted, or inverted cortisol rhythms.
– CRH neurons become hyperactive → tonic stress signaling replaces pulsatile activation.
– Persistent cortisol elevation causes GR resistance → impaired negative feedback → runaway HPA activation.
– Hippocampus and PFC lose structural integrity → weakened stress “braking” mechanisms.
– Ultradian cortisol pulses collapse → cortisol loses its temporal signaling function.
– Adrenal output becomes dysregulated (early hyperfunction → late-stage blunting).
– System-wide consequences include inflammation, metabolic dysregulation, mood reactivity, sleep disruption, and cognitive impairment.
– HPA axis breakdown is the upstream source of multi-system stress disorders.
Section V
HPA ↔ Immune System Cross-Talk:
Cytokine Signaling and the Collapse of Endocrine–Immune Balance
The HPA axis and the immune system form one of the body’s most tightly integrated regulatory loops.
In healthy physiology, cortisol and glucocorticoid receptors (GRs) maintain immune homeostasis by suppressing excessive inflammation while allowing appropriate defense responses.
Conversely, the immune system communicates threat, infection, or metabolic strain to the brain through cytokine signals that modulate CRH and ACTH secretion.
This bidirectional communication enables the organism to distinguish between safety and threat and to allocate metabolic resources accordingly.
Chronic stress disrupts this communication, transforming a finely tuned feedback loop into a self-amplifying cycle of inflammation, cortisol dysregulation, and immune dysfunction.
Understanding this cross-talk is essential for explaining why stress-related conditions – fatigue, depression, anxiety, insomnia, autoimmune flares – share a common inflammatory signature.
1. Cortisol as an Immune Regulator: The Anti-Inflammatory Baseline
Under normal conditions, cortisol provides broad anti-inflammatory control.
Through GR activation, cortisol downregulates:
- NF-κB inflammatory transcription
- IL-1β, IL-6, TNF-α production
- COX-2 and prostaglandin pathways
- mast cell activation
- microglial overexcitability
This is not suppression but optimization.
Cortisol ensures inflammation is proportionate, targeted, and time-limited.
When cortisol rhythms are intact – strong CAR, steep daytime decline, ultradian pulses – the immune system remains responsive but not overreactive.
Healthy cortisol = immune precision.
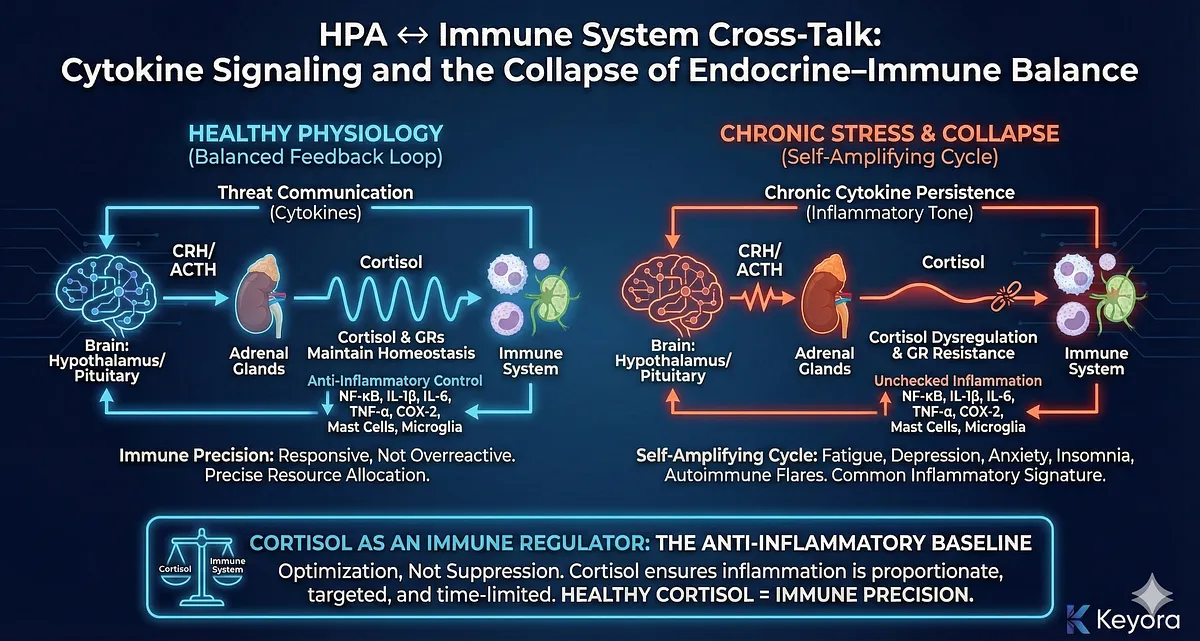
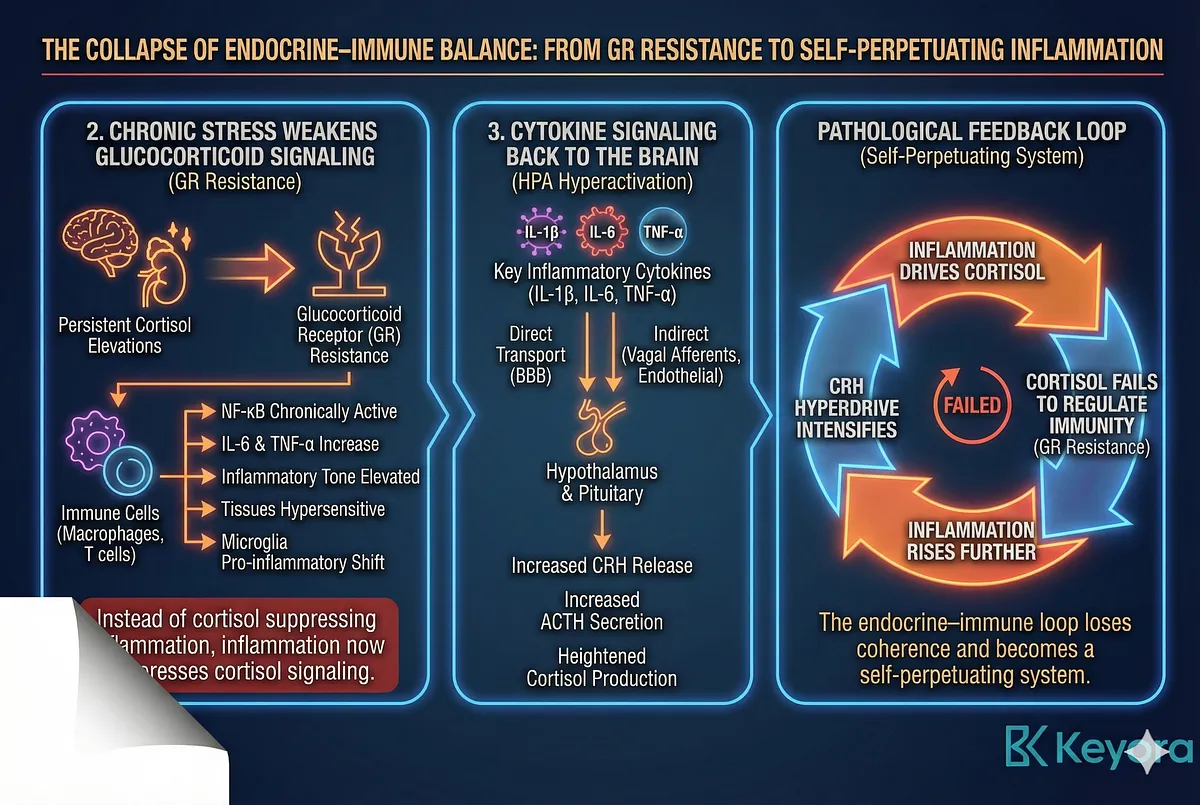
2. Chronic Stress Weakens Glucocorticoid Signaling: The Onset of GR Resistance
Persistent cortisol elevations produce glucocorticoid receptor resistance (GR resistance) – the same mechanism described in Section IV.
In the immune system, GR resistance is catastrophic.
Immune cells (especially macrophages, dendritic cells, and T cells) become less responsive to cortisol’s regulatory signals.
As a result:
- NF-κB becomes chronically active
- IL-6 and TNF-α increase
- inflammatory tone remains elevated
- tissues become hypersensitive
- microglia shift toward a pro-inflammatory phenotype
This flips the normal relationship:
Instead of cortisol suppressing inflammation, inflammation now suppresses cortisol signaling.
3. Cytokine Signaling Back to the Brain: IL-6, IL-1β, TNF-α Drive HPA Hyperactivation
Cytokines cross-talk with the CNS in two major ways:
- Directly via active transport across the blood–brain barrier
- Indirectly through vagal afferents, endothelial cells, and circumventricular organs
Key inflammatory cytokines (IL-1β, IL-6, TNF-α) signal to the hypothalamus and pituitary, inducing:
- increased CRH release
- increased ACTH secretion
- heightened cortisol production
This immune-to-brain signaling is adaptive during infection but becomes pathological during chronic stress, creating a loop where:
- inflammation drives cortisol
- cortisol fails to regulate immunity (due to GR resistance)
- inflammation rises further
- CRH hyperdrive intensifies
The endocrine–immune loop loses coherence and becomes a self-perpetuating system.
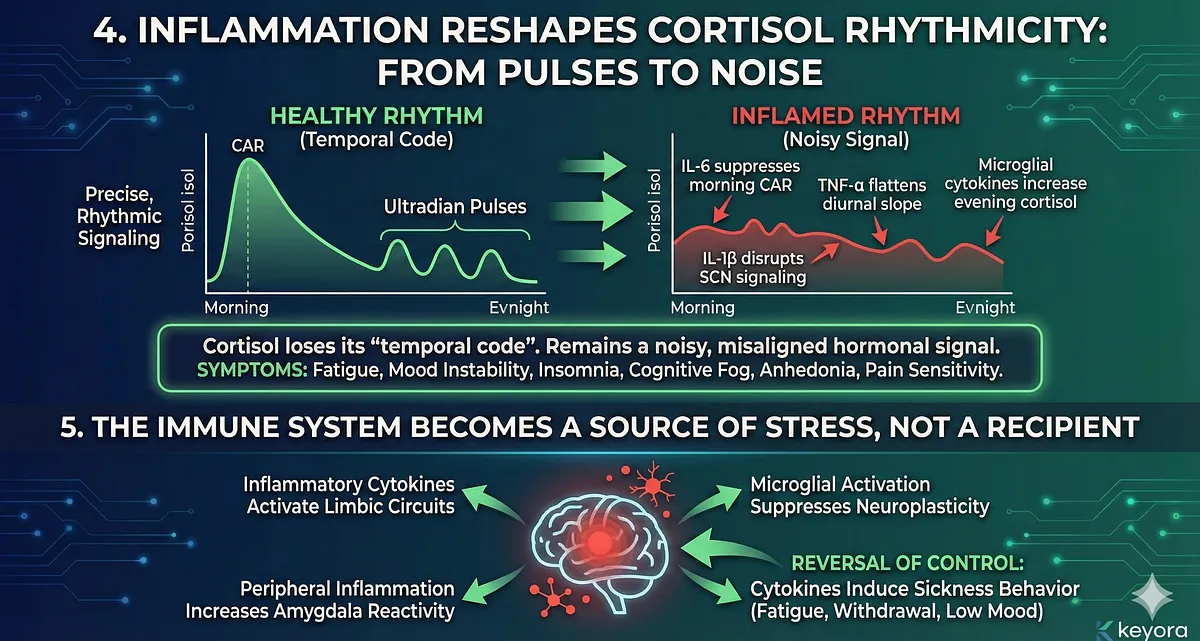
4. Inflammation Reshapes Cortisol Rhythmicity: From Pulses to Noise
Inflammatory cytokines interfere directly with cortisol’s rhythmic architecture:
- IL-6 suppresses morning CAR
- TNF-α flattens diurnal slope
- IL-1β disrupts SCN signaling
- microglial cytokines increase evening cortisol
As rhythmic precision declines, cortisol loses its “temporal code.”
What remains is a noisy, misaligned hormonal signal incapable of maintaining immune order.
This is why chronic inflammation and chronic stress share clinical features:
fatigue, mood instability, insomnia, cognitive fog, anhedonia, pain sensitivity.
5. The Immune System Becomes a Source of Stress, Not a Recipient
A critical shift occurs when immune activity no longer responds to stress but generates stress:
- inflammatory cytokines activate limbic circuits
- microglial activation suppresses neuroplasticity
- peripheral inflammation increases amygdala reactivity
- cytokines induce sickness behavior (fatigue, withdrawal, low mood)
This reverses the normal direction of control.
The immune system becomes the driver of HPA dysregulation and emotional deterioration.
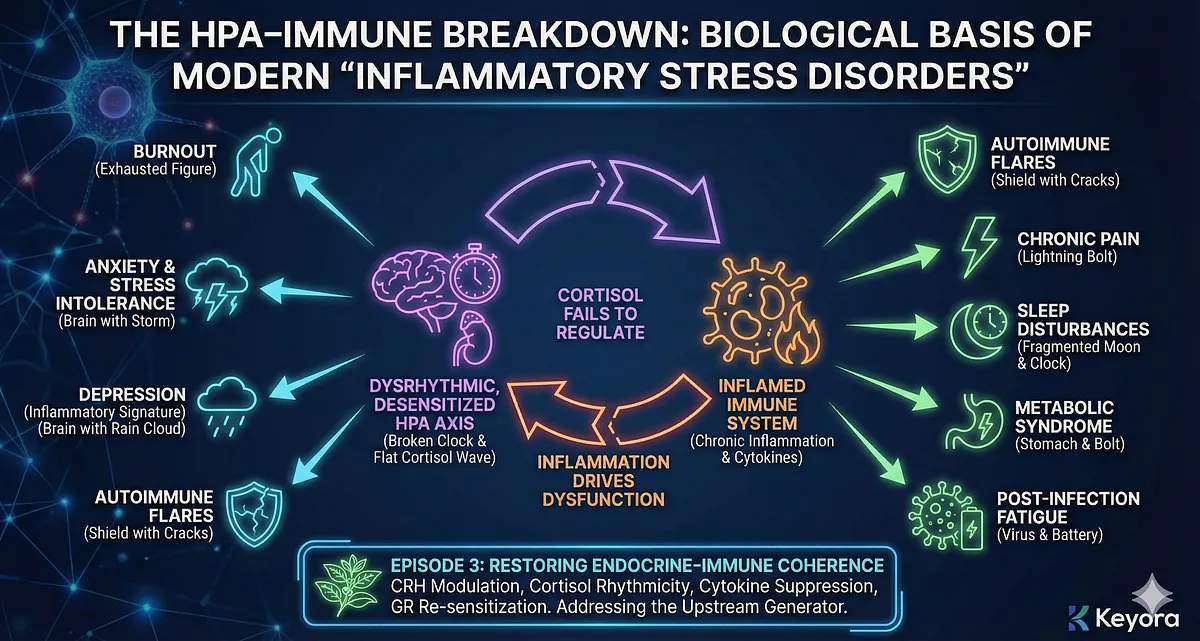
6. The HPA–Immune Breakdown Is the Biological Basis of Modern “Inflammatory Stress Disorders”
The breakdown of endocrine–immune cross-talk explains why diverse conditions share a common pattern:
- burnout
- anxiety and stress intolerance
- depression with inflammatory signature
- autoimmune flares
- chronic pain
- sleep disturbances
- metabolic syndrome
- post-infection fatigue states
Each is shaped by the same core mechanism:
an inflamed immune system that can no longer be regulated by a dysrhythmic, desensitized HPA axis.
This sets the stage for Episode 3, where we examine how Ashwagandha restores endocrine–immune coherence through CRH modulation, cortisol rhythmicity, cytokine suppression, and GR re-sensitization.
– HPA axis and immune system form a tightly integrated bidirectional regulatory loop.
– Healthy cortisol rhythms suppress excessive inflammation via GR activation.
– Chronic stress causes GR resistance → immune cells ignore cortisol → inflammation rises.
– IL-1β, IL-6, TNF-α signal back to the hypothalamus and pituitary → CRH and ACTH increase.
– Cytokines distort cortisol rhythms (CAR suppression, diurnal flattening, evening elevations).
– Inflammation begins driving stress, producing chronic hyperarousal and sickness behavior.
– HPA–immune breakdown is the shared mechanism behind burnout, depression, autoimmune flares, sleep disruption, and metabolic disorders.
Section VI
HPA ↔ Limbic Circuits:
The Amygdala–PFC–Hippocampus Loop Under Chronic Stress
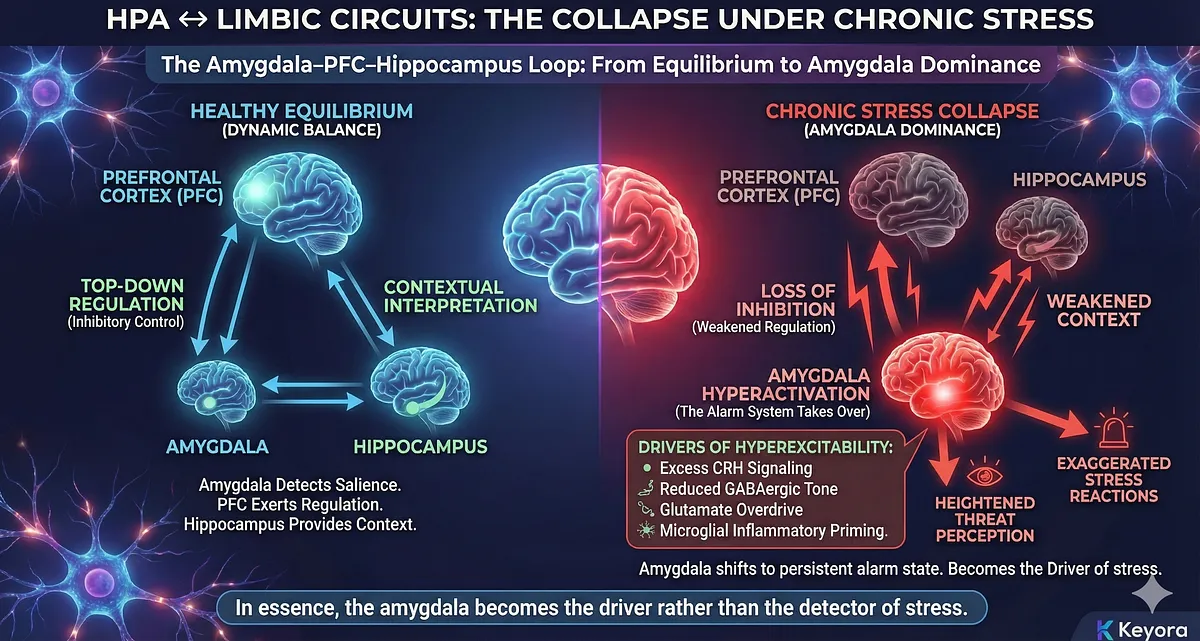
The HPA axis is not an isolated endocrine mechanism – it is tightly embedded within a core emotional–cognitive network composed of the amygdala, prefrontal cortex (PFC), and hippocampus.
These three structures form the regulatory loop that determines how the brain evaluates threat, regulates emotion, contextualizes stress, and applies inhibitory control.
Under healthy conditions, the loop maintains a dynamic equilibrium: the amygdala detects salience, the hippocampus provides memory and contextual interpretation, and the PFC exerts top-down regulation, preventing overreaction.
Chronic stress destabilizes this balance.
HPA hyperactivity, cortisol dysrhythmia, and GR resistance progressively shift control from the PFC and hippocampus toward the amygdala – producing the emotional volatility, irritability, hypervigilance, anxiety sensitivity, and cognitive–emotional instability characteristic of chronic stress states.
Below is how the collapse unfolds.
1. Amygdala Hyperactivation: The Alarm System Takes Over
The amygdala is the brain’s central salience detector. Acute stress activates the amygdala to mobilize vigilance and initiate HPA activation.
But chronic stress drives sustained amygdala hyperexcitability due to:
- Excess CRH signaling directly from the hypothalamus
- Reduced GABAergic inhibitory tone
- Glutamate overdrive and impaired reuptake
- Microglial inflammatory priming
- Loss of top-down inhibition from PFC
With these changes, the amygdala shifts to a persistent alarm state, characterized by:
- heightened threat perception
- exaggerated stress reactions
- emotional reactivity
- increased cortisol output
In essence, the amygdala becomes the driver rather than the detector of stress.
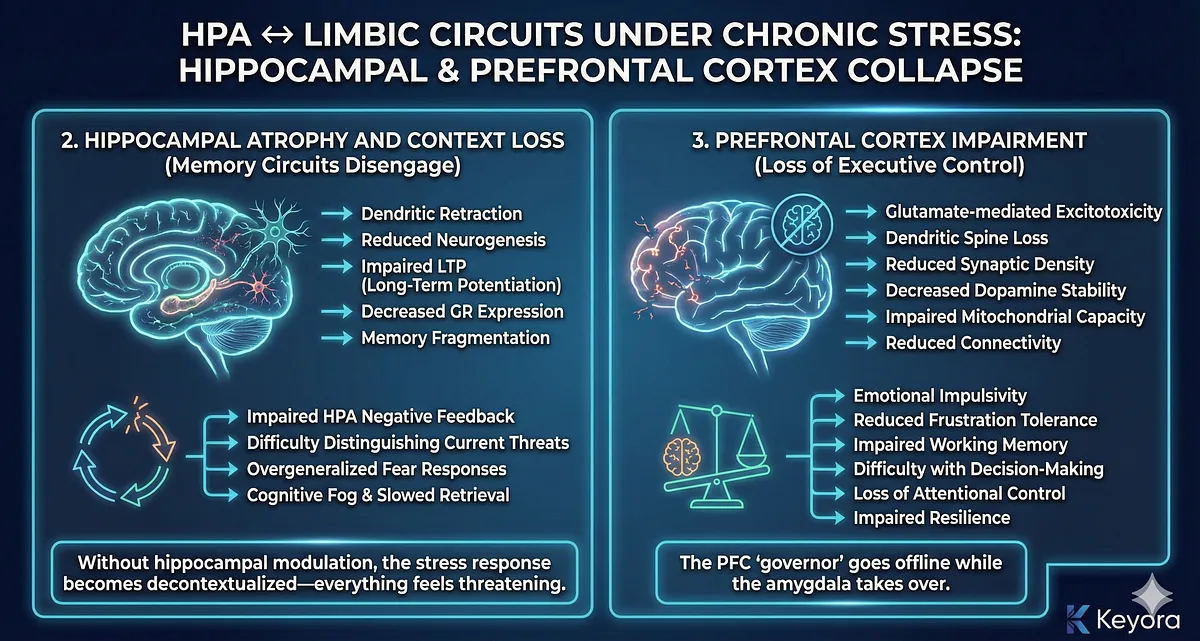
2. Hippocampal Atrophy and Context Loss: Memory Circuits Disengage
The hippocampus contextualizes threat and applies negative feedback on the HPA axis. It is densely packed with glucocorticoid receptors, making it highly sensitive to cortisol.
Under chronic stress, sustained cortisol exposure and inflammatory cytokines induce:
- dendritic retraction
- reduced neurogenesis
- impaired LTP (long-term potentiation)
- decreased GR expression
- memory fragmentation
Functionally, this produces:
- impaired HPA negative feedback
- difficulty distinguishing current threats from past memories
- overgeneralized fear responses
- cognitive fog and slowed retrieval
Without hippocampal modulation, the stress response becomes decontextualized—everything feels threatening.
3. Prefrontal Cortex Impairment: Loss of Executive Control
The PFC serves as the executive regulator of the limbic system. It applies top-down control, dampens amygdala output, and modulates emotional interpretation.
Chronic stress damages the PFC through:
- glutamate-mediated excitotoxicity
- dendritic spine loss
- reduced synaptic density
- decreased dopamine stability
- impaired mitochondrial capacity
- reduced connectivity with hippocampus and amygdala
Clinically, this manifests as:
- emotional impulsivity
- reduced frustration tolerance
- impaired working memory
- difficulty with decision-making
- loss of attentional control
- impaired resilience
The PFC “governor” goes offline while the amygdala takes over.
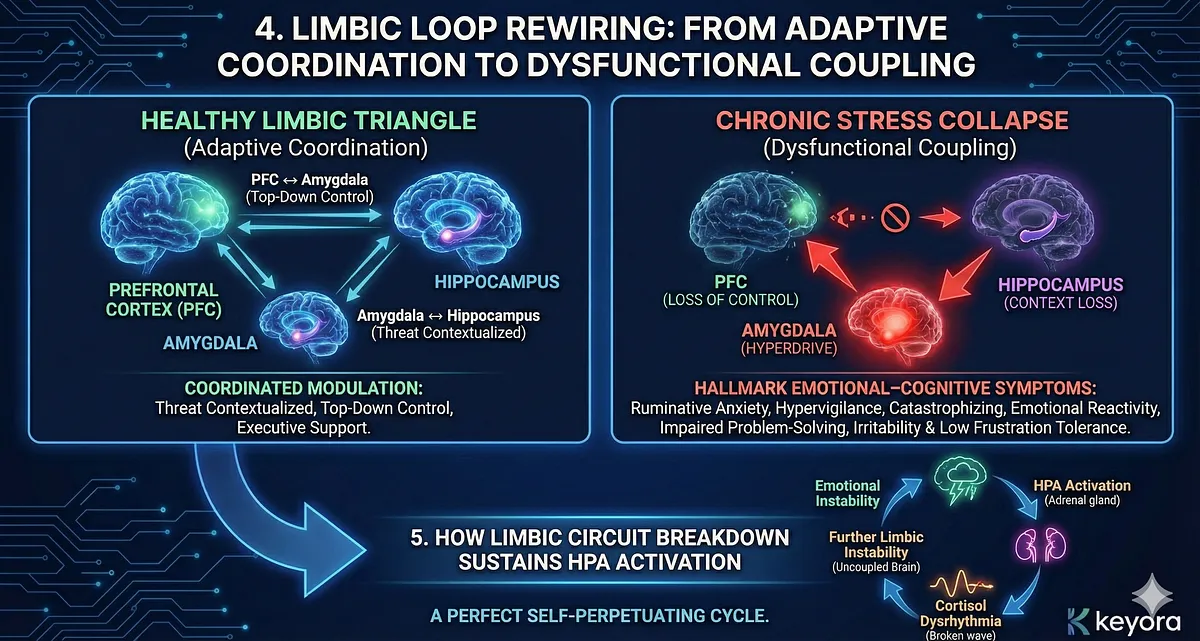
4. Limbic Loop Rewiring: From Adaptive Coordination to Dysfunctional Coupling
In healthy conditions, the amygdala, hippocampus, and PFC modulate each other in a coordinated triangle:
- Amygdala ↔ Hippocampus: threat detection contextualized by memory
- PFC ↔ Amygdala: top-down control over emotional intensity
- PFC ↔ Hippocampus: executive support for memory and planning
Chronic stress reshapes this triangle into a dysfunctional pattern:
- Amygdala dominates (hyperdrive)
- Hippocampus disconnects (context loss)
- PFC weakens (loss of control)
This produces the hallmark emotional–cognitive symptoms of chronic stress:
- ruminative anxiety
- hypervigilance
- catastrophizing
- emotional reactivity
- impaired problem-solving
- irritability and low frustration tolerance
The limbic system essentially becomes “uncoupled,” allowing the stress system to operate without regulatory constraints.
5. How Limbic Circuit Breakdown Sustains HPA Activation
As limbic stability declines, HPA output becomes increasingly driven by emotional circuits instead of external threats.
Mechanisms include:
- Amygdala sends amplified excitatory projections to the hypothalamus
- Hippocampal inhibition weakens (loss of GR-mediated feedback)
- PFC loses the capacity to apply rational override
- Limbic circuits generate spontaneous activation even without stressors
This creates a loop where:
emotional instability → HPA activation → cortisol dysrhythmia → further limbic instability
A perfect self-perpetuating cycle.
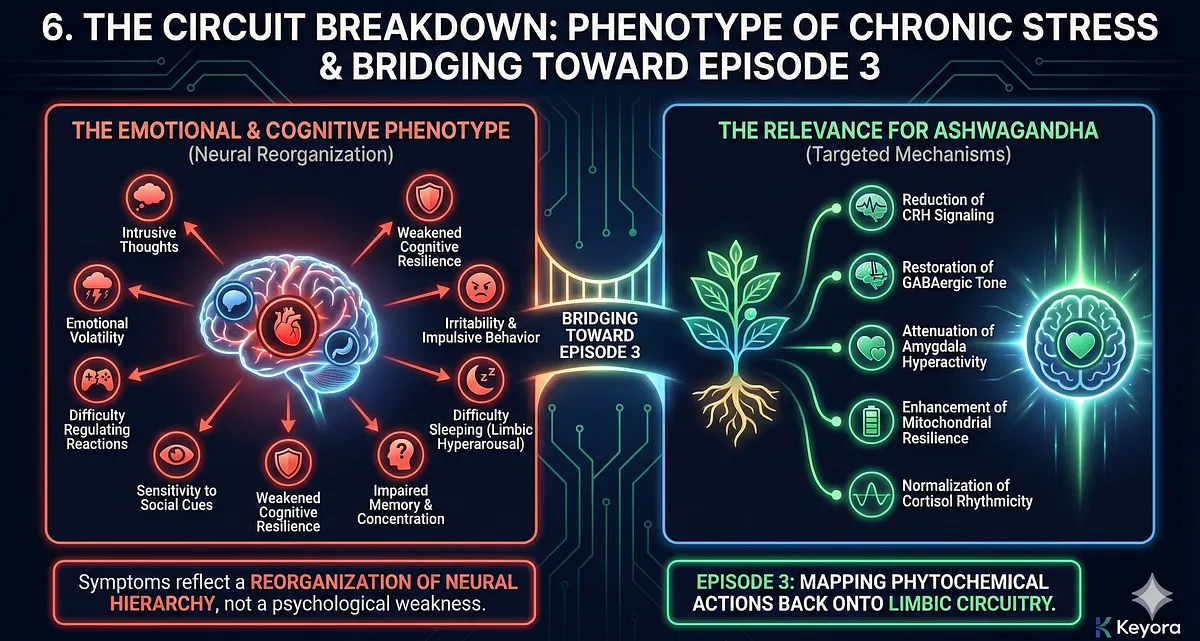
6. This Circuit Breakdown Explains the Emotional & Cognitive Phenotype of Chronic Stress
The limbic collapse model explains why individuals under chronic stress experience:
- intrusive thoughts
- emotional volatility
- difficulty regulating reactions
- sensitivity to social cues
- weakened cognitive resilience
- irritability and impulsive behavior
- difficulty sleeping due to limbic hyperarousal
- impaired memory and concentration
These symptoms reflect a reorganization of neural hierarchy, not a psychological weakness.
7. Bridging Toward Episode 3: The Relevance for Ashwagandha
Ashwagandha’s known effects – reduction of CRH signaling, restoration of GABAergic tone, attenuation of amygdala hyperactivity, enhancement of mitochondrial resilience, and normalization of cortisol rhythmicity – are directly aligned with the mechanisms described here.
Episode 3 will map these phytochemical actions back onto the limbic circuitry presented in this section.
– Limbic system (amygdala, PFC, hippocampus) forms the regulatory loop controlling emotional and stress responses.
– Chronic stress hyperactivates the amygdala → persistent alarm state and amplified HPA output.
– Cortisol and cytokines damage the hippocampus → reduced memory, context loss, weakened negative feedback.
– Chronic stress impairs the PFC → loss of executive control, emotional instability, poor decision-making.
– Limbic triangle becomes uncoupled: amygdala dominates, PFC weakens, hippocampus disconnects.
– Result = emotional reactivity, hypervigilance, anxiety, irritability, cognitive fog.
– Limbic dysfunction drives ongoing HPA activation, creating a self-perpetuating stress loop.
Section VII
HPA ↔ Mitochondrial Energy Systems:
Why Energy Failure Sustains Stress
The HPA axis is fundamentally an energy-allocation system.
Every stress response – cognitive, emotional, or immunological – requires ATP, mitochondrial resilience, and metabolic flexibility.
Chronic stress, however, degrades mitochondrial function through glucocorticoid excess, inflammatory cytokines, and sympathetic overdrive.
As mitochondria fail, the brain and body lose their capacity to regulate stress, creating a self-reinforcing loop where energy deficiency becomes both a consequence and a driver of HPA dysregulation.
This mitochondrial–HPA coupling explains the “wired-but-tired” paradox, the hallmark of modern burnout:
high arousal, low capacity; high vigilance, low energy; high cortisol, low resilience.
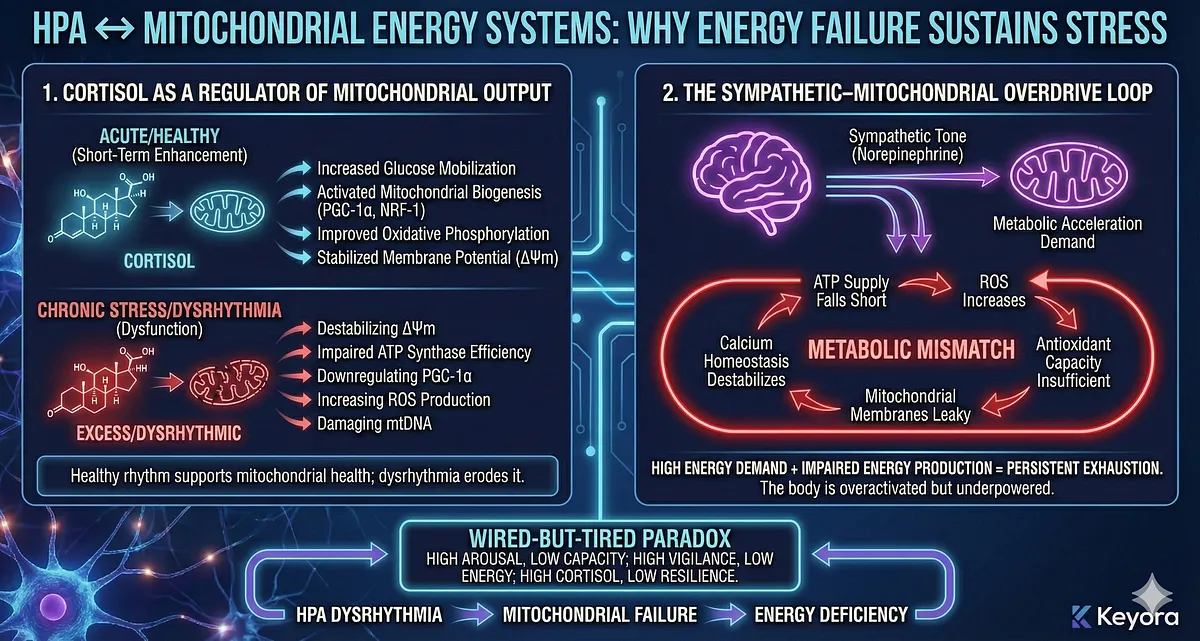
1. Cortisol as a Regulator of Mitochondrial Output
Cortisol acutely enhances mitochondrial performance by:
- increasing glucose mobilization
- activating mitochondrial biogenesis pathways
- upregulating PGC-1α and NRF-1
- improving oxidative phosphorylation efficiency
- stabilizing mitochondrial membrane potential (ΔΨm)
This short-term enhancement supports fight-or-flight responses.
But chronic cortisol exposure flips these benefits into dysfunction by:
- destabilizing ΔΨm
- impairing ATP synthase efficiency
- downregulating PGC-1α
- increasing mitochondrial ROS production
- damaging mitochondrial DNA (mtDNA)
Healthy cortisol rhythm supports mitochondrial health; dysrhythmia erodes it.
2. The Sympathetic–Mitochondrial Overdrive Loop
Chronic stress elevates sympathetic tone, increasing norepinephrine-driven metabolic acceleration.
Mitochondria attempt to match this demand by increasing oxidative phosphorylation.
But without adequate recovery time or rhythmic hormonal control:
- ATP supply falls short
- ROS increases
- antioxidant capacity becomes insufficient
- mitochondrial membranes become leaky
- calcium homeostasis destabilizes
This creates a metabolic phenotype where the body is overactivated but underpowered.
Thus emerges the “metabolic mismatch” at the core of chronic stress:
high energy demand + impaired energy production = persistent exhaustion
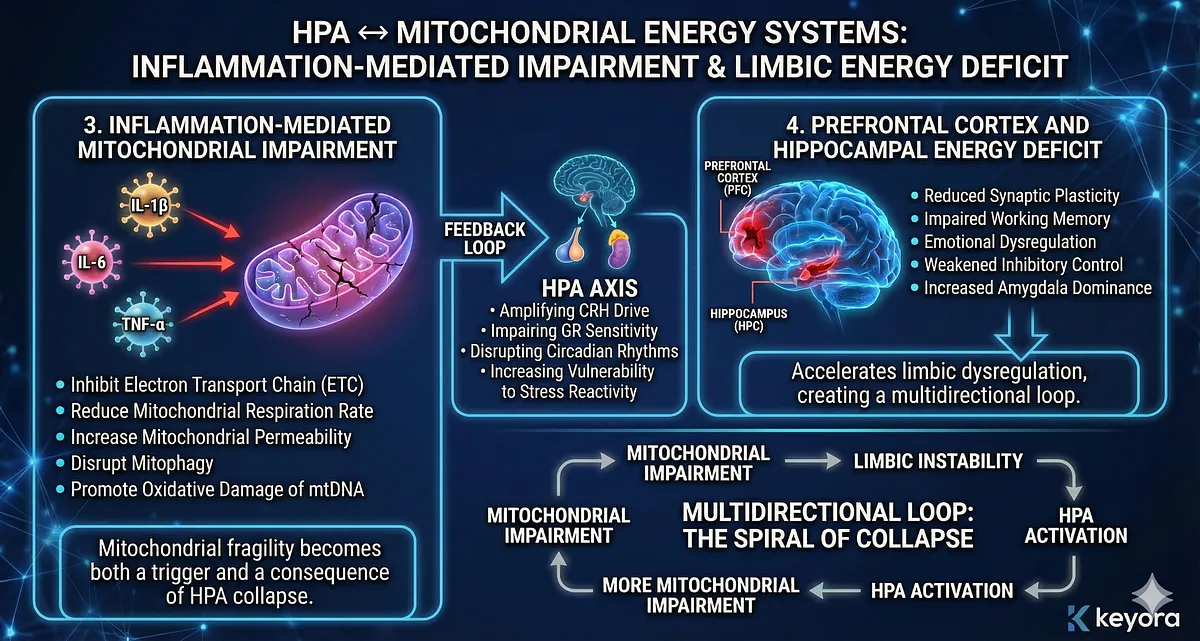
3. Inflammation-Mediated Mitochondrial Impairment
Inflammatory cytokines (IL-1β, IL-6, TNF-α), activated microglia, and NF-κB signaling interfere directly with mitochondrial function:
- inhibit electron transport chain (ETC) complexes
- reduce mitochondrial respiration rate
- increase mitochondrial permeability transition
- disrupt mitophagy
- promote oxidative damage of mtDNA
This inflammation-induced mitochondrial distress feeds back into the HPA axis by:
- amplifying CRH drive
- impairing GR sensitivity
- disrupting circadian rhythms
- increasing vulnerability to stress reactivity
Mitochondrial fragility becomes both a trigger and a consequence of HPA collapse.
4. Prefrontal Cortex and Hippocampal Energy Deficit
The PFC and hippocampus are among the body’s highest energy-demand regions.
Under chronic stress, mitochondrial impairment in these structures leads to:
- reduced synaptic plasticity
- impaired working memory
- emotional dysregulation
- weakened inhibitory control
- increased amygdala dominance
This directly accelerates limbic dysregulation described in Section VI, creating a multidirectional loop:
mitochondrial impairment → limbic instability → HPA activation → more mitochondrial impairment
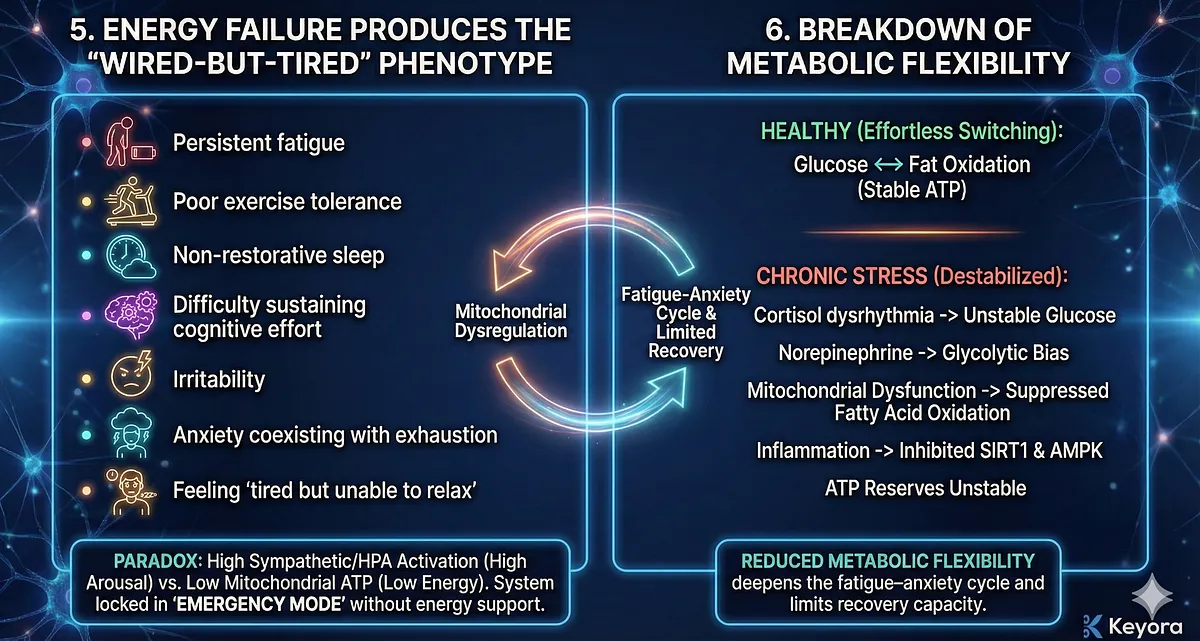
5. Energy Failure Produces the “Wired-but-Tired” Phenotype
The hallmark symptoms of chronic stress reflect mitochondrial dysregulation:
- persistent fatigue despite rest
- poor exercise tolerance
- sleep that does not restore energy
- difficulty sustaining cognitive effort
- irritability under low cognitive load
- anxiety coexisting with exhaustion
- feeling “tired but unable to relax”
This paradox arises when sympathetic and HPA activation are high but mitochondrial ATP production is low.
The system is locked in “emergency mode” without the energy to support it.
6. Breakdown of Metabolic Flexibility
Healthy stress physiology requires effortless switching between metabolic states (glucose ↔ fat oxidation).
Under chronic stress:
- cortisol dysrhythmia destabilizes glucose regulation
- norepinephrine elevates glycolytic bias
- mitochondrial dysfunction suppresses fatty acid oxidation
- inflammation inhibits SIRT1 and AMPK pathways
- ATP reserves become unstable
This reduced metabolic flexibility deepens the fatigue–anxiety cycle and limits recovery capacity.
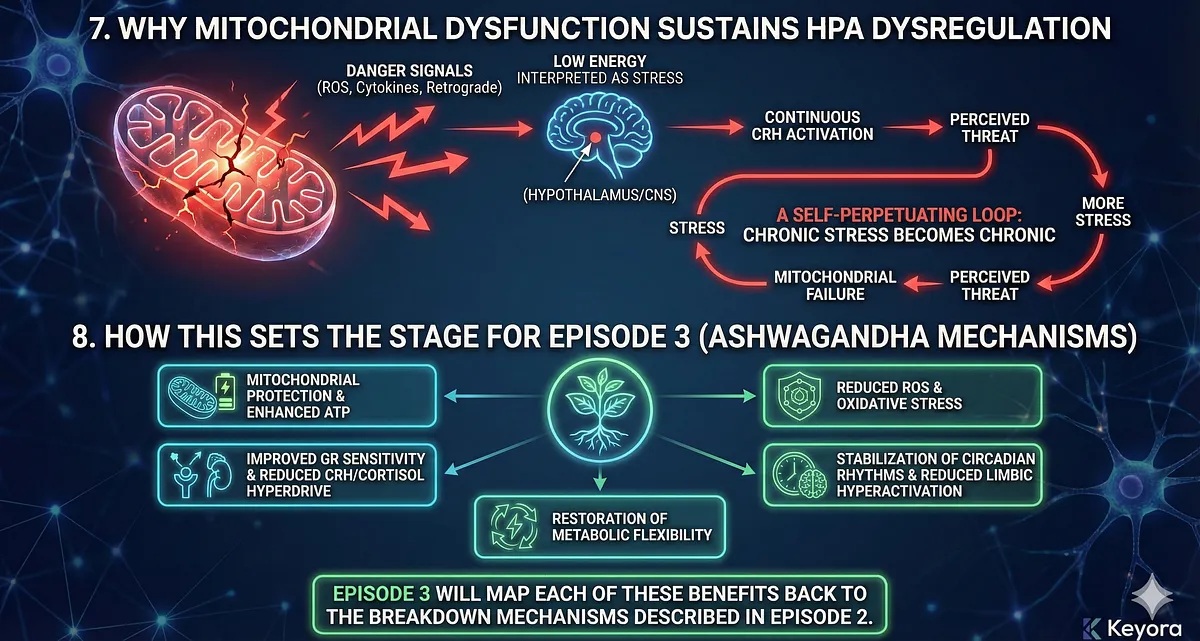
7. Why Mitochondrial Dysfunction Sustains HPA Dysregulation
Mitochondrial impairment sends danger signals back to the CNS through:
- ROS-mediated inflammatory activation
- retrograde mitochondria-to-nucleus communication
- cytokine signaling to hypothalamus
- impaired GR receptor phosphorylation
- disrupted circadian entrainment
Low energy becomes interpreted as stress, generating continuous CRH activation—even in the absence of external threat.
The organism becomes trapped in a loop where:
stress → mitochondrial failure → perceived threat → more stress
This is one of the most important upstream mechanisms that explains why chronic stress becomes chronic.
8. How This Sets the Stage for Episode 3 (Ashwagandha Mechanisms)
The mitochondrial–HPA axis coupling provides the physiological rationale for Ashwagandha’s multi-target benefits:
- mitochondrial protection
- enhanced ATP generation
- reduced ROS
- improved GR sensitivity
- reduction of CRH and cortisol hyperdrive
- stabilization of circadian rhythms
- reduction of limbic hyperactivation
- restoration of metabolic flexibility
Episode 3 will map each of these benefits back to the breakdown mechanisms described in Episode 2.
– HPA axis and mitochondria form a tightly linked energy-allocation system.
– Acute cortisol boosts ATP output; chronic cortisol damages mitochondria.
– Sympathetic overactivation creates high demand + impaired energy production.
– Inflammation disrupts ETC complexes, mtDNA, and mitophagy → energy deficit.
– PFC and hippocampus are especially vulnerable → emotional and cognitive instability.
– Energy failure produces the “wired-but-tired” paradox: high arousal, low energy.
– Loss of metabolic flexibility deepens stress reactivity and fatigue.
– Mitochondrial impairment feeds back to the CNS → CRH activation and circadian disruption.
Episode 2 – Final Conclusion & Cross-Episode Integration
Reconstructing the Stress Architecture:
From HPA Breakdown to Systems-Level Failure
Episode 2 has dissected the HPA axis not as a single hormonal loop, but as a multi-layered regulatory system embedded within circadian timing, immune signaling, emotional circuitry, and mitochondrial energy networks.
Rather than viewing cortisol as a simple stress marker, this chapter reframed the HPA axis as the central integrator of biological time, threat evaluation, metabolic allocation, and neuro-immune coordination.
Chronic stress disrupts these networks simultaneously, transforming an adaptive architecture into a self-perpetuating failure mode.
Across the seven sections, a coherent systems model emerges.
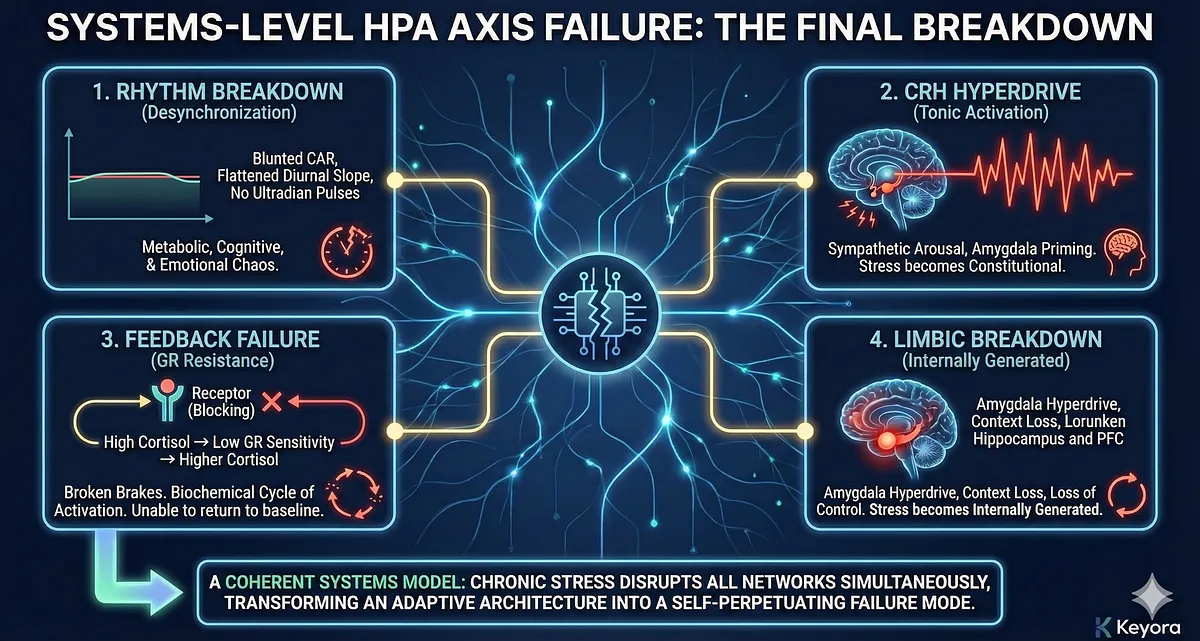
1. HPA Dysregulation Begins With Rhythm Breakdown
Cortisol’s biological power lies in its timing, not its magnitude.
When the cortisol awakening response weakens, the diurnal slope flattens, and ultradian pulses collapse, the entire organism loses its metabolic, cognitive, and emotional rhythm.
This desynchronization is the earliest – and most clinically predictive – marker of impending stress pathology.

2. CRH Hyperdrive Converts Stress From an Event Into a State
The hypothalamus shifts from adaptive responsivity to tonic activation.
Sustained CRH output amplifies sympathetic arousal, primes the amygdala for overreaction, and maintains constant vigilance even without external threat. Chronic stress is no longer “reactional,” but constitutional.
3. Feedback Failure Through GR Resistance Removes the Brakes
Glucocorticoid receptor resistance breaks the negative feedback loop that once terminated stress.
This creates a biochemical cycle of:
high cortisol → low GR sensitivity → higher cortisol, locking the system into ongoing activation and rendering the organism unable to return to baseline.
4. Limbic Circuit Breakdown Makes Stress Self-Generating
The amygdala–PFC–hippocampus loop loses structural balance:
- Amygdala hyperactivation (alarm amplification)
- Hippocampal atrophy (context loss)
- PFC suppression (loss of control)
As emotional regulation collapses, limbic circuits begin driving HPA activity, not responding to it.
Stress becomes internally generated.
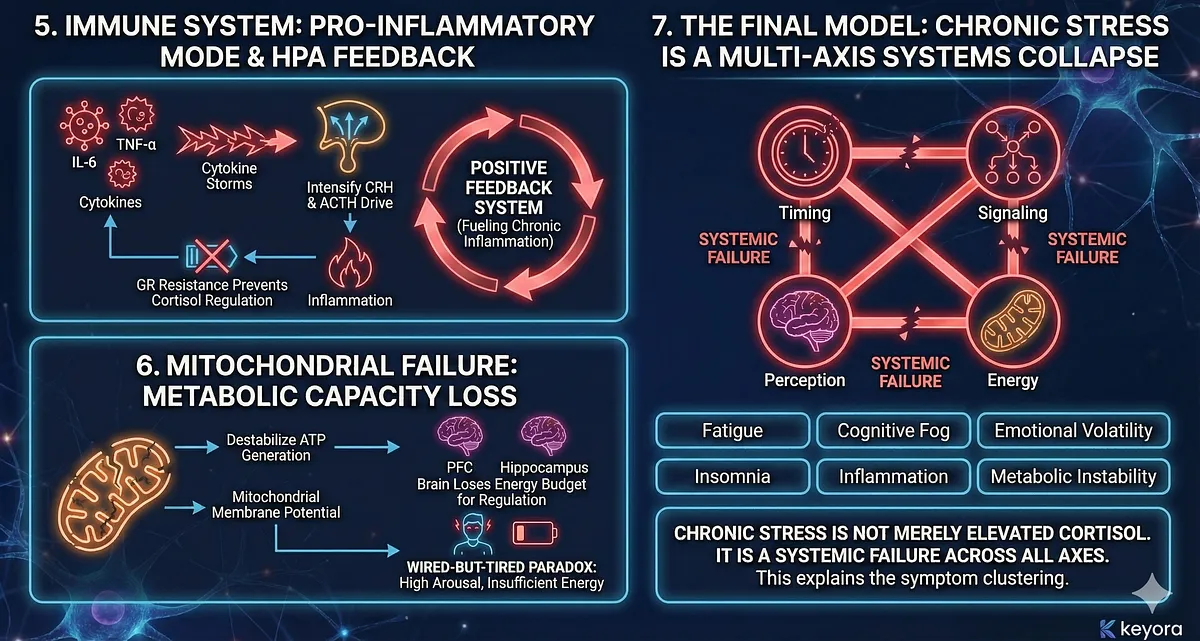
5. Immune System Enters a Pro-Inflammatory Mode That Feeds Back Into HPA Activation
Cytokine storms of IL-6, TNF-α, and IL-1β signal directly to the hypothalamus, intensifying CRH and ACTH drive.
Meanwhile, GR resistance prevents cortisol from regulating inflammation.
The endocrine–immune loop becomes a positive feedback system, fueling chronic inflammatory stress states.
6. Mitochondrial Failure Removes the Metabolic Capacity for Resilience
Chronic cortisol exposure, sympathetic overdrive, and inflammation destabilize ATP generation and mitochondrial membrane potential. The brain, especially the PFC and hippocampus, loses its energy budget for stress regulation.
This produces the “wired-but-tired” paradox – an organism stuck in high arousal with insufficient energy to support it.
7. The Final Model: Chronic Stress Is a Multi-Axis Systems Collapse
Taking these mechanisms together, Episode 2 demonstrates that chronic stress is not merely elevated cortisol or emotional overload.
It is: a systemic failure of timing, signaling, perception, and energy.
This model explains why individuals experiencing chronic stress present with a constellation of symptoms – fatigue, cognitive fog, emotional volatility, insomnia, inflammation, metabolic instability – and why these symptoms cluster across psychiatric, metabolic, and immune conditions.
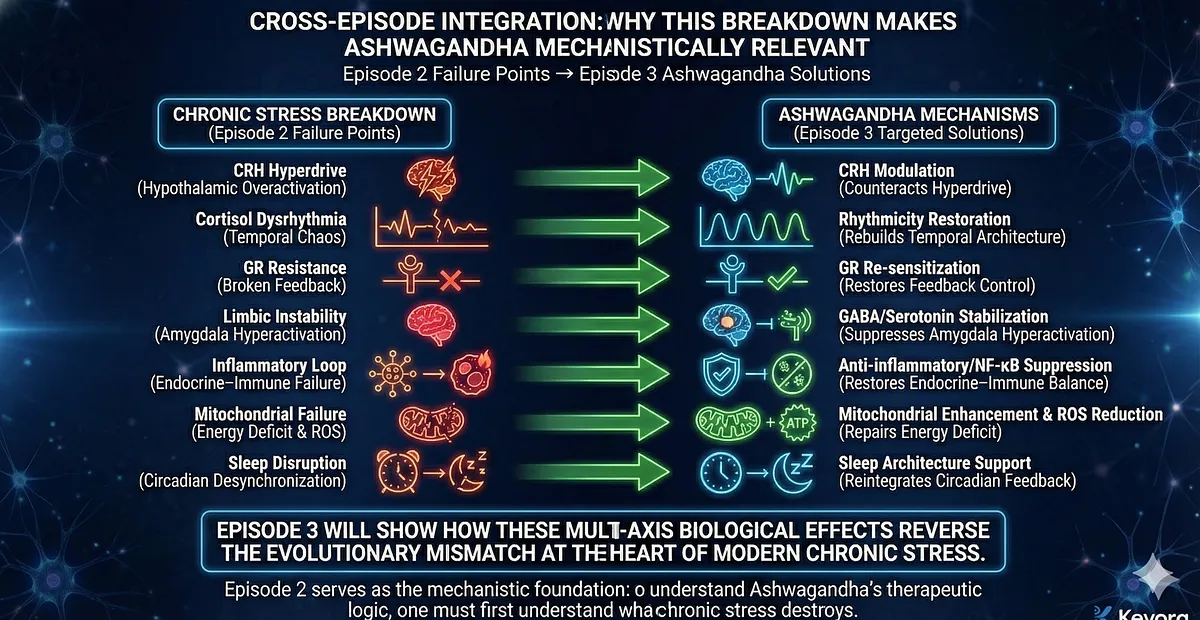
Cross-Episode Integration:
Why This Breakdown Makes Ashwagandha Mechanistically Relevant
The collapse described in Episode 2 sets the biological rationale for Episode 3.
Ashwagandha’s molecular actions map precisely onto the failure points outlined above:
- CRH modulation → counteracts hypothalamic hyperdrive
- cortisol rhythmicity restoration → rebuilds temporal architecture
- GR re-sensitization → restores feedback control
- GABAergic and serotonergic stabilization → suppresses amygdala hyperactivation
- anti-inflammatory and NF-κB suppression → restores endocrine–immune balance
- mitochondrial enhancement & ROS reduction → repairs the energy deficit
- sleep architecture support → reintegrates circadian feedback into HPA regulation
Episode 3 will show how these multi-axis biological effects make Ashwagandha uniquely suited to reverse the evolutionary mismatch at the heart of modern chronic stress.
Episode 2 thus serves as the mechanistic foundation:
to understand Ashwagandha’s therapeutic logic, one must first understand what chronic stress destroys.
– Chronic stress is a systems-level breakdown across circadian, limbic, immune, and mitochondrial axes.
– Loss of cortisol rhythm disrupts biological timing and destabilizes cognitive/emotional function.
– CRH hyperdrive and GR resistance trap the HPA axis in tonic activation.
– Limbic circuits shift from regulated to self-generating, amplifying emotional stress.
– Immune cytokines drive HPA activation while cortisol loses regulatory power.
– Mitochondrial failure reduces energy availability for stress resilience.
– This multi-axis collapse explains the global symptom pattern of chronic stress.
– Sets the foundation for Episode 3: how Ashwagandha restores the damaged architecture.
By Keyora Research Notes Series
This article contributes to Keyora’s ongoing scientific documentation series, which systematically outlines the conceptual foundations, mechanistic pathways, and empirical evidence informing our research and development approach.
ORCID: 0009–0007–5798–1996
